Clear Sky Science · en
Bovine serum albumin nanoparticles improve bacteriophage stability and antimicrobial activity against Pseudomonas aeruginosa
Why protecting helpful viruses matters
As antibiotic resistance rises, doctors are running out of ways to treat dangerous infections. One stubborn culprit is Pseudomonas aeruginosa, a bacterium that often infects the lungs of people with weakened defenses and can shrug off many drugs. This study explores a creative strategy: using friendly viruses that attack bacteria, called bacteriophages, and protecting them inside tiny protein spheres made from a common blood protein, bovine serum albumin (BSA). The goal is to keep these viruses stable and active long enough to better fight tough lung infections.
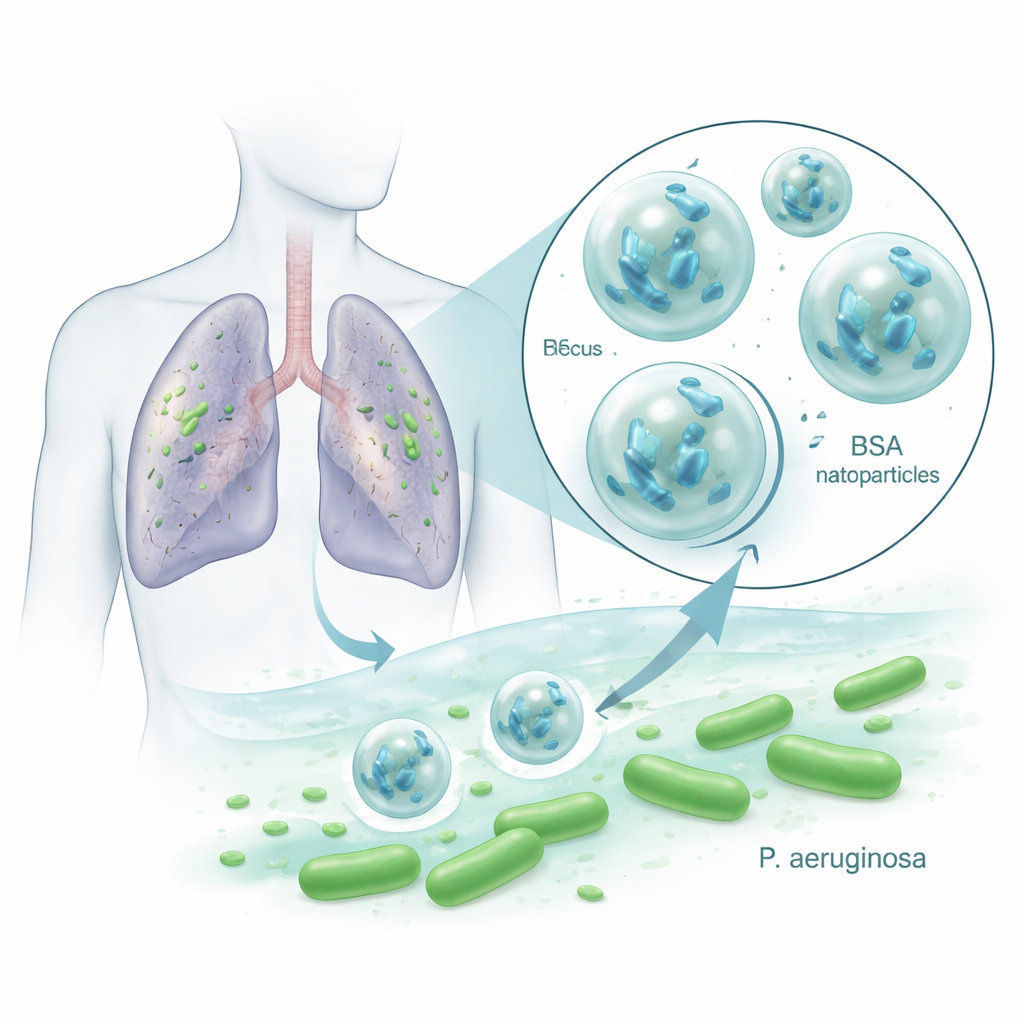
Tiny allies against stubborn lung germs
Pseudomonas aeruginosa is notorious in hospitals because it resists multiple antibiotics and forms slimy biofilms that shield it from treatment. Bacteriophages, or phages, are viruses that infect and burst specific bacteria while leaving human cells and helpful microbes largely untouched. They can multiply wherever their bacterial host is present, making them an appealing complement or alternative to antibiotics. But phages are fragile: heat, acidity, enzymes, and the immune system can quickly inactivate them, which has limited their success in real patients. The researchers asked whether packaging a Pseudomonas-killing phage called VAC1 inside BSA nanoparticles could protect it and make treatments more effective.
Building a protective protein shell
The team first had to design a particle that would not harm the phage. They tested common solvents used to form BSA nanoparticles and found that ethanol and methanol destroyed VAC1, while acetone did not, so they used acetone in their process. They mixed the phage with a BSA solution and then carefully added acetone to make the protein clump into nanoscale spheres, stabilizing them with a crosslinking chemical. These phage-loaded particles, called NPPha, averaged about 220 nanometers in diameter—far smaller than a human cell—and trapped over 95% of the phage inside. Electron microscope images showed irregularly shaped BSA particles containing denser regions that likely corresponded to phage, and tests confirmed that active viruses were slowly released over at least two days at body temperature without losing infectivity.
