Clear Sky Science · en
IL-18 promotes pancreatic fibrosis via release of IL-4 from pancreatic stellate cells and induces macrophage M2 polarization
Why scarring in the pancreas matters
Chronic pancreatitis is a long‑lasting inflammation of the pancreas that slowly destroys this vital organ, often causing severe pain, digestive problems, and a higher risk of pancreatic cancer. A key feature of the disease is fibrosis—scar tissue that replaces healthy pancreatic cells. This study asks a simple but crucial question: what signals inside the pancreas turn ongoing inflammation into permanent scarring, and could blocking these signals slow or stop the damage?
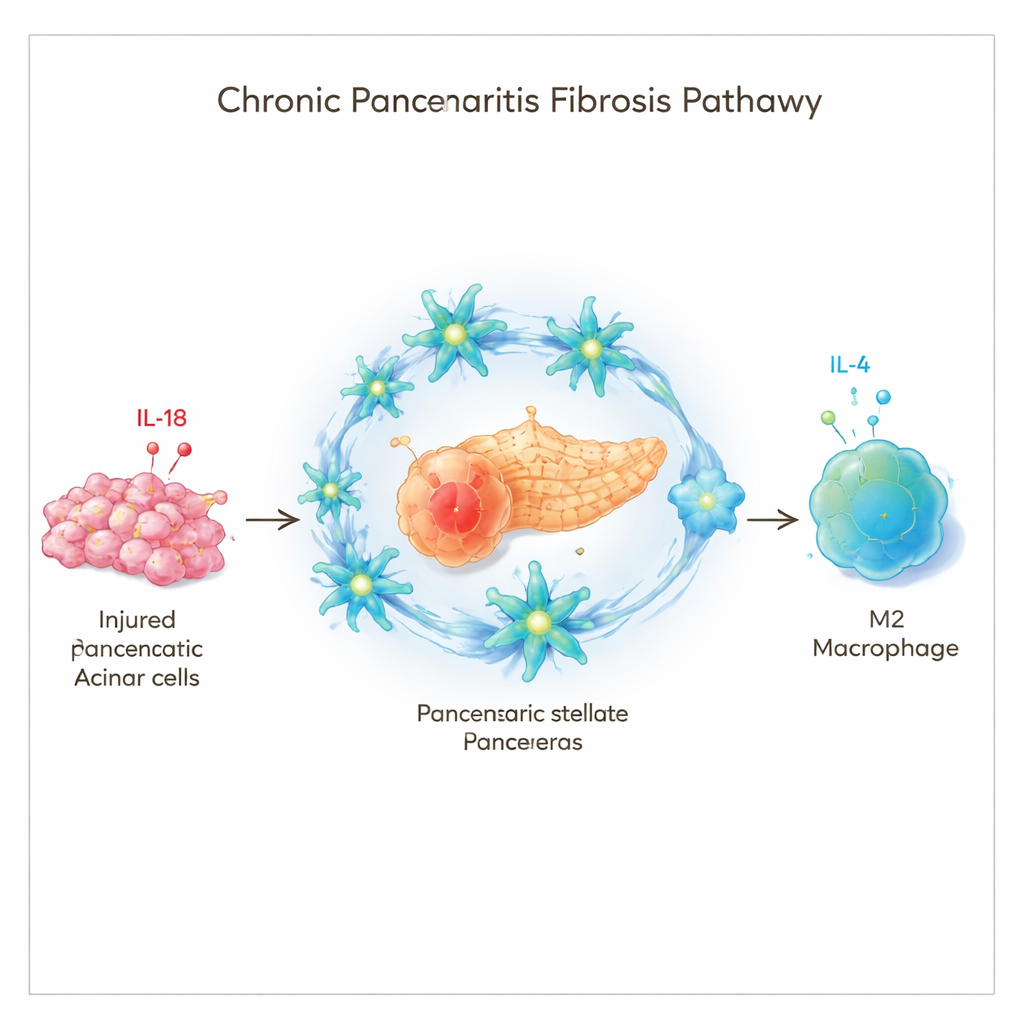
A troublemaking alarm signal
The researchers focused on a molecule called interleukin‑18 (IL‑18), part of the body’s immune “alarm system.” IL‑18 is known to rise during inflammation and has been linked to scarring in the heart, kidneys, and liver. By examining tissue samples from people with chronic pancreatitis and using mouse models of the disease, the team found that IL‑18 levels were much higher in diseased pancreases than in healthy ones. Importantly, the more IL‑18 present in a patient’s pancreas, the more extensive the fibrosis. They traced a major source of this IL‑18 to injured acinar cells—the normal digestive enzyme‑producing cells of the pancreas—which release IL‑18 when they are damaged.
The supporting cells that turn into scar builders
Scar tissue in chronic pancreatitis is mainly produced by pancreatic stellate cells, a type of support cell that normally lies quiet but can switch into an active, scar‑forming state. The study showed that these stellate cells carry the receptor for IL‑18, meaning they can directly sense this alarm signal. In mice engineered to lack this receptor, repeated injury produced far less pancreatic scarring, less activation of stellate cells, and a different pattern of immune cell invasion. This indicates that IL‑18 is not just a bystander marker of inflammation; it is a driver that helps push stellate cells and the surrounding tissue toward fibrosis.
