Clear Sky Science · en
Thermal and filler concentration modulation of charge transport mechanism and dielectric properties in high-entropy oxide (CoCrFeNiMn)3O4-acrylic polymer composite
Smarter Materials for Storing Electric Energy
Modern electronics—from electric cars to wearables—depend on materials that can safely store and release electric energy in compact spaces. This paper explores a new class of “mix-and-match” ceramic–plastic materials that aim to make capacitors smaller, more efficient, and more stable at high temperatures. By blending a flexible acrylic plastic with a complex metal-oxide powder called a high‑entropy oxide, the researchers show how tweaking temperature and filler content can finely tune how well the material stores charge.
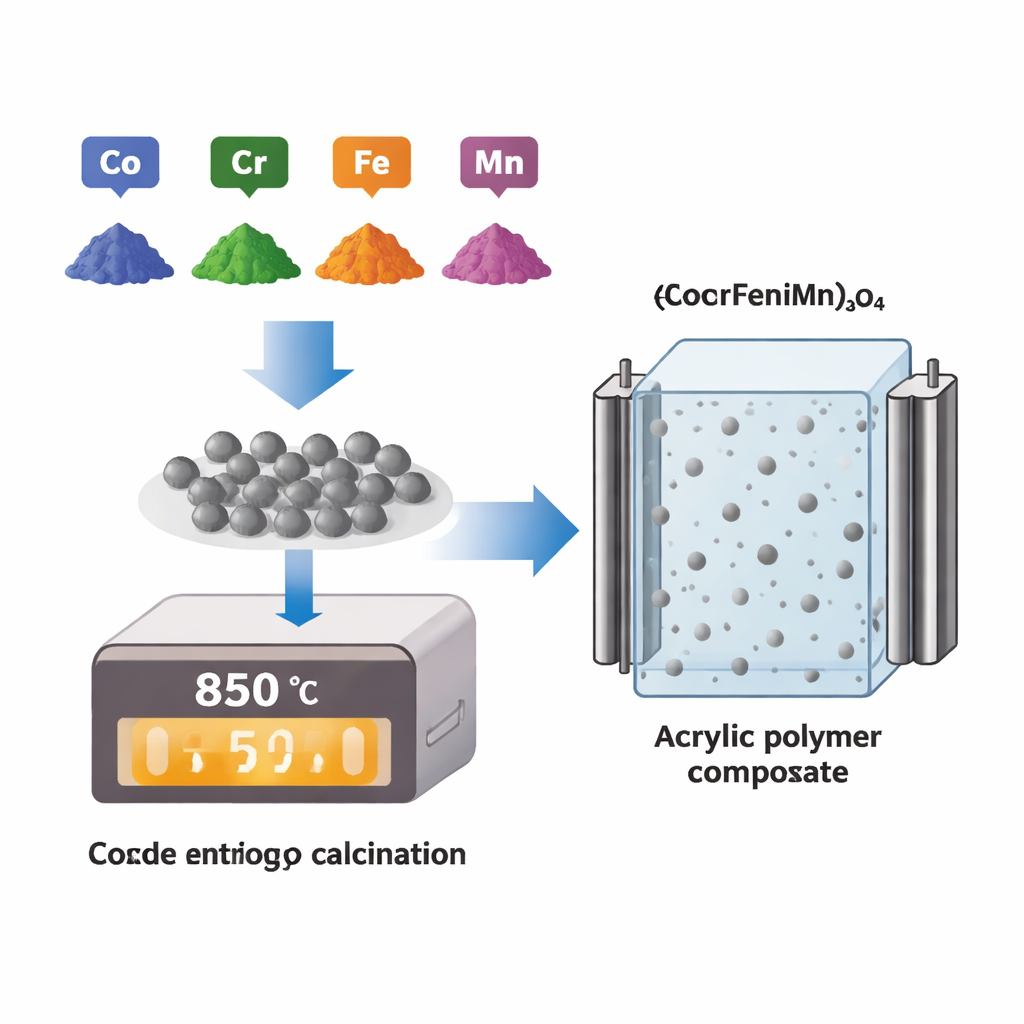
Building a Hybrid Ceramic–Plastic
The team started by creating a special ceramic powder made from five different metal oxides containing cobalt, chromium, iron, nickel, and manganese. When these are mixed and heated together, they form a single, stable crystal structure known as a high‑entropy oxide. This structure is unusually robust because many different metal atoms share the same lattice in a nearly random way, which stabilizes it even at high temperatures. The powder was calcined at 850 °C to produce uniform particles, then carefully ground and sieved so the grains were of similar size. In the next step, the powder was blended into a commercial acrylic polymer at several loadings—1, 3, 5, 10, and 15 weight percent—and hot-pressed into solid discs, creating a family of composite samples.
Checking That the Mix Plays Nicely Together
Before probing electrical behavior, the researchers confirmed that the ingredients stayed structurally sound and chemically distinct. Electron microscopy showed that the high‑entropy oxide particles were distributed throughout the plastic and that the different metals were spread fairly evenly inside each grain. X‑ray diffraction confirmed that the ceramic kept its single spinel phase after processing, while the polymer remained largely amorphous. Infrared spectroscopy indicated that no new chemical bonds formed between the ceramic and the acrylic; instead, the two phases coexist physically. This is important for capacitor applications, where one often wants a robust ceramic filler embedded in a flexible, electrically insulating host.
