Clear Sky Science · en
The hypoxic tight-skin mouse model of Group 3 pulmonary hypertension
Why this research matters
Pulmonary hypertension is a serious, often fatal rise in blood pressure in the lungs. One of its most common forms, called Group 3 pulmonary hypertension, develops in people with chronic lung diseases such as emphysema or pulmonary fibrosis, or in those exposed to long-term low oxygen. Yet doctors still lack targeted drugs for this condition, in part because scientists have not had an animal model that truly mimics what happens in human lungs. This study introduces a new mouse model that closely resembles Group 3 disease and uncovers a harmful signaling chain that could be targeted by future treatments.
A mouse that mimics damaged lungs
The researchers focused on a strain of mice known as “tight-skin” (Tsk) mice. These animals carry a mutation in a structural protein called fibrillin-1. Because of this defect, they naturally develop emphysema-like changes and scarring in their lungs, much like people with severe chronic lung disease. The team compared these mice to normal littermates while keeping them either in normal air or in low-oxygen conditions for four weeks, to mirror the chronic lack of oxygen that many human patients experience. 
Low oxygen turns a vulnerable lung into high-pressure lungs
When both normal and tight-skin mice were kept in room air, their lung blood pressures were similar and within the normal range. But under chronic low oxygen, the differences were striking. Normal mice developed only moderate increases in pressure, while tight-skin mice developed severe pulmonary hypertension, with much higher pressures in the right side of the heart and obvious thickening of the small lung arteries. Microscopy showed enlarged airspaces (emphysema) and heavy muscularization of tiny vessels in the tight-skin animals, closely mirroring the structural damage seen in patients with Group 3 disease. This indicates that pre-existing lung injury plus hypoxia together push the system into a much more dangerous state than hypoxia alone.
A danger signal in the blood
