Clear Sky Science · en
Effect of polyvinylpyrrolidone molecular weight and concentration on its dual role in the synthesis, stability and antimicrobial activity of silver nanoparticles
Why tiny silver matters in everyday life
From germ‑killing wound dressings to odor‑free sportswear and safer medical devices, products that quietly rely on silver nanoparticles are already part of daily life. But making these ultra‑small particles often involves harsh chemicals that are bad for both people and the environment. This study explores a gentler way to create silver nanoparticles in water using a common, widely used polymer called polyvinylpyrrolidone (PVP) and shows how tweaking this one ingredient can tune particle size, shape, stability, and germ‑killing power.
Making silver safely in water
The researchers set out to see whether PVP alone could play a “dual role” in silver nanoparticle production: acting both as a mild reducing agent (turning dissolved silver ions into solid silver) and as a stabilizer that keeps the particles from clumping. They tested five versions of PVP that differed only in how long their chains were, from very short (10,000, called 10K) to extremely long (1,300,000, called 1300K), and three polymer concentrations in plain water. By gently heating the mixtures and adjusting the solution to a basic pH with sodium hydroxide, they could watch nanoparticle formation in real time using light‑absorption measurements and confirm the resulting particle shapes with electron microscopy. 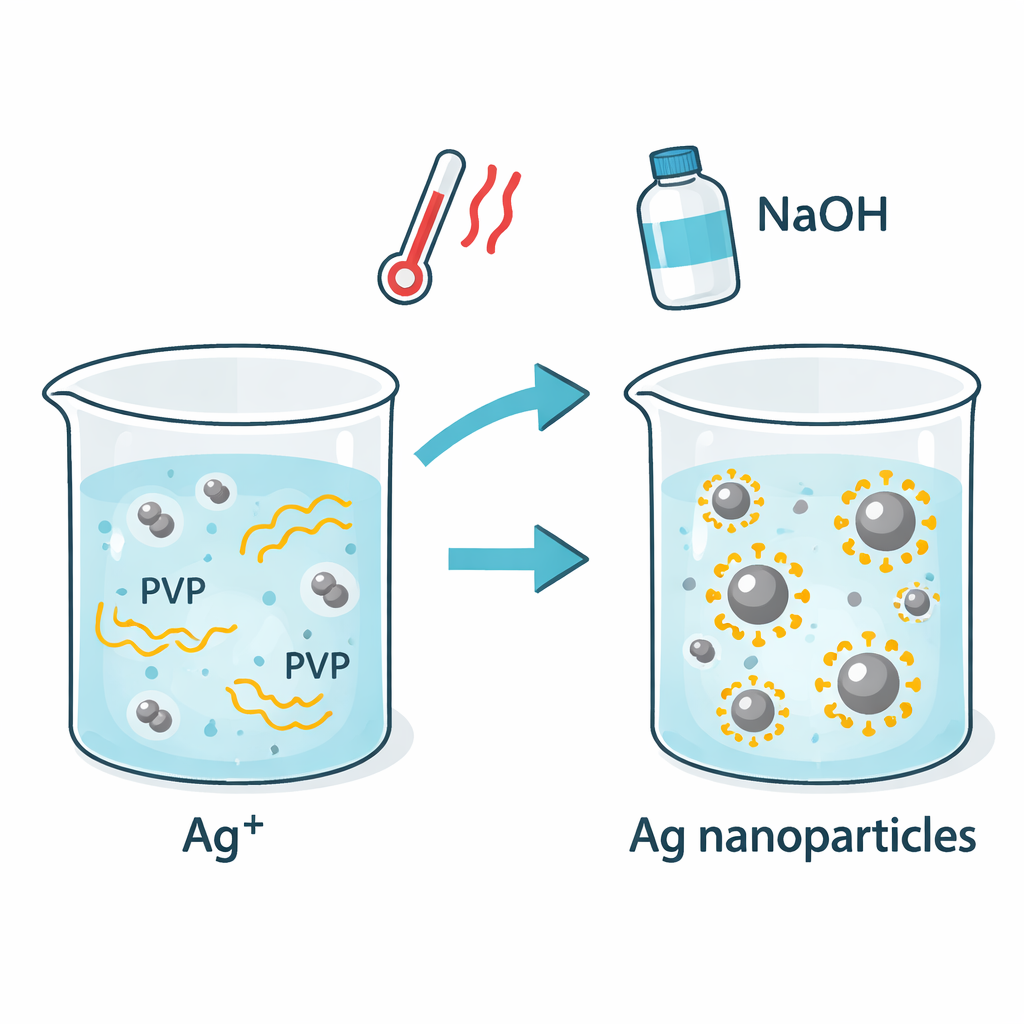
How chain length and acidity shape the particles
Silver nanoparticle formation turned out to be very sensitive to the solution’s pH and to the chain length of PVP. At a relatively high pH of 11, all but the very longest PVP (1300K) produced clear signs of nanoparticle formation within 90 minutes, with short‑chain PVP (10K) working the fastest. At a milder pH of 9, only the two shortest PVPs (10K and 40K) could still reduce silver ions effectively; at neutral pH, almost no particles formed. These results support a mechanism in which PVP’s ring structure rearranges under basic conditions to expose groups that can donate electrons to silver ions. Very long polymer chains, however, create so much crowding in solution that silver ions struggle to reach these reactive sites, so particle formation is strongly suppressed.
