Clear Sky Science · en
The potential effect of albumin replacement on immune modulation and sphingosine 1-phosphate dynamics
Why this matters for patients in intensive care
People in intensive care units often have very low levels of a blood protein called albumin. Doctors sometimes give albumin infusions, hoping to stabilize blood pressure and improve survival, but large trials have not clearly shown a benefit. This study asks a different question: instead of looking only at fluid balance, could albumin influence the immune system by carrying a small fat-like messenger molecule in the blood—and might that help explain why some patients seem to benefit more than others?
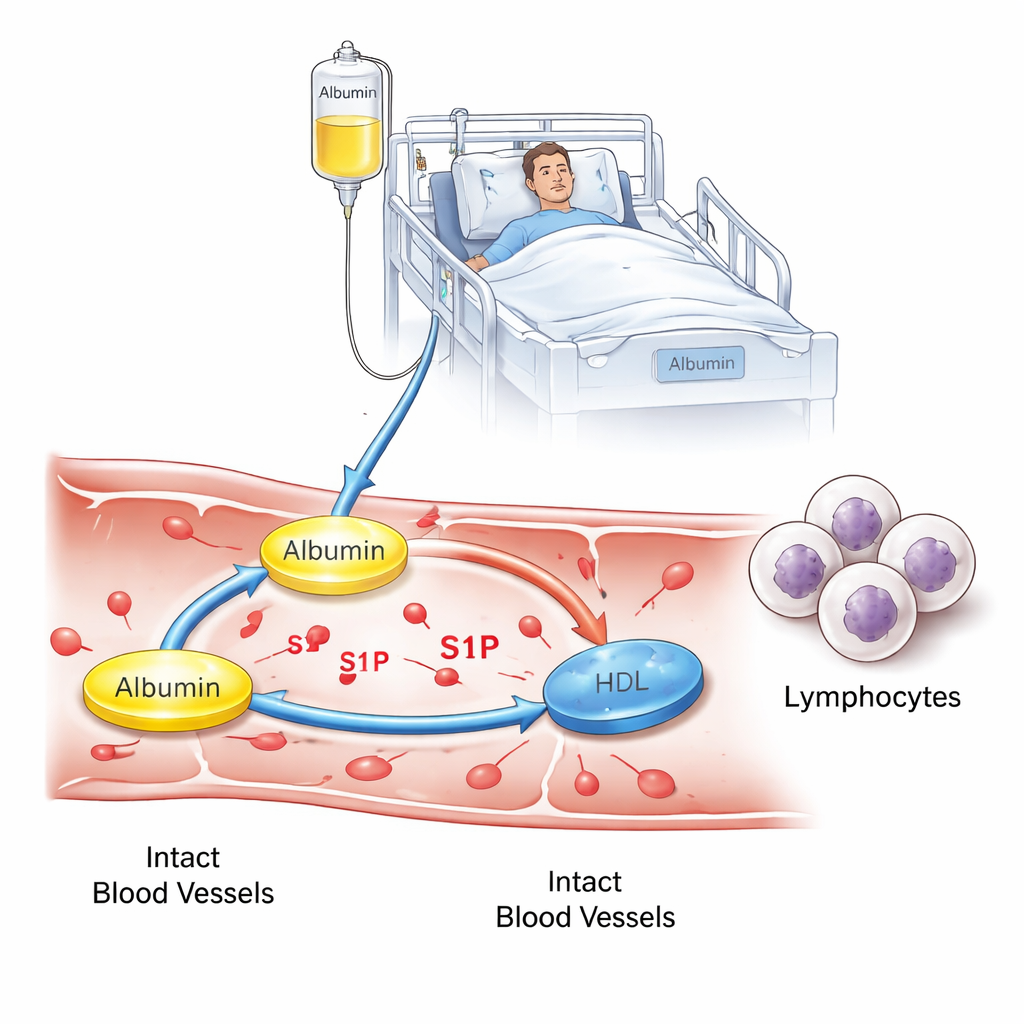
A closer look at albumin and a tiny messenger
Albumin does more than just hold water in our blood vessels. It also transports many small molecules, including sphingosine 1-phosphate (S1P), which helps control how white blood cells move through the body and how “leaky” blood vessels become during severe illness. In healthy people, S1P travels mainly on two carriers: albumin and high-density lipoprotein (HDL, often called “good cholesterol”). When people become critically ill—especially with sepsis—both albumin and HDL levels can drop, and earlier work has shown that S1P levels fall as well, which is linked to worse outcomes. The authors set out to test whether giving human albumin to intensive care patients with low albumin could change how S1P is distributed between albumin and HDL, and whether that shift would show up as changes in immune cells or blood vessel stability.
How the study was done at the bedside
The team followed 47 adults in a single intensive care unit over about a year. All had low albumin levels, but their treating doctors—not the researchers—decided who received albumin infusions. Based on this, patients were placed into three groups: a small control group with normal albumin, a larger group with low albumin who did not receive albumin, and a treatment group with low albumin who received 180 grams of human albumin over three days. Blood samples were taken for standard lab tests, detailed measurements of S1P, and counts of different white blood cell types. The researchers also used specialized methods to determine whether S1P in each sample was mainly bound to albumin or to HDL, and then ran laboratory assays to see how patient plasma affected white blood cell movement and the tightness of blood vessel cell layers.