The team then asked why tight-skin mice react so strongly to low oxygen. They homed in on a molecule called HMGB1, a “danger signal” protein released by stressed or damaged cells. Tight-skin mice already had higher HMGB1 levels in their blood at baseline, and these levels rose even further during hypoxia. Giving the mice a small peptide drug, P5779, which blocks HMGB1’s interaction with one of its key receptors (TLR4), largely prevented the development of severe pulmonary hypertension in tight-skin mice. The treated animals had lower lung pressures, less thickening of their lung vessels, and reduced enlargement of the right side of the heart. These protective effects were much more pronounced in tight-skin mice than in normal mice, pointing to HMGB1 as a central amplifier of disease in this model.
How a growth signal feeds the danger signal
Another player in this story is TGF-β, a growth factor long linked to scarring and remodeling in diseased lungs. The researchers found that tight-skin mice had higher baseline TGF-β activity in their lungs, as shown by increased activation of its downstream messenger, SMAD-3, around blood vessels. In experiments with human lung artery adventitial fibroblasts—cells that line the outer layer of blood vessels—adding TGF-β boosted production of HMGB1. In turn, HMGB1 caused both these fibroblasts and neighboring smooth muscle cells to proliferate. Importantly, this growth effect depended on the TLR4 receptor and was blocked by P5779, but not by blocking another HMGB1 receptor called RAGE. Together, these findings outline a loop in which mutant fibrillin-1 promotes TGF-β activation, TGF-β raises HMGB1 levels, and HMGB1 drives the overgrowth of vessel wall cells that narrows lung arteries. 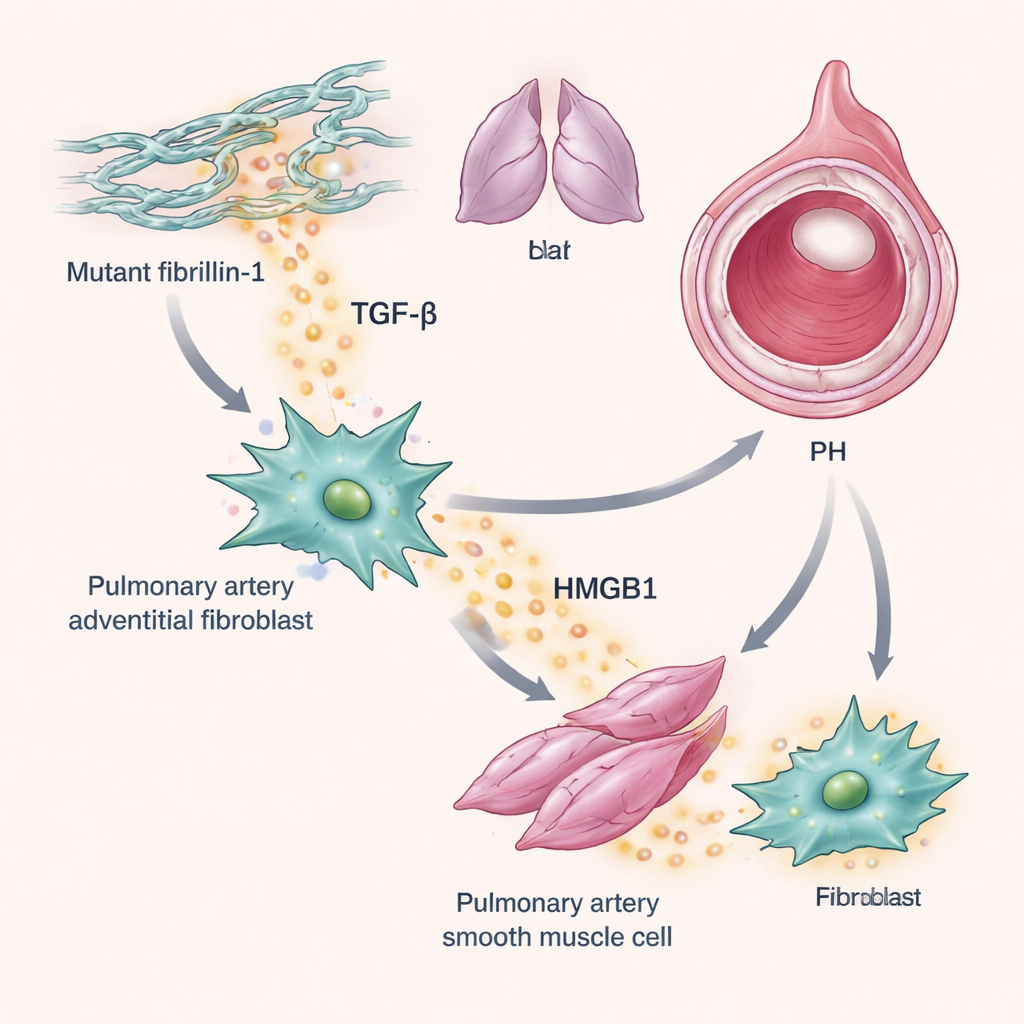
Implications for patients
By combining an emphysema-like lung structure with chronic hypoxia, the tight-skin mouse model reproduces many key features of Group 3 pulmonary hypertension that simpler hypoxia-only models miss. The work highlights a TGF-β–HMGB1–TLR4 pathway that turns injured, low-oxygen lungs into high-pressure lungs. For people living with chronic lung disease, this research suggests new routes for therapy: drugs that dampen TGF-β signaling, or that block HMGB1–TLR4 interactions like P5779 does in mice, might one day help prevent or slow the rise in lung blood pressure. The model itself also offers a powerful new platform for testing such treatments before they reach the clinic.
Citation: Chi, L., Foley, A.E., Goodarzi, G. et al. The hypoxic tight-skin mouse model of Group 3 pulmonary hypertension. Sci Rep 16, 6968 (2026). https://doi.org/10.1038/s41598-026-38174-7
Keywords: pulmonary hypertension, chronic lung disease, hypoxia, HMGB1, TGF-beta