Balancing small, round, and stable
Changing how much PVP was present added another layer of control. At lower polymer levels, shorter chains tended to produce many small, mostly spherical particles, while longer chains favored fewer, larger particles and, in some cases, flat triangular or hexagonal “nanoplates.” At higher PVP concentrations, mid‑length chains (around 80K) gave a particularly narrow size distribution, suggesting an optimal balance between helping silver ions react and coating the growing particles to keep them from merging. Across most conditions, typical particle sizes fell between about 17 and 23 nanometers—tens of thousands of times smaller than the width of a human hair. When stored as simple water suspensions, all of these PVP‑coated nanoparticles remained stable and well dispersed for at least six months, indicating that the polymer shell provides strong long‑term protection even without significant electrical charge on the particle surface.
Controlling how strongly the particles fight germs
Because these nanoparticles are often used as antimicrobial agents, the team also tested how well they inhibited two common bacteria: the Gram‑positive Staphylococcus aureus and the Gram‑negative Escherichia coli. They placed drops of nanoparticle suspensions into wells in bacteria‑coated agar plates and measured the clear “inhibition zones” that formed after a day. Surprisingly, a higher PVP concentration around the particles usually led to smaller zones, even when the amount of silver was the same. This suggests that a thicker polymer coating slows the release of silver ions, which many researchers believe are key to killing bacteria. Particle shape also mattered: samples containing more non‑spherical, plate‑like particles (often formed with higher‑molecular‑weight PVP) tended to create larger inhibition zones, especially against the more resistant E. coli. 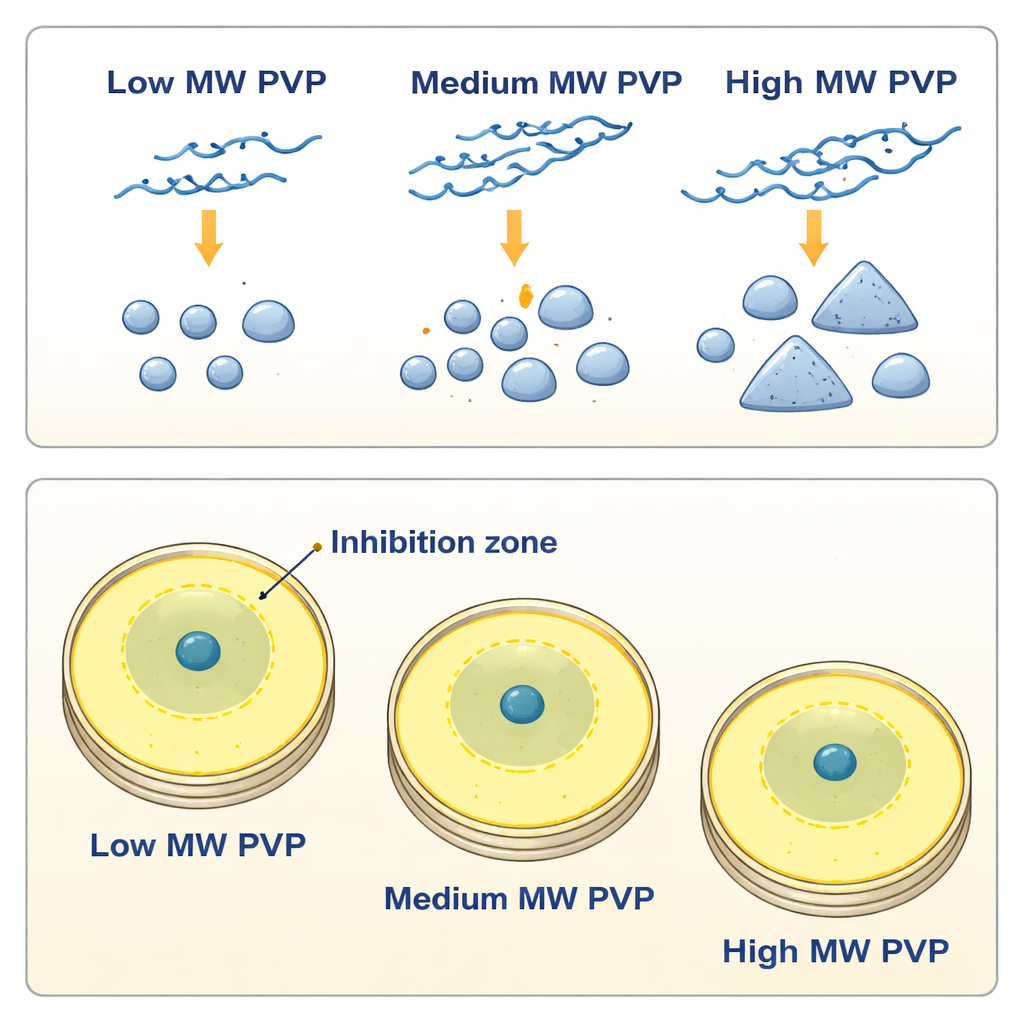
What this means for future silver‑based products
For non‑specialists, the main message is that a single, widely available polymer can be used to both create and stabilize silver nanoparticles in water, avoiding harsher chemical reducers. By carefully choosing the chain length and amount of PVP, manufacturers could “dial in” particles that are small, uniform, long‑lived, and have a desired level of antimicrobial strength, while using a process that is more environmentally friendly. This work offers a toolkit for designing safer silver‑based coatings and materials that resist germs without relying on toxic synthesis routes.
Citation: Rashid, A., Irfan, M., Javid, A. et al. Effect of polyvinylpyrrolidone molecular weight and concentration on its dual role in the synthesis, stability and antimicrobial activity of silver nanoparticles. Sci Rep 16, 7562 (2026). https://doi.org/10.1038/s41598-026-38124-3
Keywords: silver nanoparticles, green synthesis, polyvinylpyrrolidone, antimicrobial coatings, nanomaterials