Clear Sky Science · en
PERK inhibition attenuates multi-program cell death through Nrf2/HO-1 activation in diabetic retinopathy with integrated proteomics and functional validation in HRECs
Why protecting sight in diabetes matters
For millions of people living with diabetes, one of the most feared complications is the gradual loss of vision known as diabetic retinopathy. This damage to the light-sensing tissue at the back of the eye often starts silently, long before symptoms appear. The study behind this article digs into what actually kills the tiny blood vessel cells that keep the retina healthy—and tests a new way to protect them. By understanding and calming the cell “stress responses” triggered by high blood sugar, researchers hope to slow or even prevent blindness in people with diabetes.
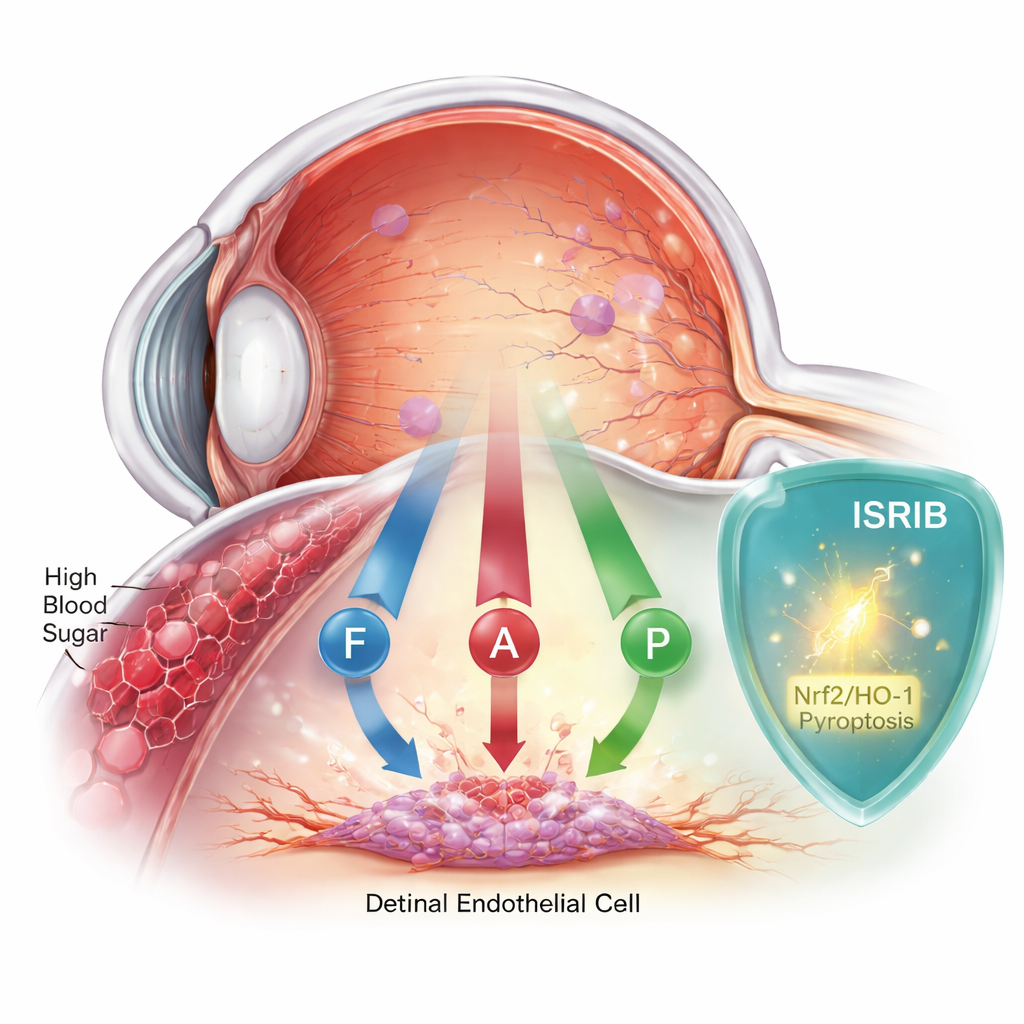
When blood sugar quietly injures the eye
Diabetic retinopathy begins when high blood sugar harms the delicate inner lining of retinal blood vessels. These cells form a barrier that keeps the retina nourished and protected. For years, scientists thought that a single form of cell death, called apoptosis, was mainly to blame. But mounting evidence suggests the story is more complicated. This team focused on human retinal endothelial cells grown in the lab and exposed them to sugar levels similar to those seen in poorly controlled diabetes, then watched how the cells responded over two days.
