Clear Sky Science · en
Traceable stiffness calibration of colloidal AFM probes for biomechanical measurements
Why tiny forces matter for living tissues
When doctors and biologists probe living cells or soft tissues, they increasingly rely on atomic force microscopy (AFM)—a technique that can feel forces a trillion times smaller than the weight of an apple seed. To turn those delicate touches into trustworthy numbers about how stiff or soft a sample is, the tiny AFM “spring” must itself be measured very accurately. This paper tackles a hidden source of error in that process and shows how to correct it, making AFM-based biomechanical measurements more reliable.

Feeling softness with a microscopic spring
In AFM, a hair‑thin cantilever acts like a miniature diving board with a tip at its free end. When this tip pushes on a sample, the cantilever bends; by knowing how stiff the cantilever is, researchers can convert that bend into force and, ultimately, into a measure of the sample’s mechanical properties. For many biological studies, researchers replace the sharp tip with a glued-on glass or plastic bead, creating a so‑called colloidal probe. The larger, rounded surface is gentler to cells and tissues and gives more predictable contact, but it also makes the calibration of the cantilever’s stiffness trickier.
A hidden hitch: sliding and friction
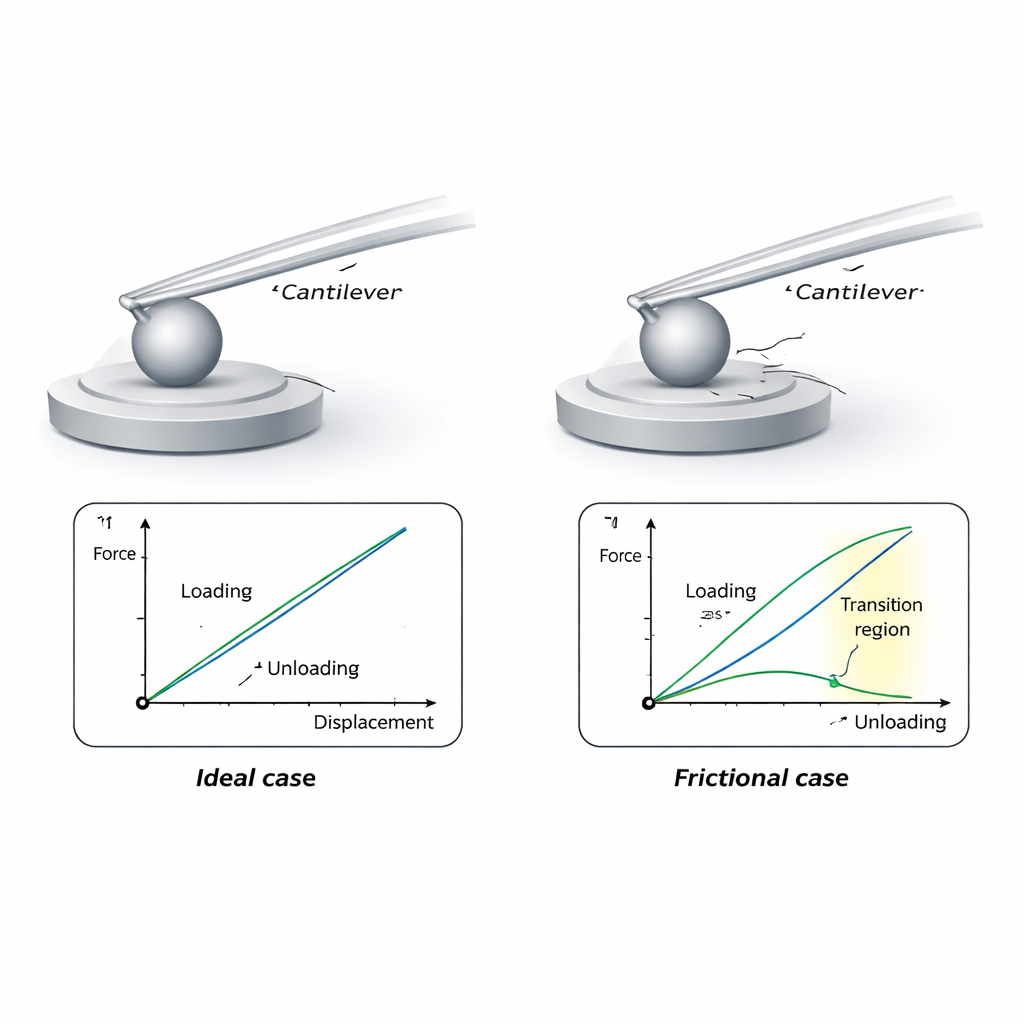
Standard, highly accurate calibration methods press the AFM tip against a reference surface mounted on a precision balance and record how much force is needed to bend the cantilever by a given distance. This works well for sharp, smooth tips. However, when a big bead is glued to the cantilever, its rough surface and large size cause it to drag and slide over the reference surface as the cantilever bends. That sliding introduces friction, which changes how the cantilever bends and makes the simple “force versus deflection” line appear steeper during loading and shallower during unloading. If friction is ignored, the calculated stiffness of the cantilever can be noticeably wrong.
New model to separate stiffness from friction
The authors develop a compact analytical model that describes how both the normal pushing force and the sideways friction force combine to bend the cantilever. By comparing the slope of the loading curve with the slope of the unloading curve, their formulas allow experimenters to back out the true spring stiffness and, at the same time, estimate the friction between bead and reference surface. The model also predicts a small “transition zone” at the start of unloading, where the direction of sliding reverses and the curve temporarily deviates from a straight line. Using this transition-free portion of the data yields cleaner stiffness values.
Testing the idea in simulations and real hardware
To check their theory, the team first used finite element simulations—computer models that track how shapes deform—to mimic a cantilever with a glued microsphere pushing and sliding on a rounded button. As they dialed up the friction in the model, the simulated force–deflection curves behaved exactly as predicted: loading and unloading slopes split apart, and a distinct kink appeared at the start of unloading. Next, the researchers built a dedicated micro‑force measurement facility around a traceably calibrated balance and a nanopositioning stage. With this setup, they measured both conventional sharp AFM probes and several colloidal probes with glass and polymer spheres, comparing results against an established reference system at another laboratory. The two systems agreed within about one and a half percent.
What this means for studying soft matter
By explicitly including friction, the new approach restores confidence in stiffness values obtained from bead‑based AFM probes, even when large spheres and rough surfaces are involved. The study also provides practical friction coefficients for common material pairs—such as glass on diamond or glass on ruby—that other researchers can use when designing and interpreting AFM experiments. In plain terms, the work offers a clearer way to tell how stiff an AFM spring really is, which in turn sharpens measurements of how soft or rigid cells, tissues, and other delicate materials are. That improved accuracy can help make AFM a more dependable tool in fields ranging from cancer diagnosis to the development of advanced biomaterials.
Citation: Li, Z., Cherkasova, V., Gao, S. et al. Traceable stiffness calibration of colloidal AFM probes for biomechanical measurements. Sci Rep 16, 5243 (2026). https://doi.org/10.1038/s41598-026-38158-7
Keywords: atomic force microscopy, colloidal probes, stiffness calibration, biomechanics, nanofriction