Clear Sky Science · en
Design, synthesis, characterization, pharmacological evaluation and in silico ADMET and molecular docking and dynamics simulations of a novel series of N-substituted pyrazole from chalcone derivatives
New Hope Against Stubborn Infections
Antibiotic resistance and chronic inflammation often go hand in hand, making common infections harder to treat and pain more difficult to control. This study explores a fresh family of lab‑made molecules that aim to tackle both problems at once: they are designed to kill harmful bacteria while also calming inflammation, all with fewer stomach side effects than some current drugs.
Why New Medicines Are Needed
Decades of heavy antibiotic use have helped bacteria evolve ways to dodge many of our best drugs. At the same time, widely used anti‑inflammatory medicines, such as some painkillers, can irritate the stomach and cause ulcers when taken for long periods. The researchers set out to build “two‑in‑one” compounds that could fight a broad range of bacteria and reduce swelling and pain, while being gentler on the digestive system.
Building the New Molecules
The team focused on two chemical building blocks that already appear in many successful medicines: pyrazoles and chalcones. By cleverly linking these cores and extending them with extra ring systems, they created a small library of related compounds. Subtle changes—such as adding a chlorine atom, a sulfur‑containing ring, or an antipyrine fragment—allowed them to tune how each molecule behaved. Standard laboratory techniques confirmed that the new structures were made correctly and were pure.
Testing Germ‑Killing Power and Pain Relief
The compounds were then tested against six different bacterial species, including familiar troublemakers like E. coli, Staphylococcus aureus, and Klebsiella pneumoniae. Several candidates, especially those labeled 4c, 5c, and 12, stopped bacterial growth at lower doses than the widely used antibiotic levofloxacin, or worked just as well. In animal tests of swelling in the paw, many of the compounds also eased inflammation strongly. Notably, 5c and 12 slightly outperformed the prescription pain and arthritis drug celecoxib in reducing swelling over several hours.
Safer for the Stomach
Powerful anti‑inflammatory drugs often carry a hidden cost: they can damage the stomach lining and lead to ulcers. To check this risk, the team examined the stomachs of rats given the most promising new compounds. While celecoxib produced a noticeable level of damage, leading molecules such as 9, 5b, 5c, 10, 11, and especially 12 caused far milder changes. In parallel computer‑based safety checks, one standout compound, 4c, showed a good balance of size, fat‑solubility, and predicted absorption, along with low chances of genetic damage or heart‑related side effects.
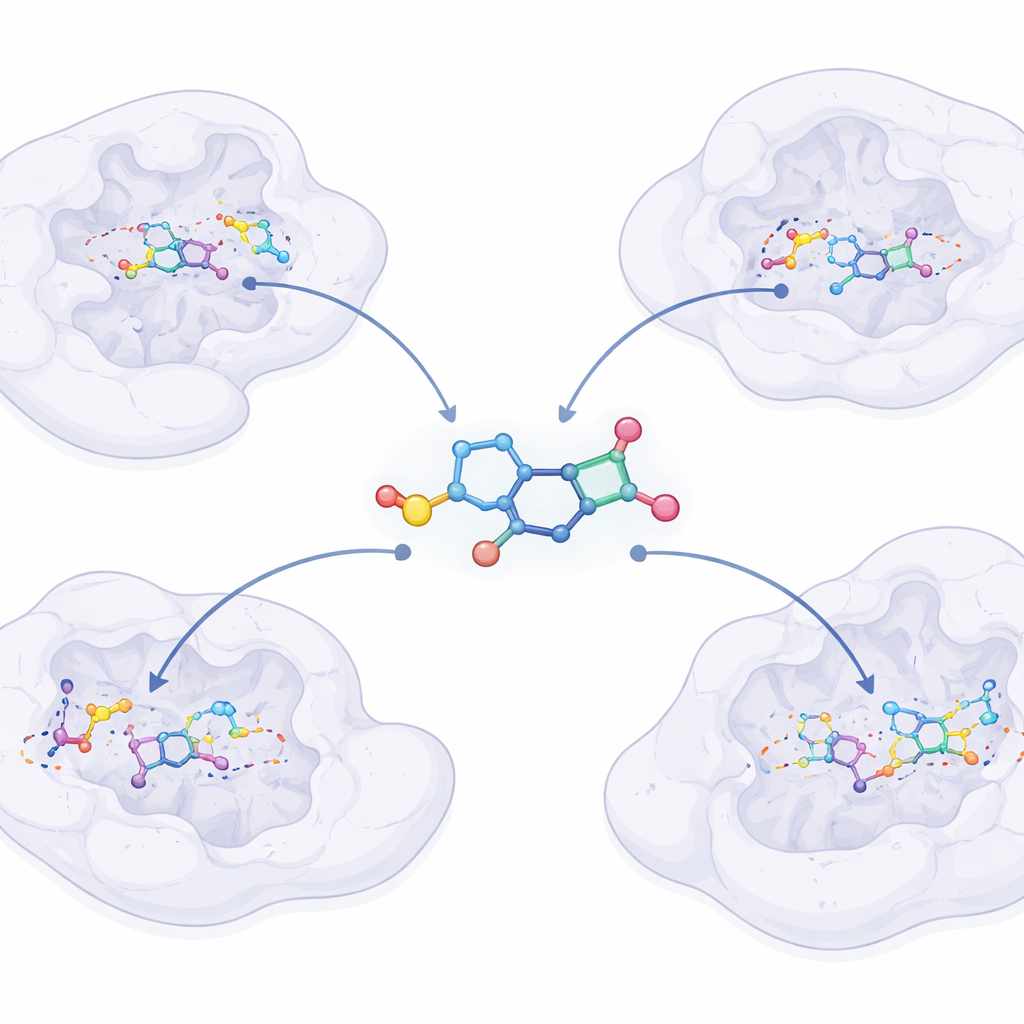
Peeking Inside at the Molecular Workings
Using advanced computer simulations, the researchers asked how these molecules might latch onto their bacterial targets. They modeled how the new compounds sit inside the pockets of several essential bacterial proteins, including enzymes that twist and copy DNA, build cell components, and help germs invade the body. The top molecules formed tight, stable fits at these sites, often gripping key amino acids more strongly than levofloxacin. Further simulations over time suggested that these bindings remain stable in a watery, dynamic environment that resembles the inside of a cell.
What This Could Mean for Future Treatments
Altogether, the study introduces a new class of small molecules that combine strong antibacterial action with potent anti‑inflammatory effects and reduced risk of stomach ulcers. While these compounds are still at an early stage and have only been tested in cells, animals, and computer models, they offer a promising starting point for future drugs that can both clear infections and soothe the body’s response. With further refinement and testing, such dual‑action agents could become valuable tools in treating tough, drug‑resistant infections without adding to patients’ discomfort.
Citation: Hafez, H.N., Otaif, H.Y., Alshammari, B.H. et al. Design, synthesis, characterization, pharmacological evaluation and in silico ADMET and molecular docking and dynamics simulations of a novel series of N-substituted pyrazole from chalcone derivatives. Sci Rep 16, 7931 (2026). https://doi.org/10.1038/s41598-026-38237-9
Keywords: antibiotic resistance, dual-action drugs, pyrazole chalcone, anti-inflammatory agents, drug design