What changed in the blood—and what did not
As expected, albumin infusions clearly raised blood albumin levels in the treated group, confirming that the treatment worked in a basic sense. Surprisingly, total S1P levels in the blood did not increase with albumin therapy and stayed lower than in ICU patients whose albumin was normal from the start. The important change was where S1P was carried: after treatment, more S1P was found bound to albumin and less to HDL, showing a measurable redistribution between carriers without altering the overall amount. At the same time, counts of two key types of immune cells in the bloodstream—CD4 T cells and B cells—fell over the three treatment days. This pattern fits with the idea that changes in S1P handling might alter how immune cells recirculate, though the study design cannot prove cause and effect.
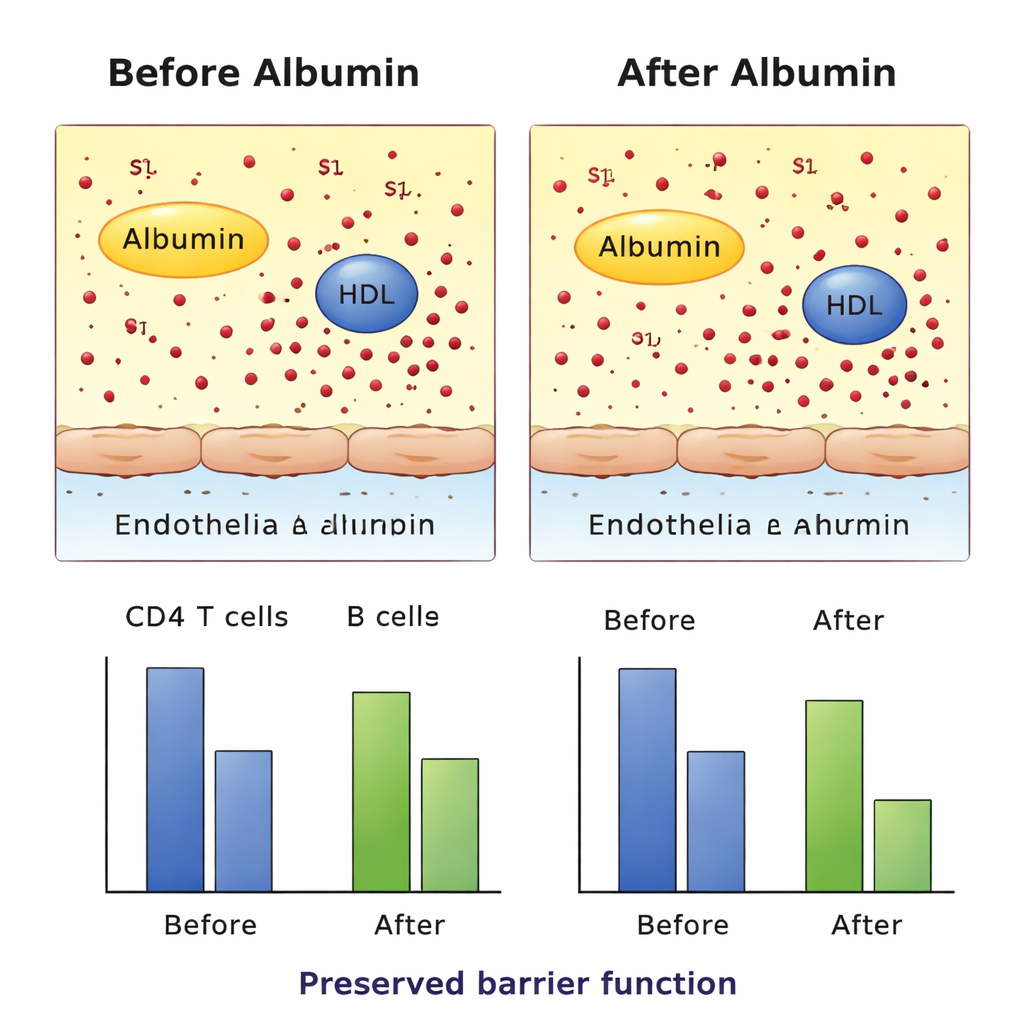
Testing vessel protection and cell movement in the lab
To find out whether this carrier switch had harmful side effects, the researchers tested patient samples in several controlled laboratory systems. They exposed cultured blood vessel cells to plasma from different patient groups and monitored how well the cell layer resisted electrical current, a sign of how tight the barrier remained. They also used a migration assay in which fluorescent immune cells moved through a membrane toward patient plasma, simulating S1P-driven “homing.” In these ex vivo experiments, albumin-treated plasma did not weaken the vessel barrier and did not significantly reduce S1P-driven movement of immune cells compared with controls. Additional receptor-level tests showed that S1P carried on either HDL or albumin could still activate its target receptors, at least under the conditions studied.
What this means for future treatment choices
For non-specialists, the take-home message is that albumin infusions in very ill patients do not seem to restore the overall amount of this important messenger molecule S1P, but they do shift it from HDL to albumin in the bloodstream. That shift is linked to subtle changes in circulating immune cells without obvious harm to blood vessel stability in lab tests. The findings support the idea that albumin has immune-modulating roles beyond simply pulling fluid into the circulation and that its benefits—if they exist—may be limited to particular patient types, such as those with very low HDL or marked inflammation. Larger, targeted studies will be needed to learn whether carefully selected patients can gain a meaningful clinical advantage from this more nuanced role of albumin therapy.
Citation: Winkler, M.S., Enzmann, F., Schilder, M. et al. The potential effect of albumin replacement on immune modulation and sphingosine 1-phosphate dynamics. Sci Rep 16, 5412 (2026). https://doi.org/10.1038/s41598-026-38157-8
Keywords: albumin therapy, critical illness, sphingosine 1-phosphate, sepsis immunology, vascular barrier