Clear Sky Science · en
Quantification of overall tumor burden using longitudinal magnetic resonance imaging improves response assessment in orthotopic murine hepatocellular carcinoma models
Why Tracking Tumors Better Matters
Liver cancer is one of the deadliest cancers worldwide, and it usually grows in livers already damaged by long‑term disease such as scarring or fatty liver. Doctors now treat some patients with powerful immunotherapies that help the body’s own defenses attack tumors. But to know whether these treatments truly work, researchers first need animal studies and, crucially, a precise way to measure how the cancer changes over time inside the body rather than only at the end of an experiment. This study shows how advanced MRI scanning can provide a much more honest picture of tumor growth in mouse models that closely resemble human liver cancer.
Realistic Mouse Models of Liver Cancer
The researchers used two mouse models designed to mirror the main ways human liver cancer develops. In one, mice received a chemical that triggers liver tumors plus repeated injury that causes scarring, resembling cancer that arises in fibrotic or cirrhotic livers. In the other, the same cancer‑causing chemical was paired with a Western‑style high‑fat diet, leading to a fatty, inflamed liver similar to metabolic dysfunction‑associated steatotic liver disease (MASLD), now a major cause of liver cancer worldwide. In both models, many small tumors appeared across the liver, just as they often do in patients, making simple visual inspection or weighing the liver at the end of life an unreliable way to judge overall disease.
Using MRI to See the Full Tumor Load
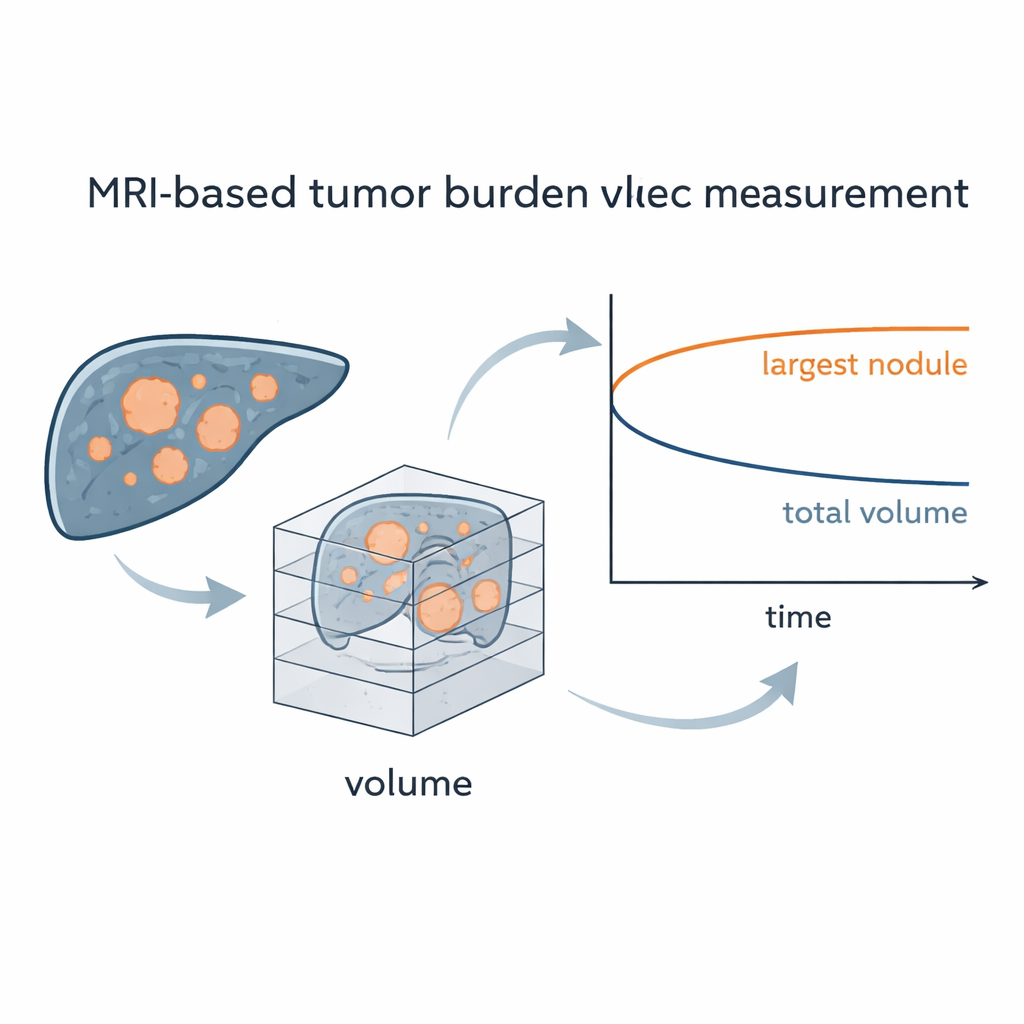
To tackle this, the team turned to high‑resolution magnetic resonance imaging (MRI). They optimized scanning settings so that tiny tumors, as small as one millimeter, stood out clearly against the background of either scarred or fatty liver tissue. For each scan, they traced every visible tumor on each image slice and combined these slices to calculate the total tumor volume inside the liver — a measure they call overall tumor burden. This approach allowed them to follow all tumors in each mouse over many weeks without surgery or repeated tissue sampling, reducing animal use and stress while revealing detailed growth patterns that cannot be seen from a single end‑point measurement.
Testing Immunotherapy in Fibrotic and Fatty Livers
Once early tumors were confirmed by MRI, some mice received an antibody therapy that blocks PD‑L1, a target of widely used immune checkpoint drugs in humans, while others were left untreated. In the fibrosis‑based model, treated mice survived longer than controls, largely because their largest individual tumors grew more slowly. However, when the researchers looked at the MRI‑derived overall tumor burden — the combined volume of all tumors — they found that total cancer volume inside the liver did not clearly shrink with treatment. In other words, focusing only on the biggest nodule suggested a stronger benefit than actually occurred when all tumors were counted together.
Why Fatty Liver Cancer Resists Treatment
The story was different in the MASLD model, where fatty, inflamed livers are thought to alter immune responses. Here, the PD‑L1‑blocking therapy had little detectable effect. Survival times were similar in treated and untreated mice, and both the size of the largest tumor and the total tumor volume increased along nearly overlapping paths. This mirrors growing clinical evidence that human liver cancers arising from metabolic fatty liver often respond poorly to current immunotherapies. Because the MRI method could track all tumors over time, it provided strong support that these mouse models faithfully reproduce the treatment resistance seen in many patients with fatty‑liver‑related cancer.
What This Means for Future Research and Patients
By showing that detailed MRI‑based measurements give a truer picture of how liver tumors respond to therapy than simple diameter or liver weight, this work highlights the risk of overestimating drug benefits when only the largest tumor is followed. The study argues that counting the full tumor burden will make preclinical trials more reliable and more closely aligned with what happens in people. At the same time, repeatedly scanning the same animals over time reduces the number that must be euthanized at multiple checkpoints, improving animal welfare. Together, these advances should help researchers design better immunotherapy studies and, ultimately, select treatments that are more likely to make a real difference for patients with liver cancer.
Citation: Lurje, I., Werner, W., Hilbert, N. et al. Quantification of overall tumor burden using longitudinal magnetic resonance imaging improves response assessment in orthotopic murine hepatocellular carcinoma models. Sci Rep 16, 5247 (2026). https://doi.org/10.1038/s41598-026-38125-2
Keywords: hepatocellular carcinoma, liver cancer, magnetic resonance imaging, immunotherapy, fatty liver disease