Clear Sky Science · en
Machine learning interatomic potential for the structural properties of iron oxides
Why Rusty Rocks Matter
Iron oxides – the minerals that give rust its color – quietly support much of modern life. They are the main source of iron for steel, key ingredients in batteries and solar cells, and even help clean polluted water. Yet, despite their importance, we still struggle to predict how these materials behave under real-world conditions, especially at the atomic level. This article describes how researchers used modern artificial intelligence to build a fast, precise digital model of one crucial iron oxide, hematite, opening the door to more reliable virtual experiments on everything from ore processing to clean energy devices.
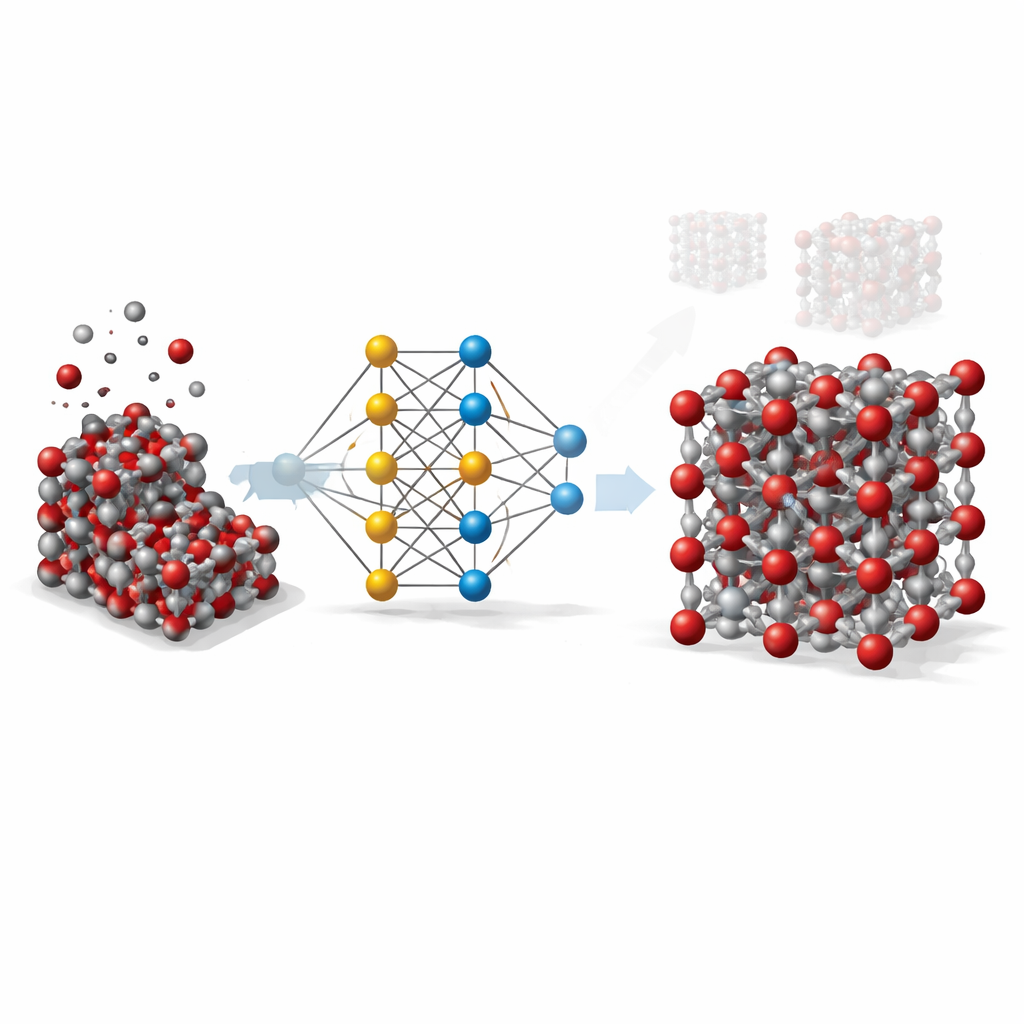
From Costly Calculations to Smart Shortcuts
To understand a solid like hematite in detail, scientists ideally rely on quantum-mechanical methods that track how electrons and atoms interact. These methods, though very accurate, are so computationally expensive that they are impractical for simulating large samples or long time spans. Classical models, in contrast, are fast but crude: they rely on simple formulas tuned for specific situations and often fail when temperatures, pressures, or crystal shapes change. The work presented here aims to bridge this gap by using machine learning to imitate the accuracy of quantum calculations while keeping the speed of traditional models.
Teaching a Neural Network About Atoms
The team built what is known as a graph neural network potential for hematite. In this approach, each atom is treated as a node in a network, and the bonds and neighboring atoms are the connections between nodes. To teach this network how atoms in hematite push and pull on each other, the researchers first generated thousands of atomic snapshots using standard simulations across a wide range of temperatures, pressures, and crystal distortions, including both bulk crystals and exposed surfaces. They then used a high-level quantum method (DFT+U) to compute the energy, forces, and internal stresses for each snapshot, and trained the neural network to reproduce these values as closely as possible.
