Clear Sky Science · en
SHH pathway inhibition and astrocyte co-culture induce distinct responses in glioblastoma and cancer stem cells
Why brain tumors are so hard to treat
Glioblastoma is one of the deadliest brain cancers, and even with surgery, radiation, and chemotherapy, only a small fraction of patients live beyond five years. One reason is that these tumors are not made of just one kind of cell. They include a small group of especially stubborn “cancer stem” cells that can survive treatment and help the tumor grow back. This study asks a key question: how do these different tumor cells respond when a growth‑driving signal is blocked, and how does the presence of normal brain support cells called astrocytes change that response?
A closer look at the tumor’s toughest cells
The researchers worked with two main types of cells from a common glioblastoma model: the bulk tumor cells and a tiny subpopulation marked by a protein called CD133, which behave like cancer stem cells. These stem‑like cells divided less, looked smaller and rounder, and formed tighter clusters than the bulk cells. The team also grew the tumor cells together with astrocytes, the star‑shaped cells that help maintain healthy brain tissue. This mixed culture better mimics the real tumor environment, where cancer cells constantly “talk” with their neighbors. 
Blocking a developmental signal that tumors reuse
Many glioblastomas, especially their stem‑like cells, hijack a molecular communication route called the Sonic Hedgehog (SHH) pathway, normally active during brain development. The team used cyclopamine, a compound that blocks a key SHH component, to see how both bulk tumor cells and cancer stem cells respond. They measured activity of SHH‑related genes and proteins, how fast cells moved through the cell cycle, and how often they underwent programmed cell death (apoptosis). Cyclopamine changed SHH signaling in a way that depended strongly on cell type and whether astrocytes were present. Cancer stem cells produced the most SHH signal when grown alone, but this secretion dropped when they were grown with astrocytes and exposed to the drug, suggesting that the surrounding brain cells can dampen the tumor’s chemical “self‑support.”
How the neighborhood shifts growth and death
