Clear Sky Science · en
HPLC–DAD analysis of functional dietary supplements followed by liquid–liquid microextraction-assisted FTIR identification of IR-active ingredients
Why checking what’s really in your drink matters
From sports drinks promising faster muscle recovery to vitamin waters that claim to boost immunity, functional beverages and dietary supplements have become everyday products. Yet what appears on the label does not always match what is actually inside the bottle, and hidden additives or wrong doses can affect both health and performance. This study describes a new laboratory approach that helps scientists and regulators verify the true chemical makeup of complex drinks, using a combination of separation and light-based fingerprinting techniques to see through the clutter of sugars, salts, and other ingredients.
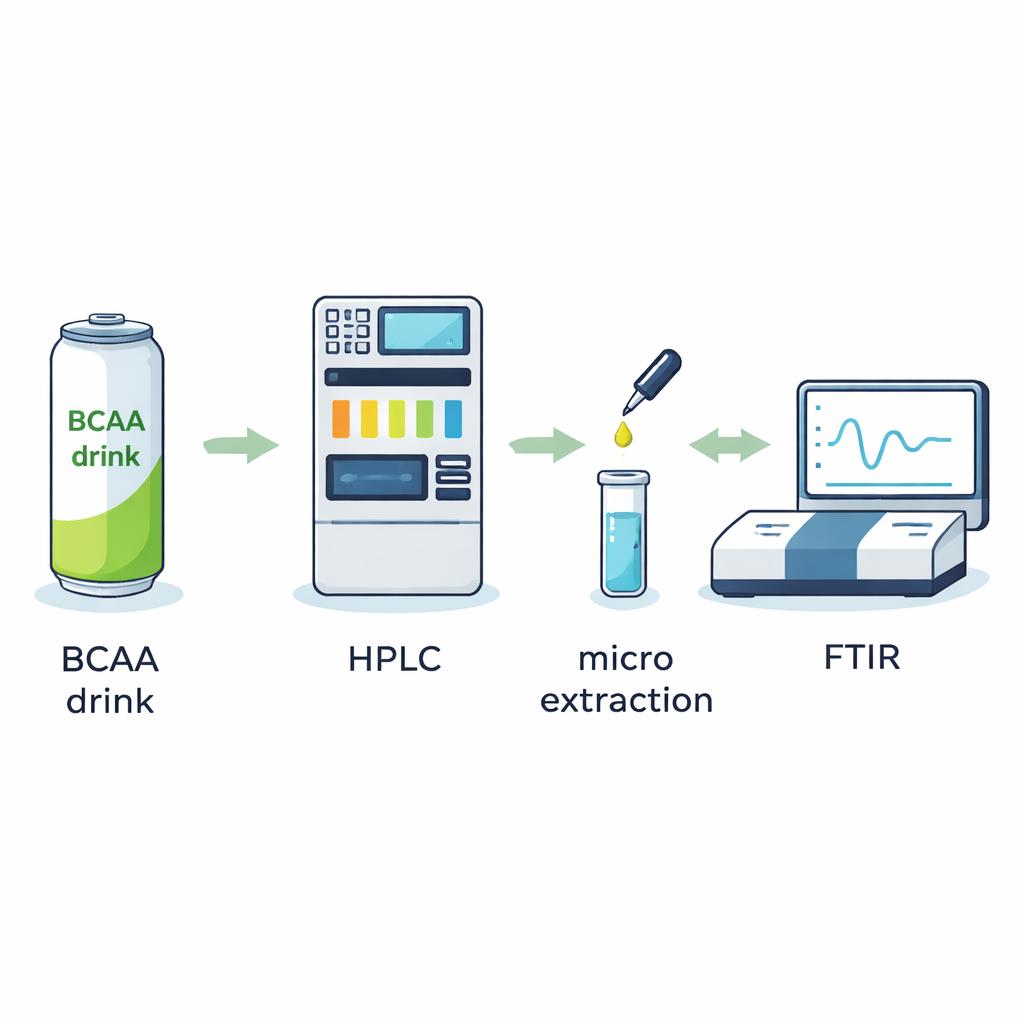
Untangling crowded ingredient lists
Modern functional drinks, such as branched-chain amino acid (BCAA) beverages, usually contain a cocktail of active ingredients—amino acids like valine, leucine, and isoleucine, plus acids, sweeteners, caffeine, and vitamins. Identifying each of these precisely is not trivial, because they are mixed into a thick chemical “soup” that also includes preservatives, flavorings, and sometimes contaminants. Traditional quality checks rely heavily on high-performance liquid chromatography (HPLC), which separates a sample into individual chemical bands, and on mass spectrometry, a powerful but expensive technique. The authors instead explore how to pair HPLC with Fourier transform infrared (FTIR) spectroscopy, a method that reads each molecule’s unique infrared light fingerprint, to create a more accessible tool for routine quality control.
The problem with water and salts
Although FTIR is fast and information-rich, it stumbles when used directly with liquid samples from standard HPLC systems. The issue is that water, common organic solvents, and especially dissolved buffer salts strongly absorb infrared light and leave behind their own spectral signatures. After the mobile liquid phase is evaporated, crystals from buffer salts can completely cover or distort the signal of the real ingredients. The authors first carefully examined how different surfaces for holding dried samples, such as various metals and crystals, influenced background signals. They found that plain aluminum foil offered high reflection and a relatively clean background, making it a simple and effective support for later measurements.