Checking the Model Against Reality
Once trained, the new potential – called Fe-MLIP – was rigorously tested. The authors compared its predictions for basic structural quantities such as lattice dimensions and how the crystal stretches under stress with both experiments and several widely used classical models. Fe-MLIP reproduced the known crystal structure of hematite within a few percent and captured its elastic behavior almost as well as direct quantum calculations, clearly outperforming other force fields for many properties. It also did well on more subtle tests, such as how the material expands with temperature and how its atoms vibrate, which are important for heat transport and spectroscopy. These vibrational frequencies, never shown explicitly during training, were closer to measured values than those from competing models.
Reaching Beyond a Single Mineral
The researchers then explored how far this hematite-based model could be pushed. They applied it to related iron oxides – maghemite and magnetite – that share similar atomic building blocks but differ in crystal arrangement and iron charge states. Even though Fe-MLIP was not trained on these phases, it produced reasonable values for their lattice sizes and stiffness, often matching or beating specialized classical models. The potential also captured the relative stability of key crystal surfaces and even trends in the energy cost of creating atomic vacancies, features crucial for understanding corrosion, catalysis, and battery performance.
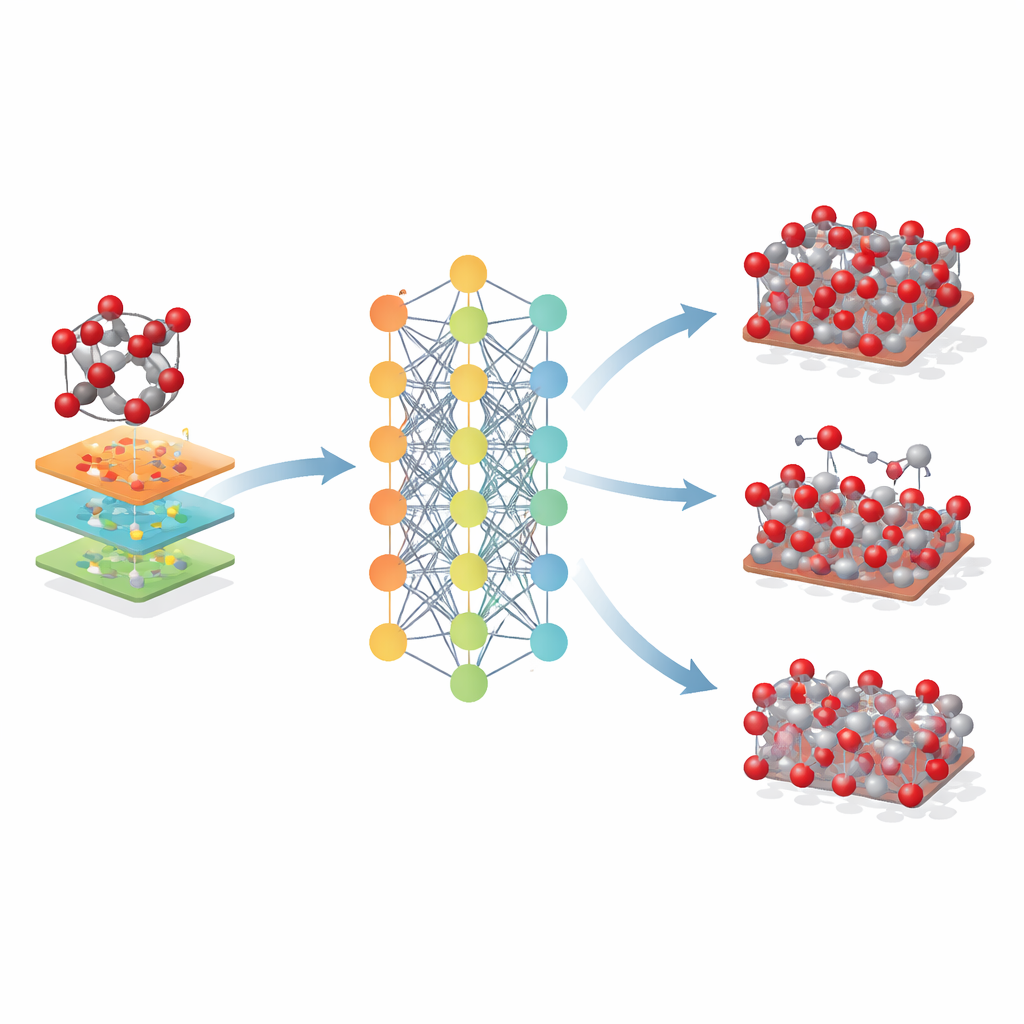
What This Means for Future Materials Design
For non-specialists, the takeaway is that this work provides a powerful new "digital twin" for iron oxides. The Fe-MLIP model allows researchers to run large, long simulations of hematite and related materials with nearly quantum-level reliability but at a fraction of the cost. While it inherits some limitations of the underlying quantum method and is currently focused on iron and oxygen, it already enables more realistic studies of how these minerals respond to stress, heat, surfaces, and defects. In practical terms, such a tool can accelerate the design of better steelmaking processes, more efficient catalysts and batteries, and improved environmental technologies that rely on iron oxides – all by letting scientists test ideas on a computer before turning to the lab or the mine.
Citation: Torres, A., de Oliveira, A.B., Barbosa, M.d.S. et al. Machine learning interatomic potential for the structural properties of iron oxides. Sci Rep 16, 8576 (2026). https://doi.org/10.1038/s41598-026-38096-4
Keywords: hematite, iron oxides, machine learning potential, graph neural networks, molecular dynamics