Clear Sky Science · en
INHBA: a mitochondrial-related pan-cell death gene associated with the prognosis and immunity of OSCC
Why this matters for mouth cancer
Oral squamous cell carcinoma, a common form of mouth cancer, can rob people of basic abilities such as speaking, eating, and even smiling comfortably. Despite surgery, radiation, and chemotherapy, many patients still face recurrence and poor long‑term survival. This study takes a fresh look at mouth cancer by focusing on how a single gene, called INHBA, helps shape cell death, the tumor’s neighborhood of support cells, and the success or failure of modern treatments like immunotherapy.
A closer look at a deadly mouth cancer
Most head and neck cancers begin in the lining of the mouth, and more than 90% of these are oral squamous cell carcinoma (OSCC). Worldwide, cases and deaths from OSCC are rising and are expected to increase by about 40% by 2040. Standard care relies on surgery plus radiation and chemotherapy, but many tumors resist treatment and come back. Newer immunotherapies, which aim to awaken the body’s own defenses, help only a fraction of patients. To move beyond trial‑and‑error treatment, researchers need to understand which genes drive OSCC and how they interact with the immune system.
Cell death, power plants, and a new gene category
Our cells are programmed to die in several carefully controlled ways, including processes with names like apoptosis, autophagy, ferroptosis, and others. In cancer, these death programs are often rewired so that tumor cells survive when they should not. The team behind this study focused on a broad group they call “pan‑cell death” genes—genes that sit at the crossroads of different death pathways—and narrowed in further on those tied to mitochondria, the tiny power plants in cells. By combining large public cancer datasets with a specialized mitochondrial gene catalog, they defined a set of “mitochondria‑associated pan‑cell death genes” and hunted for those most strongly linked to OSCC behavior and patient outcome.
Finding INHBA among many suspects
Using advanced statistics and nine different machine‑learning algorithms, the researchers sifted through thousands of genes and identified 19 mitochondrial pan‑cell death genes with strong ties to prognosis in OSCC. Among these, INHBA stood out. Its activity was consistently higher in tumor samples than in normal mouth tissue and was linked to worse survival in both OSCC and the broader group of head and neck cancers. When tumors were split into high‑ and low‑INHBA groups, those with higher levels showed more aggressive biological signals, including pathways related to tissue invasion, blood vessel growth, and stress in mitochondria. Laboratory tests on patient samples confirmed that INHBA’s message levels were indeed elevated in real‑world tumors.
The hidden role of support cells around the tumor
To understand where INHBA is most active, the team turned to single‑cell RNA sequencing and spatial transcriptomics, technologies that map gene activity in individual cells and across tissue slices. They found that INHBA is not mainly produced by the cancer cells themselves, but by cancer‑associated fibroblasts—support cells that remodel tissue and shape the immune environment. A particular subtype, called myofibroblastic CAFs, showed especially high INHBA levels. Detailed communication maps suggested that INHBA‑rich fibroblasts talk intensely with immune cells and tumor cells, helping create an immunosuppressive, scar‑like niche around the cancer. This microenvironment appears to encourage tumor growth and may blunt some forms of immunotherapy.
