Clear Sky Science · en
Integrated metabolomics and 16S rRNA sequencing reveal the mechanism of total flavones of Abelmoschus manihot (L.) Medic against liver fibrosis
Flower Power for a Scarred Liver
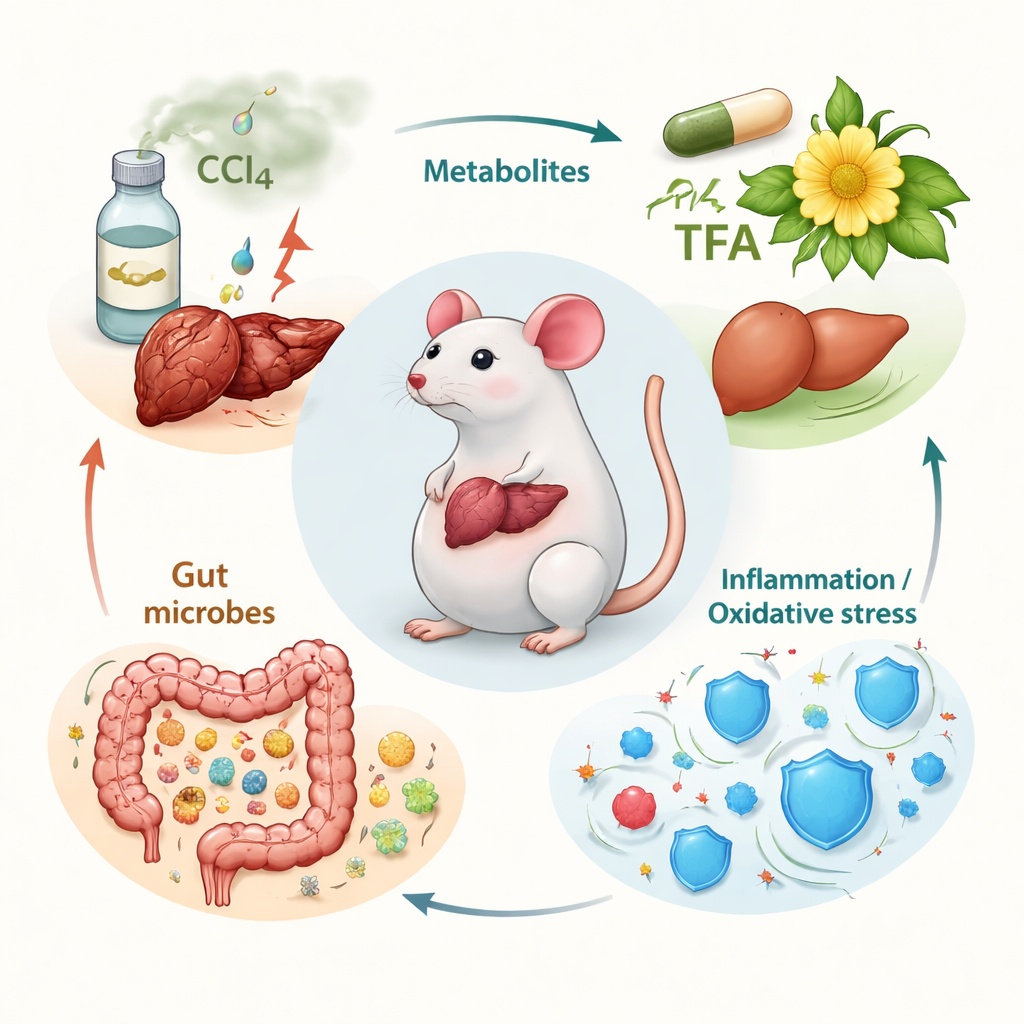
Liver scarring, or fibrosis, is a silent danger that can slowly lead to cirrhosis and liver failure. Modern medicine still lacks safe, broadly effective drugs to stop or reverse this process. This study explores whether natural compounds from the bright yellow flowers of Abelmoschus manihot—a traditional Chinese medicinal plant already used for kidney disease—can protect the liver from long‑term toxic damage, and how they might do it by working on both the body’s chemistry and the microbes in our gut.
