Clear Sky Science · en
Prediction of estimated glomerular filtration rate slope and kidney prognosis of patients with chronic kidney disease
Why this matters for everyday health
Chronic kidney disease often progresses silently for years before symptoms appear, yet it can lead to heart problems, the need for dialysis, and even death. Family doctors see most patients long before they reach a kidney specialist, but they have few simple tools to look into the future and judge whose kidneys are likely to worsen quickly. This study from Japan introduces a machine-learning tool that uses routine clinic data from a single visit to predict how fast kidney function will decline over the next few years, helping doctors act earlier and more confidently.
Kidneys under quiet strain
Chronic kidney disease affects tens of millions of adults in Japan alone and is tightly linked to heart disease and early death worldwide. Because there are far more patients than kidney specialists, most people with mild to moderate damage are followed by primary care doctors. These doctors rely on a blood test called estimated glomerular filtration rate, or eGFR, which reflects how well the kidneys filter waste. Until now, most risk tools focused on whether a patient would eventually reach kidney failure, a distant outcome. The authors argue that the pace of change in eGFR over time—the eGFR “slope”—is a more practical yardstick for day-to-day care, because it captures the speed of decline rather than a single yes-or-no event.
Turning routine clinic data into a time machine
The team drew on J-CKD-DB-Ex, Japan’s largest electronic health record database devoted to kidney disease, which contains information on about 250,000 patients from 15 university hospitals. From this pool they selected 10,474 adults with chronic kidney disease who were cared for as outpatients and had at least four eGFR measurements spread over several years. For each person, they collected basic information that any clinic could obtain: age, sex, blood values such as creatinine, albumin, sodium, and potassium, urine protein results, common diagnoses like diabetes and high blood pressure, and whether certain kidney-protective drugs were prescribed. Using eGFR values over three years, they calculated each patient’s true eGFR slope—the rate at which kidney function rose or fell per year.
Putting machine learning to the test
The researchers then compared three ways of predicting each patient’s eGFR slope. A traditional approach simply extended past eGFR readings into the future using straight-line statistics. Two modern machine-learning methods, called LightGBM (a type of decision-tree boosting) and LSTM (a neural network geared to sequences), instead learned patterns linking single-visit information to later kidney decline. The data were split so that one portion trained the models and another, never seen during training, tested how well they performed. Accuracy was judged by how closely predicted slopes matched actual slopes, summarized as an average error. The simple statistical method missed the mark by a wide margin, while both machine-learning models were far more precise, with LightGBM performing best.
How accurate is “accurate enough” for real patients?
In practical terms, the LightGBM model misestimated the yearly rate of kidney function change by about 3 units on average, compared with more than 15 units for the simple method. Over three years, this translates to roughly a 9-unit typical uncertainty in predicted kidney function, and for most patients the error would stay within about 20 units. While not perfect, this level of precision is tight enough to help decide when to intensify treatment or refer someone to a kidney specialist. Importantly, the model works even if only a single eGFR value and standard lab and clinical information are available, a common situation in primary care where regular long-term testing may be patchy.