Beyond SHH itself, the team tracked other gene networks that steer whether cells keep dividing, pause, or begin to specialize. In simple tumor‑only cultures, blocking SHH pushed bulk tumor cells toward a stall in the late stage of division (the G2/M phase), while stem‑like cells tended to accumulate earlier, in the resting or entry phase (G0/G1). When astrocytes were added, these patterns changed: cancer stem cells, which had been more quiescent, shifted toward G2/M arrest, implying a different set of “brakes” being applied in the mixed environment. The researchers also saw that signals linked to cell fate and tissue shaping—such as WNT, BMP, and TGF‑β pathways—were tuned differently in bulk cells and stem‑like cells when astrocytes were present, meaning the microenvironment can tilt cells toward either continued stem‑like behavior or a more differentiated, less aggressive state.
Triggering cell death in the most resistant cells
The study further examined how readily cells underwent apoptosis. Genetic readouts and a DNA‑damage–sensing test (TUNEL assay) showed that cyclopamine favored an “external” route to cell death, driven by a protein called CASP8, particularly in the cancer stem‑cell population. Interestingly, in tumor cells grown alone, overall cell death sometimes decreased after SHH blockade, suggesting that these cells could activate backup survival strategies. By contrast, when stem‑like cells were co‑cultured with astrocytes, cyclopamine increased markers of apoptosis, indicating that the surrounding brain cells can make these otherwise stubborn cells more vulnerable once SHH signaling is blocked. 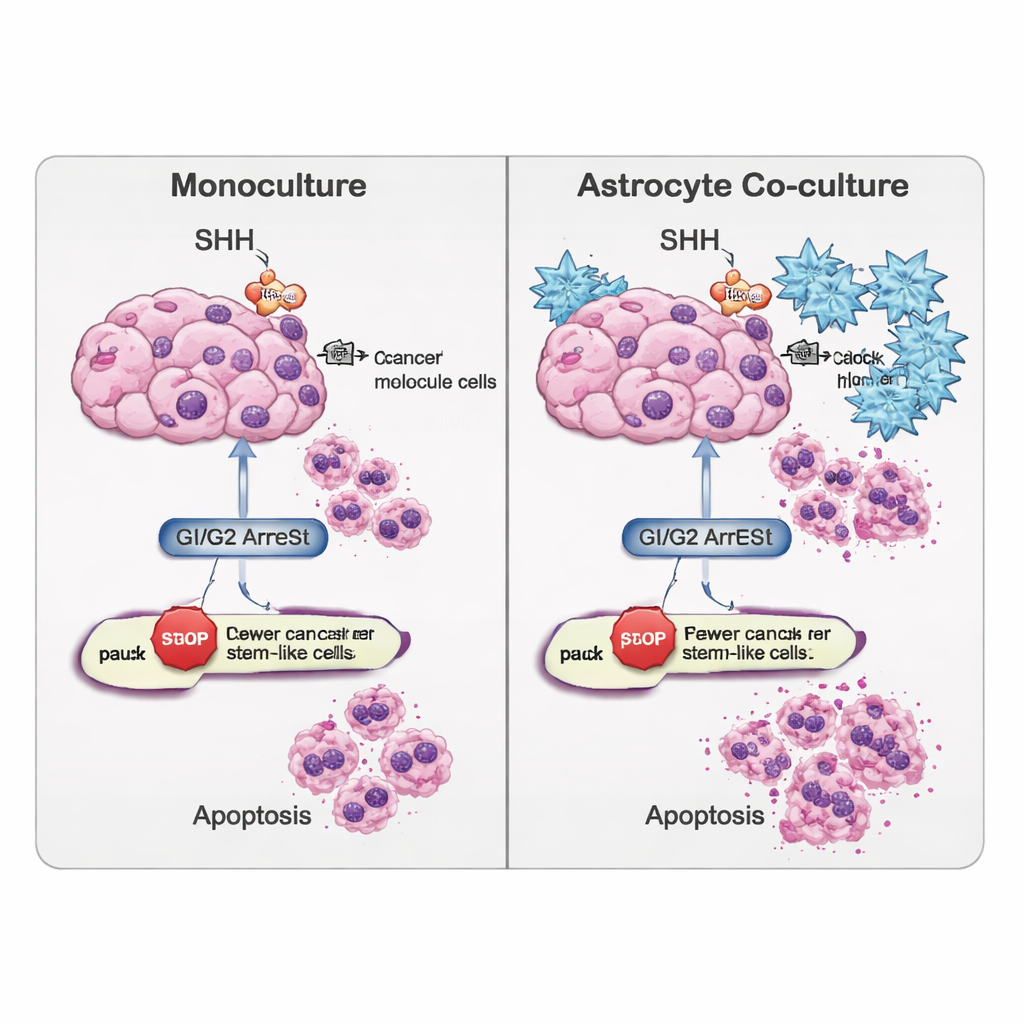
What this means for future brain cancer treatments
For a non‑specialist, the key message is that targeting a single growth signal in glioblastoma is not enough if we ignore the tumor’s neighborhood. This work shows that astrocytes—the normal cells that share space with the tumor—can profoundly change how both bulk cancer cells and cancer stem cells respond to SHH pathway blockade. Cancer stem cells, in particular, became more prone to cell‑cycle arrest and to a form of cell death when astrocytes were present. The authors conclude that drugs aimed at SHH and related pathways are more likely to work if they are tested and eventually used in ways that reflect the real brain environment, possibly in combination with other treatments that attack the tumor’s backup survival routes. In short, understanding and exploiting the “conversation” between tumor cells and their neighbors may open more precise and durable strategies against this otherwise relentless cancer.
Citation: Kocaturk, D.C., Ozdil, B., Adali, Y. et al. SHH pathway inhibition and astrocyte co-culture induce distinct responses in glioblastoma and cancer stem cells. Sci Rep 16, 7712 (2026). https://doi.org/10.1038/s41598-026-38199-y
Keywords: glioblastoma, cancer stem cells, Sonic Hedgehog pathway, tumor microenvironment, astrocytes