Clear Sky Science · en
Characteristics of strontium adsorption onto aquatic sediments in Southwest China
Why this metal in mud matters
When heavy metals get into rivers and lakes, they do not just float away; many end up stuck to the mud on the bottom. One such metal is strontium, which behaves a lot like calcium in our bodies and can become dangerous when its radioactive forms are present. This study looks at how sediments from rivers and lakes in Southwest China hold on to strontium, and how temperature, water chemistry, and competing elements like calcium control whether that metal stays locked in the mud or is released back into the water.
Rivers, lakes, and an invisible safety net
The researchers focused on three types of aquatic sediments collected from a river, a creek, and a lake in the Mianyang area of Sichuan Province. These sediments act as an invisible safety net: they can trap dissolved strontium from the water above and from groundwater below. The team carefully measured basic properties of the sediments, including how alkaline they were, how much organic matter they contained, and how many microscopic sites on their surfaces could bind positively charged particles. These surface sites, largely controlled by tiny bits of clay and organic material, set the stage for how much strontium the sediments can hold.
How the experiments were done
To probe strontium behavior, the scientists ran controlled lab tests known as batch adsorption experiments. They shook small amounts of sediment with water containing different levels of strontium, then measured how much metal remained in the water and how much had moved into the sediment. They repeated this across a range of temperatures close to natural river and lake conditions and added different amounts of dissolved calcium salt to see how strongly calcium would compete with strontium. Along the way, they closely tracked changes in water acidity (pH), because even small shifts in pH can tilt the balance between metals attached to sediment and metals roaming free in the water.
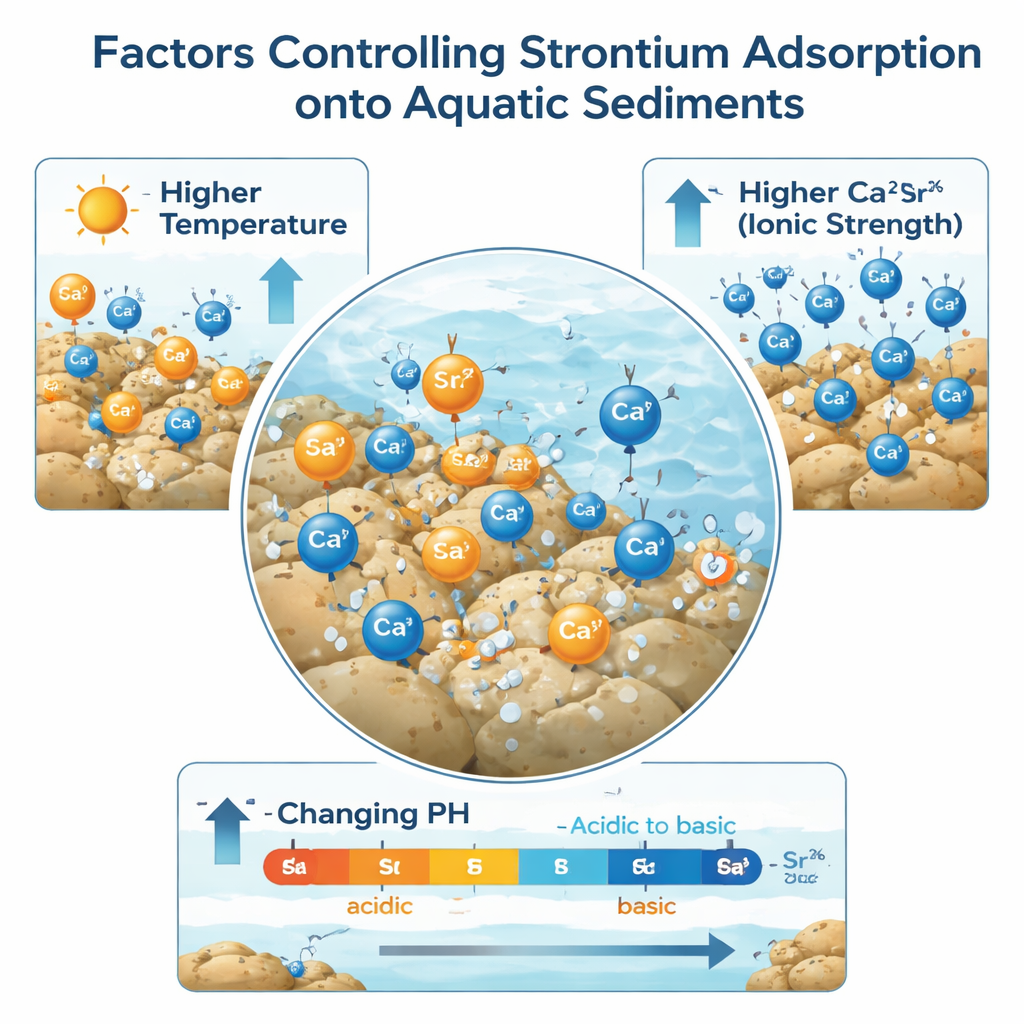
Warmer water and richer mud hold more strontium
The results showed that strontium attachment to the sediments followed well-known patterns used by chemists to describe how surfaces become coated. In everyday terms, as the water warmed, the sediment could hold more strontium. Over the tested temperature span, the maximum amount of strontium that could stick to the sediment increased by roughly 1 to 2 times, depending on the site. Warmer conditions sped up the motion of dissolved strontium and made it easier for the ions to reach and occupy binding spots in the mud. Among the three sites, the lake sediment, which had a much larger surface area and more sites capable of swapping charged particles, stored the most strontium per unit weight.
When calcium crowds out strontium
Calcium, a close chemical cousin of strontium, turned out to be a powerful rival. When the researchers added more calcium to the water, the sediments took up far less strontium. At the highest calcium levels tested, the capacity of some sediments to hold strontium dropped by nearly twentyfold. This indicates that strontium is often held by relatively weak electrostatic forces that can be reversed when many other positively charged ions are present. The team also found that as more strontium was taken up, the water became slightly more acidic, suggesting that hydrogen ions were released as strontium swapped into place on the sediment surfaces.
pH, removal efficiency, and what it means for safety
Even within the modest pH range typical of natural waters, the acidity of the solution made a clear difference. As the starting pH in the experiments rose from slightly acidic toward mildly alkaline, the share of strontium removed from the water increased, reaching about half removal in the most favorable cases. In real environments this means that modest changes in water chemistry, such as pollution that acidifies a river or the influx of salts that raise calcium levels, could cause previously captured strontium to be released from sediment back into the water. Overall, the study shows that sediments can act as an important temporary storehouse for strontium, but their grip is not permanent. Knowing how temperature, calcium, and pH tip this balance helps scientists and regulators better predict where strontium will end up and how to design cleanup and protection strategies for contaminated waters.
Citation: Luo, X., Zhang, D., Zhou, M. et al. Characteristics of strontium adsorption onto aquatic sediments in Southwest China. Sci Rep 16, 6948 (2026). https://doi.org/10.1038/s41598-026-38190-7
Keywords: strontium, aquatic sediments, heavy metal pollution, water quality, radionuclide contamination