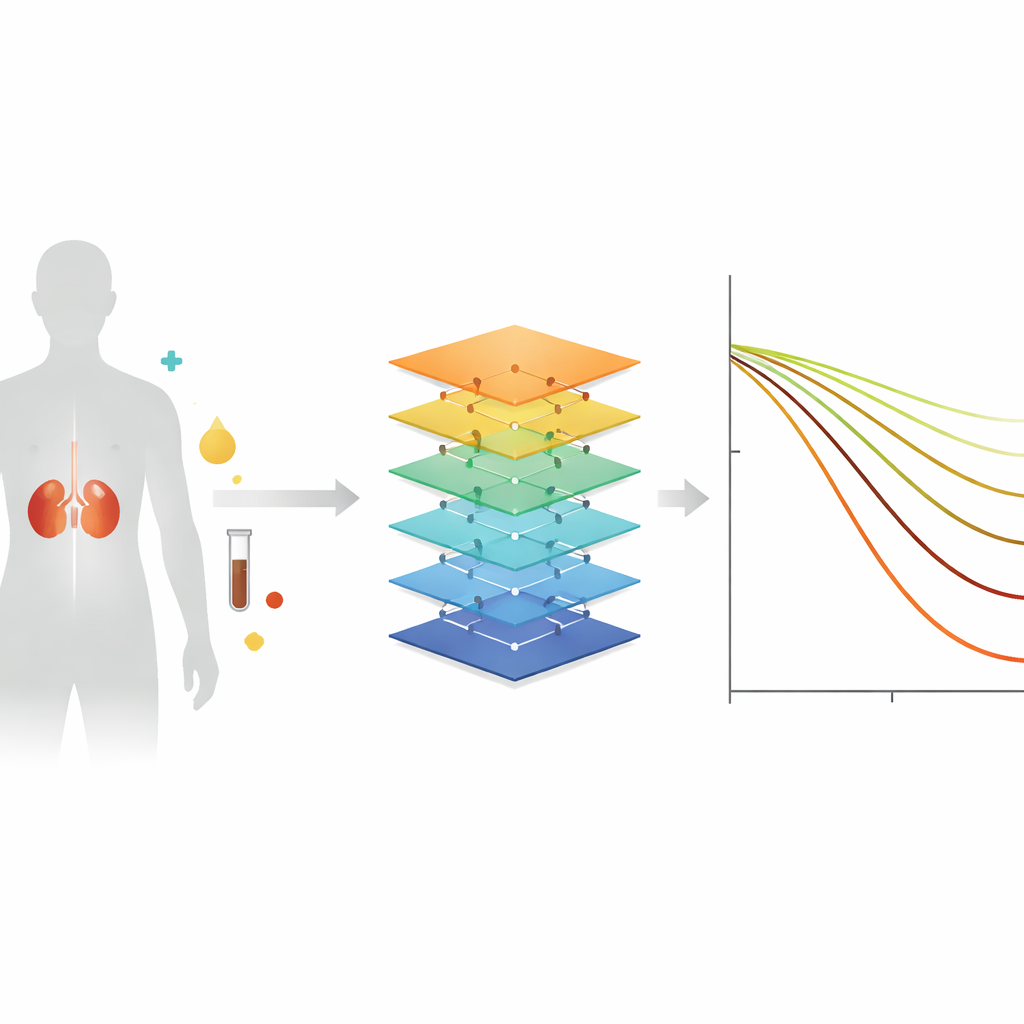
From complex code to a simple clinic screen
To make the tool usable outside research centers, the team wrapped the best-performing model in a web-based application. A clinician can enter a patient’s age, sex, lab results, and key diagnoses, and the tool immediately draws a projected line of kidney function over the next three years. This visualization turns abstract numbers into a clear picture of whether function is stable, gently drifting downward, or plunging at a worrying rate. By highlighting patients whose kidneys may deteriorate quickly, the system encourages earlier lifestyle counseling, medication adjustments, and timely referral to specialists, while also helping to reassure those with a more favorable outlook.
What this means for people with kidney disease
This study shows that a carefully trained machine-learning model can act like a short-range forecast for kidney health, using only information that most clinics already collect. While the tool does not replace medical judgment and still needs testing in more diverse groups, it offers a way for front-line doctors to spot high-risk patients years before crisis strikes. For people living with chronic kidney disease, that earlier warning could mean more time to slow the damage, avoid or delay dialysis, and stay healthier overall.
Citation: Nagasu, H., Nakashima, T., Ihara, K. et al. Prediction of estimated glomerular filtration rate slope and kidney prognosis of patients with chronic kidney disease. Sci Rep 16, 8883 (2026). https://doi.org/10.1038/s41598-026-38246-8
Keywords: chronic kidney disease, kidney function prediction, machine learning in medicine, primary care tools, eGFR slope