A Growing Global Liver Problem
Hundreds of millions of people worldwide live with chronic liver disease caused by viruses, alcohol, fat buildup, or toxins. When the liver is injured again and again, it responds by laying down scar tissue, much like a wound that never fully heals. Over time this fibrosis can stiffen the organ, block blood flow, and progress to cirrhosis, a leading cause of liver‑related deaths. Because many different triggers can drive this damage at once, researchers are especially interested in plant‑based mixtures that can act on several targets simultaneously, rather than single‑molecule drugs that hit only one pathway.
Testing a Traditional Remedy in Mice
The team focused on the “total flavones” of Abelmoschus manihot (TFA)—a concentrated mix of seven related plant compounds previously shown to be anti‑inflammatory and antioxidant. They used a well‑established mouse model in which repeated exposure to the industrial chemical carbon tetrachloride (CCl4) causes chronic liver injury and scarring similar to human fibrosis. Male mice were divided into groups: healthy controls, CCl4‑damaged “model” mice, a standard drug group, and three TFA dose groups. After weeks of CCl4 exposure, the researchers treated the animals with TFA by mouth and then examined their blood, liver tissue, and gut contents.
Cleaner Blood Tests and Healthier Liver Tissue
TFA had clear protective effects. Blood levels of liver enzymes ALT and AST—clinical indicators of liver cell damage—rose sharply in mice exposed to CCl4, but fell by about half in animals given TFA. Microscopic examination told the same story: untreated fibrotic livers showed distorted architecture, dead cells, heavy collagen deposits, and inflammatory cell swarms. TFA‑treated livers had fewer scar “pseudolobules,” less collagen staining, and reduced signs of cell death. Markers of oxidative stress improved as well: antioxidant defenses (SOD and GSH‑Px) went up, while damaging by‑products (MDA) dropped. Key inflammatory messengers, including TNF‑α, IL‑6, and TGF‑β1, were also lowered, along with chemical markers of fibrosis such as hydroxyproline, collagen IV, and hyaluronic acid.
Rebalancing Body Chemistry and Gut Microbes
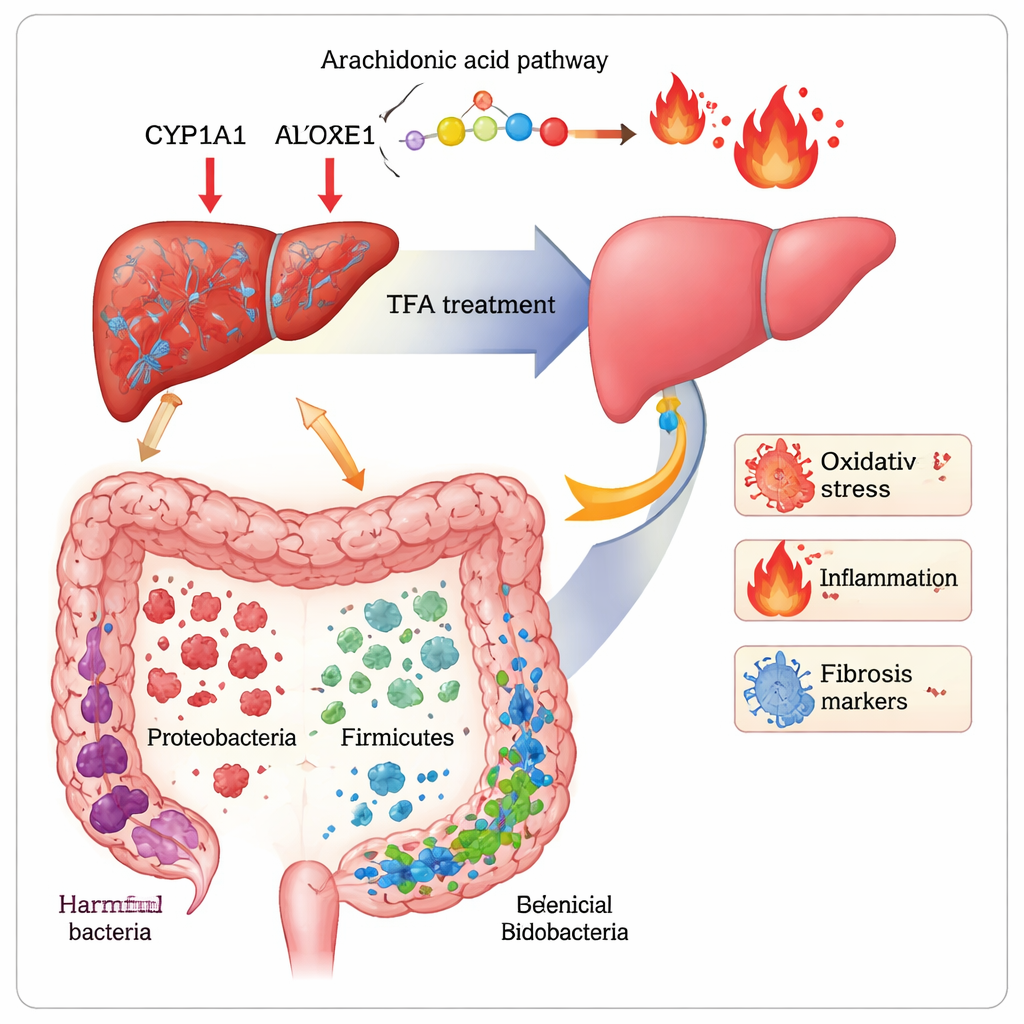