How immune cells are pushed toward a scarring mode
Another key player in chronic pancreatitis is the macrophage, an immune cell that can adopt different “personalities.” In one state (often called M1), macrophages are aggressive defenders; in another (M2), they shift toward tissue remodeling and, unfortunately, can promote scarring. The scientists found that mice lacking the IL‑18 receptor had fewer M2‑type macrophages in their pancreases. Surprisingly, IL‑18 by itself did not directly turn macrophages into the M2 type in lab dishes. Instead, IL‑18 acted on stellate cells, prompting them to release another signaling molecule, interleukin‑4 (IL‑4). IL‑4 is well known for steering macrophages toward the M2, pro‑fibrosis state. When macrophages were exposed to fluid from IL‑18‑treated stellate cells, they strongly shifted to the M2 pattern—but this effect largely disappeared when IL‑4 was blocked.
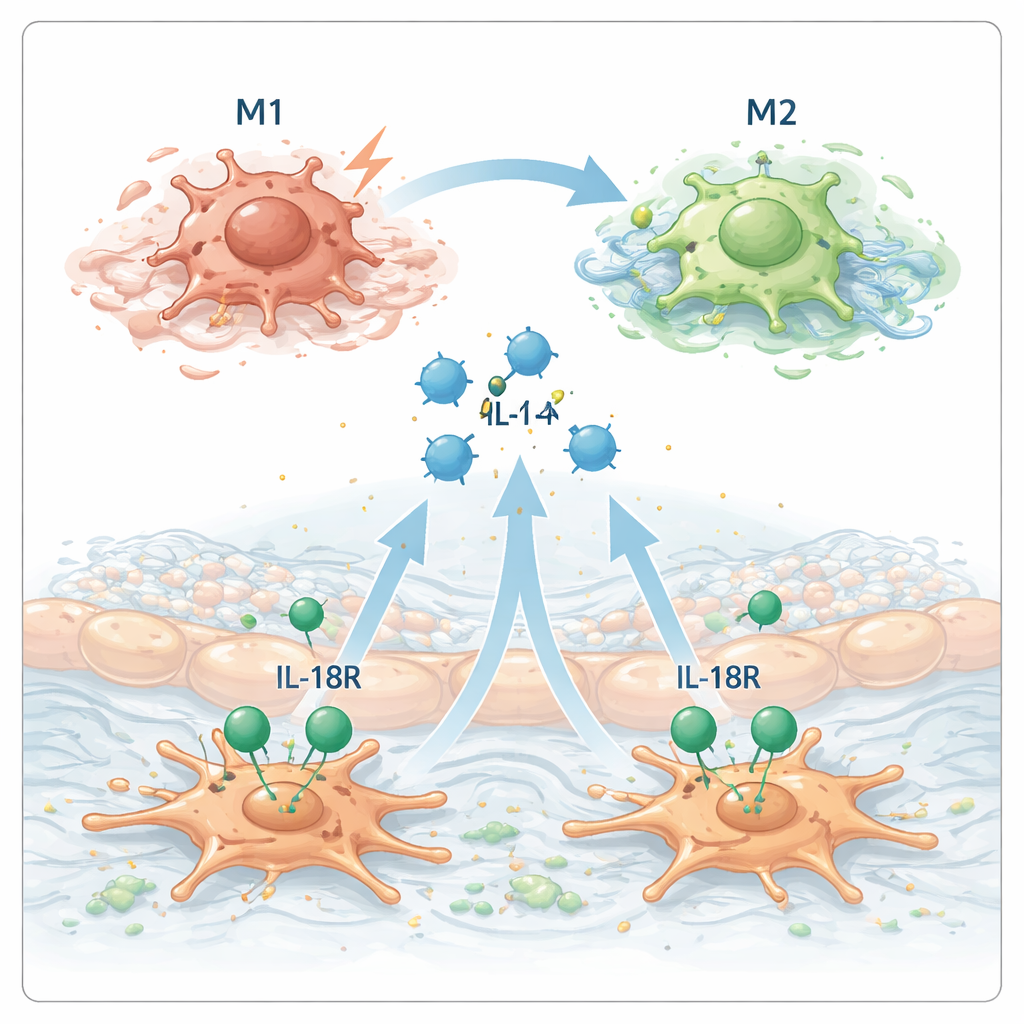
A chain reaction that worsens scarring
Putting these pieces together, the researchers propose a chain reaction inside the diseased pancreas. First, acinar cells are injured and release IL‑18. Next, IL‑18 activates nearby stellate cells through its receptor. In response, stellate cells secrete IL‑4, which then instructs macrophages to adopt the M2, scar‑promoting state. These M2 macrophages, in turn, further stimulate stellate cells and help lay down collagen and other components of scar tissue, deepening the fibrosis. In mouse experiments, giving extra IL‑18 during chronic pancreatitis made the disease more severe and the scarring worse, but blocking IL‑4 blunted these harmful effects.
What this means for future treatments
For non‑specialists, the core message is that chronic pancreatitis scarring is not random; it is driven by a specific conversation between damaged pancreatic cells, stellate cells, and immune cells. IL‑18 sits near the top of this communication chain, and IL‑4 acts as a key middle messenger. By interfering with IL‑18, its receptor on stellate cells, or IL‑4 signaling to macrophages, future therapies may be able to reduce or slow the buildup of scar tissue in chronic pancreatitis, potentially preserving pancreatic function and improving patients’ quality of life.
Citation: Tu, G., Peng, C., Xie, S. et al. IL-18 promotes pancreatic fibrosis via release of IL-4 from pancreatic stellate cells and induces macrophage M2 polarization. Sci Rep 16, 7540 (2026). https://doi.org/10.1038/s41598-026-38168-5
Keywords: chronic pancreatitis, pancreatic fibrosis, IL-18, IL-4, macrophage polarization