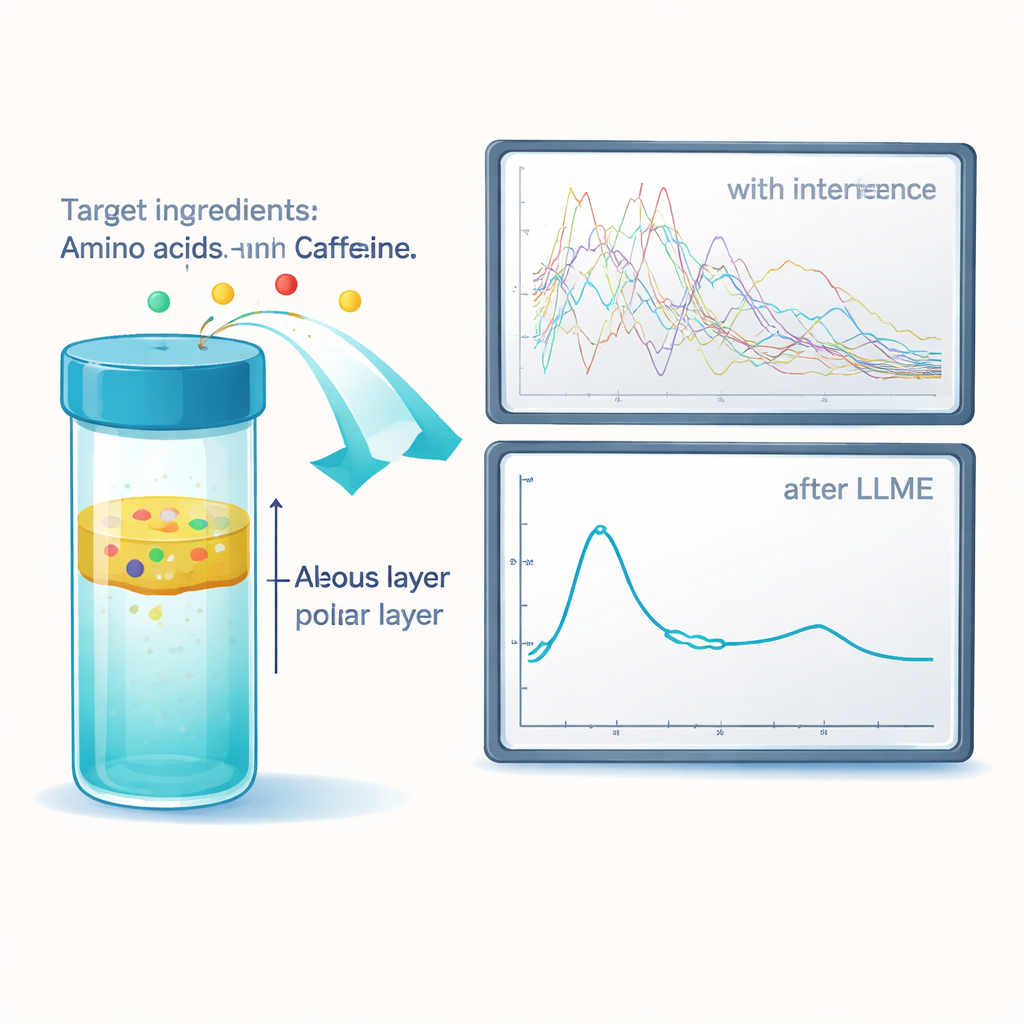
A tiny extraction step that makes a big difference
To overcome solvent and salt interference, the researchers introduced a liquid–liquid microextraction (LLME) step between HPLC and FTIR. In this approach, each collected HPLC fraction from a BCAA drink is mixed with a very small volume of a heavy, non-polar liquid that does not absorb in the infrared region. During vigorous mixing, the desired ingredients move from the watery phase into this special solvent, while most salts and other troublesome components stay behind. The team tested several candidate solvents and showed that one of them, a fluorinated compound, consistently pulled out enough of each target ingredient to provide clear FTIR fingerprints. Compared with simply evaporating the original solvent, LLME either alone or combined with evaporation produced much cleaner spectra, even when complex solvent mixtures and buffers were present.
Putting the method to work on a real sports drink
With the technique optimized, the authors turned to a commercially available BCAA beverage. After separating the drink’s contents by HPLC and applying the LLME cleanup, they recorded FTIR spectra of each purified fraction. The resulting fingerprints allowed them to confidently identify key ingredients including citric acid, vitamin C (L-ascorbic acid), the three BCAAs (valine, leucine, isoleucine), and caffeine. Importantly, all of this was done using standard laboratory instruments—an ordinary HPLC system, a benchtop FTIR microscope, and common glassware—rather than highly specialized or extremely costly equipment. This means the method could be adopted by many quality-control laboratories that currently lack advanced mass spectrometers.
What this means for consumers and regulators
The study shows that a simple added step—microextraction with an infrared-friendly solvent—turns a difficult analytical puzzle into a manageable routine test. While the new HPLC–LLME–FTIR method does not replace high-end mass spectrometry for ultra-trace or highly detailed studies, it offers a practical, low-cost way to confirm the main active ingredients in complex drinks and supplements and to check whether labels are truthful. For consumers, this translates into better safeguards against misleading products; for regulators and manufacturers, it provides a robust tool to support safer, more transparent markets for functional foods, sports nutrition, and related health products.
Citation: Pavelek, D., Kaykhaii, M., Jampilek, J. et al. HPLC–DAD analysis of functional dietary supplements followed by liquid–liquid microextraction-assisted FTIR identification of IR-active ingredients. Sci Rep 16, 7028 (2026). https://doi.org/10.1038/s41598-026-38160-z
Keywords: dietary supplements, functional beverages, quality control, infrared spectroscopy, branched-chain amino acids