To uncover how TFA exerts these benefits, the researchers combined two powerful tools: metabolomics, which profiles hundreds of small molecules in the blood, and 16S rRNA sequencing, which maps the types of bacteria in the gut. In fibrotic mice, dozens of metabolites involved in fat handling and amino acid breakdown were disturbed. TFA partly restored balance in pathways linked to cell membranes (glycerophospholipids), the mood‑related amino acid tryptophan, and a fatty acid called arachidonic acid that feeds inflammatory signals. Levels of two arachidonic acid products, 20‑HETE and 16(R)‑HETE—both tied to oxidative stress and fibrosis—were pushed back toward normal. In the liver, TFA lowered the abundance of three enzymes (CYP1A1, CYP2E1, ALOX15) that help generate these harmful molecules. At the same time, CCl4 disrupted the gut microbiota, shrinking beneficial groups such as Firmicutes, Lactobacillus, and Bifidobacterium and allowing potentially harmful Proteobacteria to flourish. TFA reversed much of this shift, increasing diversity and restoring “friendly” bacteria associated with better barrier function and lower inflammation.
A Multi‑Target Strategy on the Gut–Liver Highway
By linking bacterial changes with blood chemicals and disease markers, the study suggests that TFA acts along the “gut–liver axis”—the busy highway of signals, nutrients, and toxins traveling from the intestine to the liver. Harmful bacteria and their products appear to track with higher inflammation and scarring, whereas beneficial microbes correlate with healthier metabolic profiles and less fibrosis. TFA seems to nudge this entire system back toward balance: it strengthens antioxidant defenses, calms inflammatory pathways fueled by arachidonic acid, reshapes the microbial community in the gut, and ultimately eases the scarring response in the liver.
What This Could Mean for Future Treatments
For non‑specialists, the key message is that a flower extract long used in traditional medicine showed real promise in a modern, rigorous test of chronic liver scarring. In mice, TFA did more than just protect liver cells—it tuned down toxic chemistry and harmful gut microbes at the same time. While these results do not yet prove benefit in people, and were obtained only in male animals using one type of liver injury, they highlight a potential multi‑target therapy that works with the body’s own networks rather than against a single molecule. With further testing, such plant‑derived flavones could one day become part of a broader toolbox to slow or even reverse liver fibrosis.
Citation: Li, D., Ge, H., Zhang, Y. et al. Integrated metabolomics and 16S rRNA sequencing reveal the mechanism of total flavones of Abelmoschus manihot (L.) Medic against liver fibrosis. Sci Rep 16, 7342 (2026). https://doi.org/10.1038/s41598-026-38192-5
Keywords: liver fibrosis, gut microbiota, metabolomics, flavonoids, traditional Chinese medicine