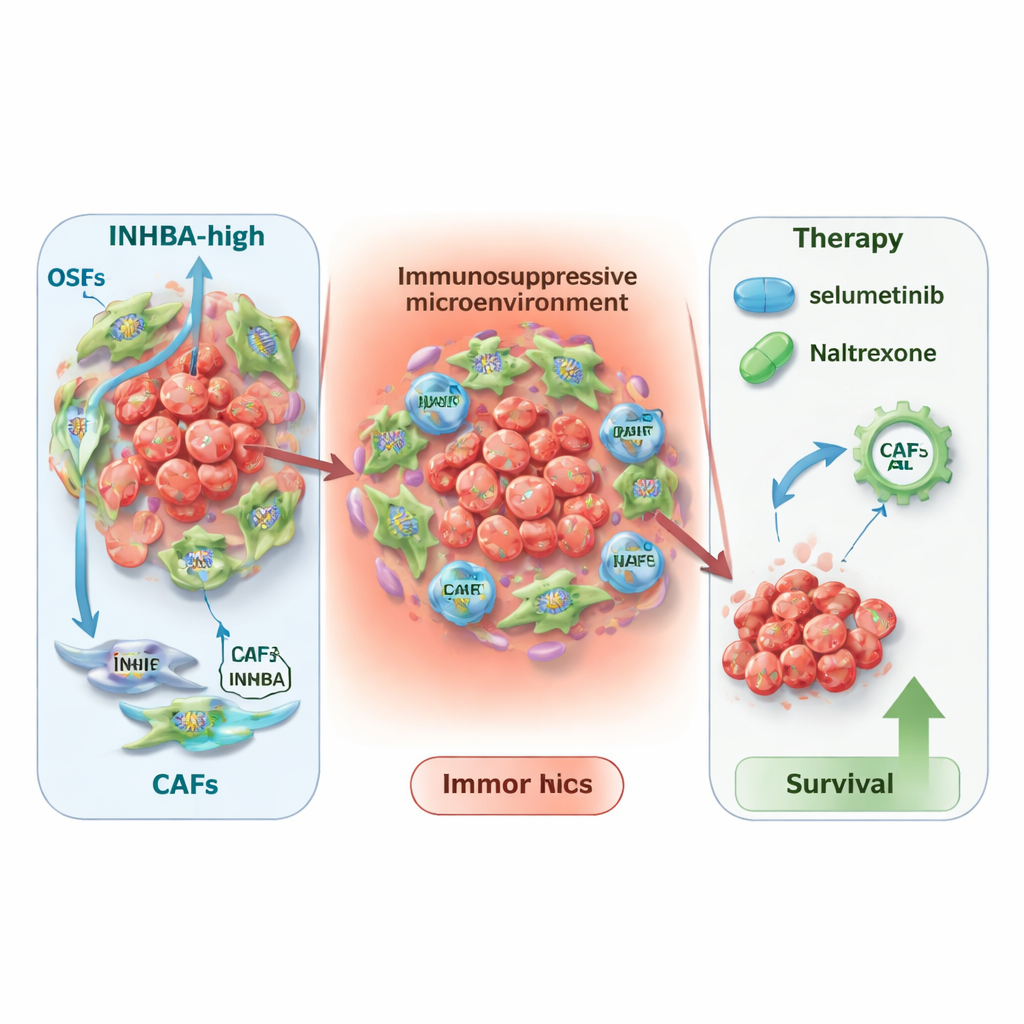
New treatment angles and broader impact
Knowing that INHBA marks a more dangerous tumor neighborhood, the researchers asked how this might guide therapy. In patients receiving immunotherapy, low INHBA levels were linked to better outcomes with common drugs that target PD‑L1 or CTLA4, whereas high INHBA levels seemed to predict better benefit from CAR‑T cell approaches. By mining two large drug‑response databases, they also flagged two existing medicines—selumetinib and naltrexone—as especially promising in tumors with high INHBA. Computer modeling showed that both drugs could bind tightly to the INHBA protein, hinting at a direct effect. Finally, a pan‑cancer analysis revealed that INHBA is elevated and linked to poor survival in many other tumor types, suggesting that its importance extends well beyond mouth cancer.
What this means for patients
In plain terms, this study pins down INHBA as a “master switch” gene that helps deadly mouth tumors grow, spread, and evade the immune system, largely by acting through specialized support cells rather than the cancer cells alone. Measuring INHBA in tumors could help doctors identify high‑risk patients, predict who will benefit from different kinds of immunotherapy, and choose targeted drugs that may work better in this setting. Because INHBA plays a similar harmful role in several other cancers, therapies aimed at this gene and its fibroblast partners could ultimately benefit a wide range of patients, moving care closer to precise, personalized treatment.
Citation: Zhang, X., Sun, M., Qiu, T. et al. INHBA: a mitochondrial-related pan-cell death gene associated with the prognosis and immunity of OSCC. Sci Rep 16, 7642 (2026). https://doi.org/10.1038/s41598-026-38131-4
Keywords: oral squamous cell carcinoma, INHBA, tumor microenvironment, cancer-associated fibroblasts, immunotherapy