How Charge Moves and Collects Inside
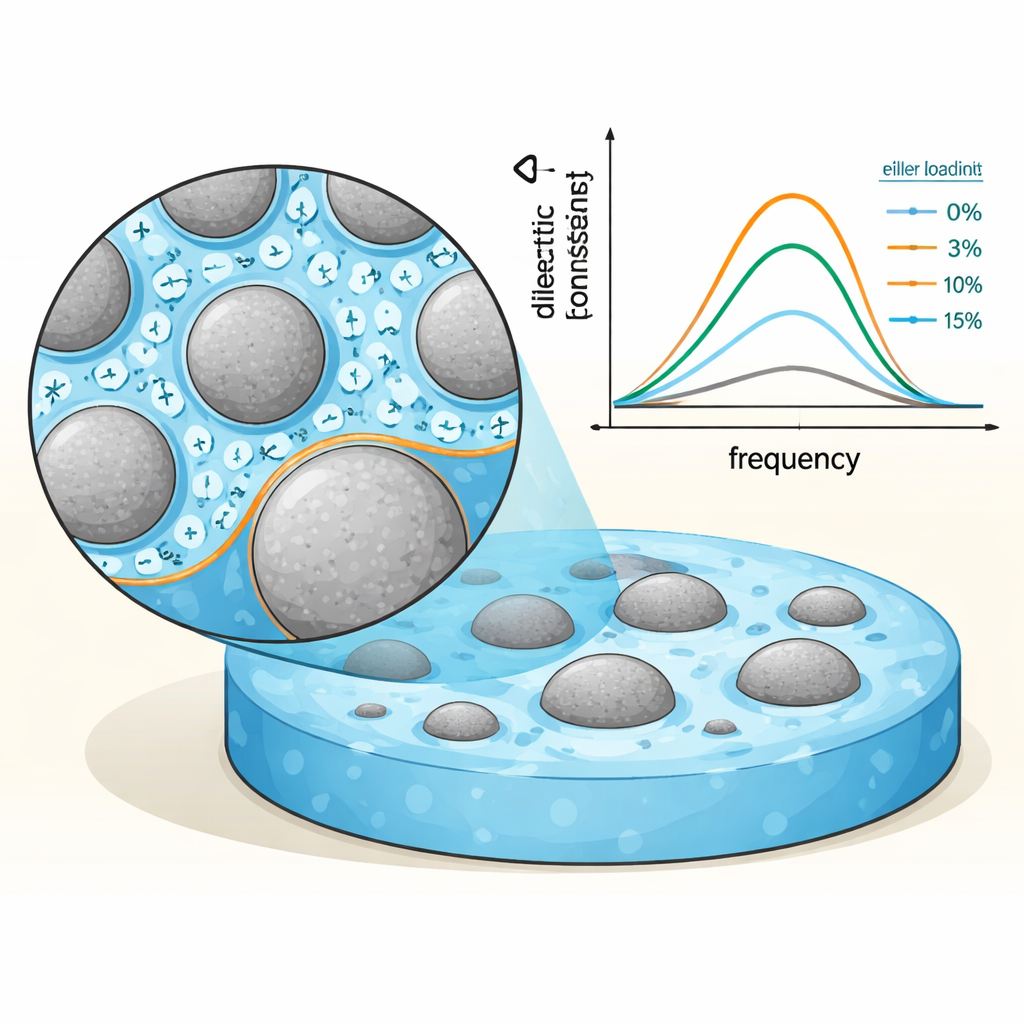
To understand how these composites store and lose electric energy, the team used broadband dielectric spectroscopy, applying an alternating electric field over a wide range of frequencies and temperatures (from −90 to 90 °C). They tracked both how much energy the material can store (the dielectric constant) and how much is lost as heat (dielectric loss and conductivity). At low ceramic content and moderate temperatures, the high‑entropy oxide particles introduce extra interfaces inside the plastic. Charges tend to pile up at these boundaries, a process called interfacial polarization, which boosts the dielectric constant. As temperature rises, charge carriers gain energy, hop more easily between atomic sites of different metals, and form “polarons” (charges coupled to local lattice distortions). This hopping behavior changes the way current flows, shifting from simple tunneling at low temperatures to more thermally driven hopping at higher temperatures.
Finding the Sweet Spot in Filler Content
The most striking result is that the dielectric response does not simply rise with more ceramic. Instead, there is an optimal filler concentration near 10 percent by weight. Around this level, a near‑continuous network of particles forms inside the plastic, dramatically enhancing both the dielectric constant and conductivity—a behavior linked to the “percolation threshold,” where separate islands of filler start to connect. Below this threshold, too few particles are close enough to cooperate; above it, at 15 percent loading, overly connected paths act more like leaky channels, so the ability to store energy drops again and losses increase. The relaxation peaks in the data shift to higher frequencies with temperature, meaning the material’s internal dipoles can reorient more quickly as they receive more thermal energy.
What This Means for Future Electronics
Overall, the study shows that by precisely choosing how much high‑entropy oxide to add and at what temperature the device operates, engineers can tune a simple acrylic plastic into a highly responsive dielectric material. The composite with about 10 percent ceramic filler offers the best balance: a high capacity to store charge, reasonable losses, and stability over a wide temperature range. Because these behaviors are rooted in the flexible electronic structure of the multi‑metal oxide and the way charges move through and between the particles, the same design ideas could guide future hybrid materials for capacitors, power electronics, and energy storage systems that are smaller, more robust, and better suited to demanding environments.
Citation: Daradkeh, S.I., Alsoud, A., Spusta, T. et al. Thermal and filler concentration modulation of charge transport mechanism and dielectric properties in high-entropy oxide (CoCrFeNiMn)3O4-acrylic polymer composite. Sci Rep 16, 7309 (2026). https://doi.org/10.1038/s41598-026-38245-9
Keywords: high entropy oxide, polymer composite, dielectric materials, energy storage, charge transport