Three ways cells can die at once
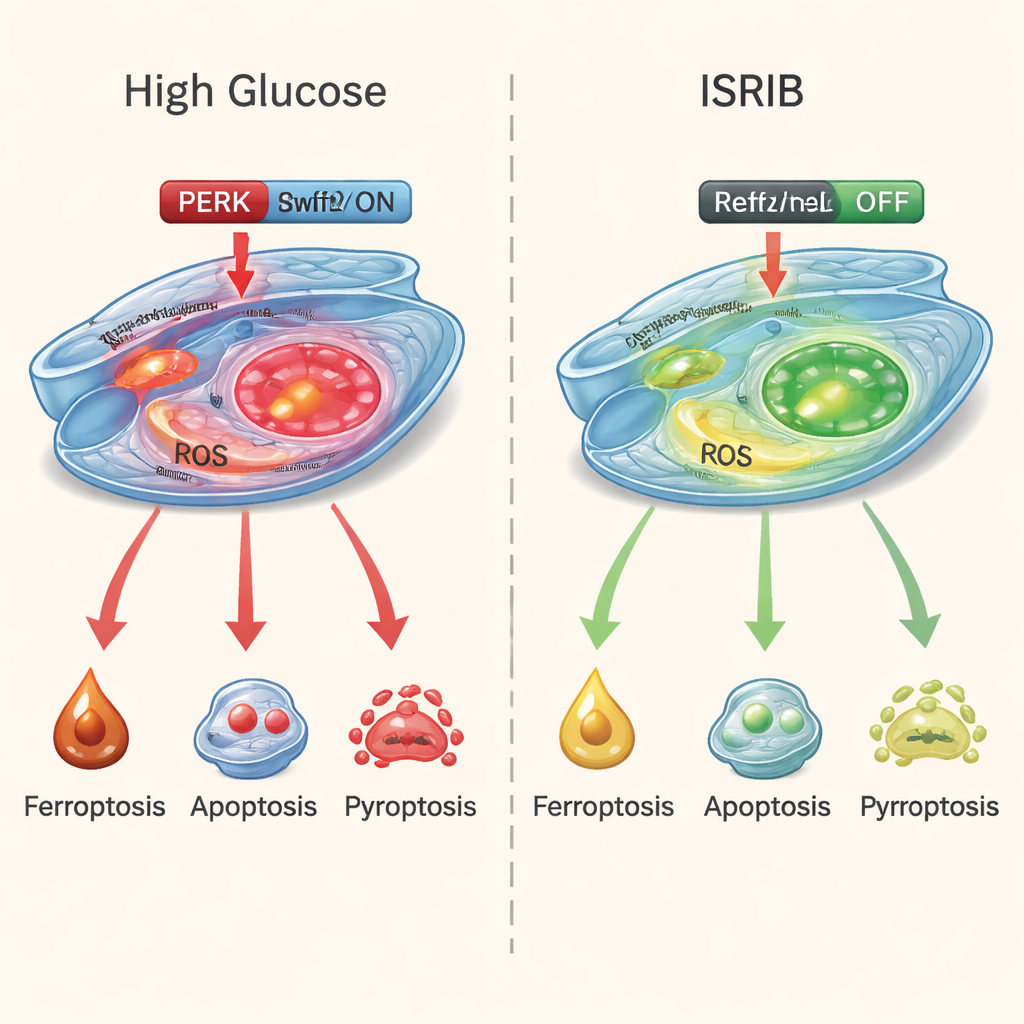
Using a powerful protein-measuring technique called proteomics, the researchers found that high sugar levels disturbed hundreds of proteins, especially those involved in stress responses, metabolism, and iron handling. Follow-up tests showed that the cells were not just dying one way, but three: classic programmed death (apoptosis), a fiery, inflammation-linked form (pyroptosis), and a newer iron-driven form called ferroptosis. Markers of oxidative damage, iron overload, and depleted antioxidants all rose, while protective enzymes fell. Together, these changes painted a picture of cells overwhelmed by stress, inflammation, and toxic build-up of reactive molecules.
A stress switch at the heart of the problem
The team then zeroed in on a key stress sensor inside cells called PERK, which sits on the membrane of the endoplasmic reticulum, a structure that helps fold and process proteins. Under high sugar conditions, PERK became overactive, acting like a stuck emergency alarm that keeps blaring. This appeared to feed into all three death programs. At the same time, a natural defense system that usually helps cells cope with oxidative damage—the Nrf2/HO-1 antioxidant pathway—was weakened. In simple terms, the danger signals were turned up while the internal fire extinguishers were turned down.
Turning down the stress, turning up protection
To see if they could break this destructive cycle, the researchers treated the high-sugar cells with ISRIB, a small molecule that effectively dampens PERK’s stress signaling. The results were striking. ISRIB improved cell survival, reduced markers of iron-driven damage and oxidative stress, and lowered chemical signs of both apoptosis and pyroptosis. At the same time, it boosted the Nrf2/HO-1 defense pathway, helping the cells restore their antioxidant capacity. A separate drug that specifically blocks ferroptosis also helped, but ISRIB stood out because it calmed all three death routes at once rather than just one.
What this could mean for people with diabetes
This work suggests that early damage in diabetic retinopathy is not caused by a single “bad actor,” but by a convergence of several self-reinforcing death programs driven by chronic cellular stress. By dialing down the PERK stress switch and reactivating the cell’s own protective systems, drugs like ISRIB might offer a more unified way to keep retinal blood vessels alive. While these experiments were done in cells, not yet in patients, they point toward future treatments that target stress responses inside the eye—potentially adding a new layer of protection alongside blood sugar control and current eye injections to help preserve sight in diabetes.
Citation: Xie, L., Zhang, X., Tian, M. et al. PERK inhibition attenuates multi-program cell death through Nrf2/HO-1 activation in diabetic retinopathy with integrated proteomics and functional validation in HRECs. Sci Rep 16, 7225 (2026). https://doi.org/10.1038/s41598-026-38213-3
Keywords: diabetic retinopathy, retinal cell death, oxidative stress, PERK pathway, Nrf2 HO-1