Stronger bacteria-killing power in the lab
The researchers then compared how well free VAC1 and NPPha controlled Pseudomonas in liquid culture. When bacteria were exposed to NPPha, their growth was suppressed much more strongly than with the same phage given alone or with empty nanoparticles. Over 24 hours, cultures treated with NPPha produced roughly one hundred thousand times more new phage particles than cultures treated with free VAC1, suggesting that the steady release from the nanoparticles created a more sustained virus-bacteria battle. Importantly, the BSA nanoparticles—whether filled with phage or empty—did not harm human liver-derived cells in toxicity tests, supporting their potential safety as a delivery vehicle. Stability experiments at 37 °C showed that free phage rapidly lost activity within two days, while phage inside NPPha remained infective for up to five days.
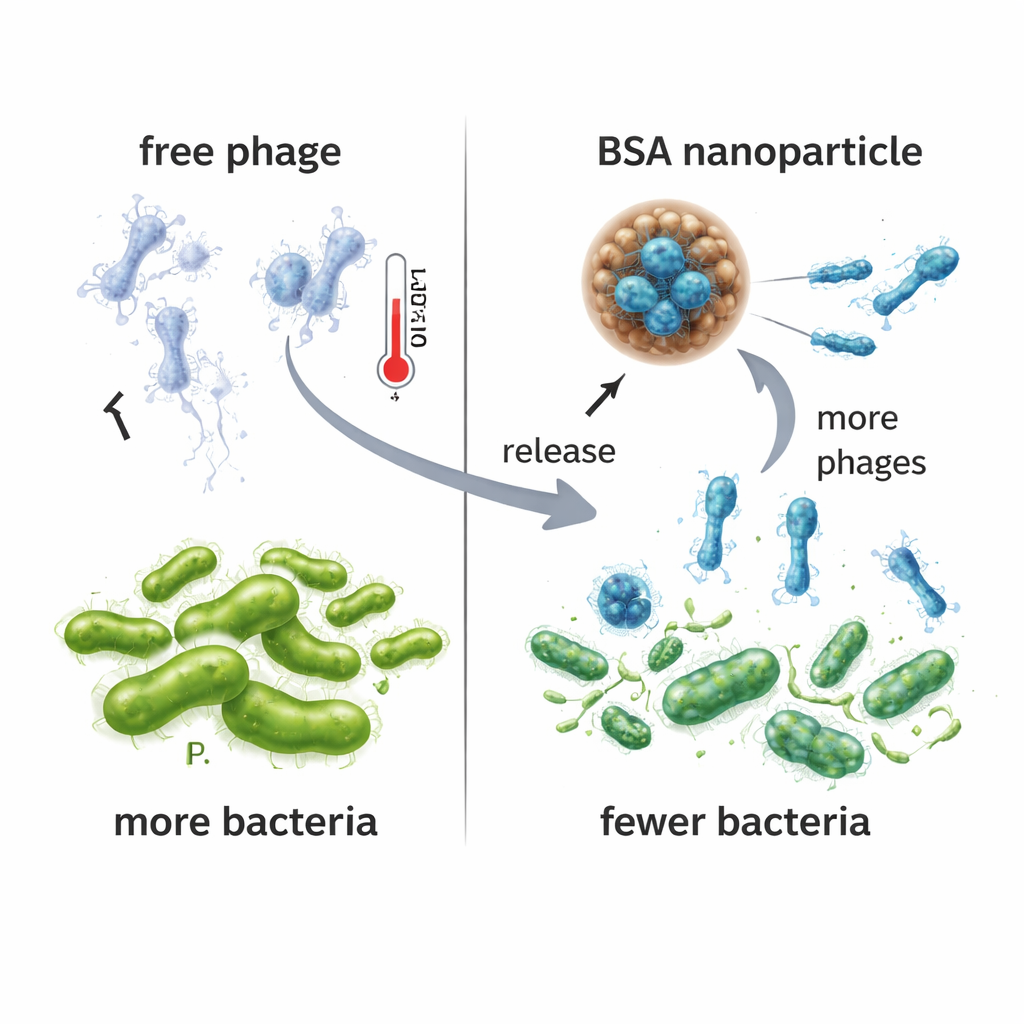
Testing the approach in infected mice
To see whether these benefits carried over to living animals, the team used a mouse model of acute Pseudomonas lung infection. Mice were infected through the nose and, an hour later, treated with NPPha, free VAC1, empty nanoparticles, or a salt solution. In this very severe model, all animals, regardless of treatment, died within 12 hours, so survival did not improve. However, when the researchers examined the lungs, they found that mice given NPPha had fewer bacteria and were more likely to still harbor detectable phage than those treated with free VAC1. Tissue slices from NPPha-treated lungs showed less structural damage, thinner walls between air sacs, and reduced inflammatory cell buildup compared with other infected groups, indicating that the infection was somewhat blunted even though it was not enough to save the animals under these harsh conditions.
What this means for future infection treatments
For non-specialists, the key message is that packaging bacteriophages inside tiny protein-based bubbles can keep them alive and potent longer, helping them better attack hard-to-treat bacteria like Pseudomonas aeruginosa. In dishes and in mouse lungs, BSA nanoparticles boosted phage numbers, reduced bacterial growth, and lessened lung damage, though they did not yet prevent death in a very aggressive infection model. The work suggests that albumin nanoparticles offer a simple, low-cost, and seemingly safe way to stabilize therapeutic phages. With refined dosing, timing, and perhaps use in less extreme or more chronic infections, such nano-packaged phages could become a valuable tool alongside antibiotics in the fight against multidrug-resistant bacteria.
Citation: Cunha, G.A.d., Marangoni, G.S., Durante, M.F.R. et al. Bovine serum albumin nanoparticles improve bacteriophage stability and antimicrobial activity against Pseudomonas aeruginosa. Sci Rep 16, 7146 (2026). https://doi.org/10.1038/s41598-026-38106-5
Keywords: phage therapy, nanoparticles, Pseudomonas aeruginosa, antibiotic resistance, lung infection