Clear Sky Science · en
Obinutuzumab induces lysosomal destabilization via sphingomyelin-dependent inhibition of TRPML2
Why this antibody story matters
For many people with B‑cell lymphomas, modern antibody drugs have turned a once-uniformly deadly diagnosis into a treatable disease. Yet not all antibodies are created equal: some kill cancer cells more powerfully and more reliably than others. This paper explores why one such drug, obinutuzumab, is better at directly killing lymphoma cells than its predecessor rituximab, by zooming in on an unexpected culprit deep inside the cell—the cell’s own recycling centers, called lysosomes, and a fine balance of fats and ion channels in their membranes.
From surface target to internal self‑destruct
Both rituximab and obinutuzumab recognize the same target on B cells, a protein called CD20. But the authors show that obinutuzumab is taken up into the cell much faster and more efficiently. Once bound to CD20 on the cancer cell surface, the antibody–receptor complexes are pulled inward through an endocytic process and delivered to acidic compartments, including lysosomes. There, obinutuzumab triggers lysosomal membrane permeabilization: the normally tough lysosomal membrane becomes leaky, allowing digestive enzymes such as cathepsins to spill into the cell and set off cell‑death pathways. Changes in the salt and water balance of these compartments, which make lysosomes swell, were found to greatly amplify this lethal effect.
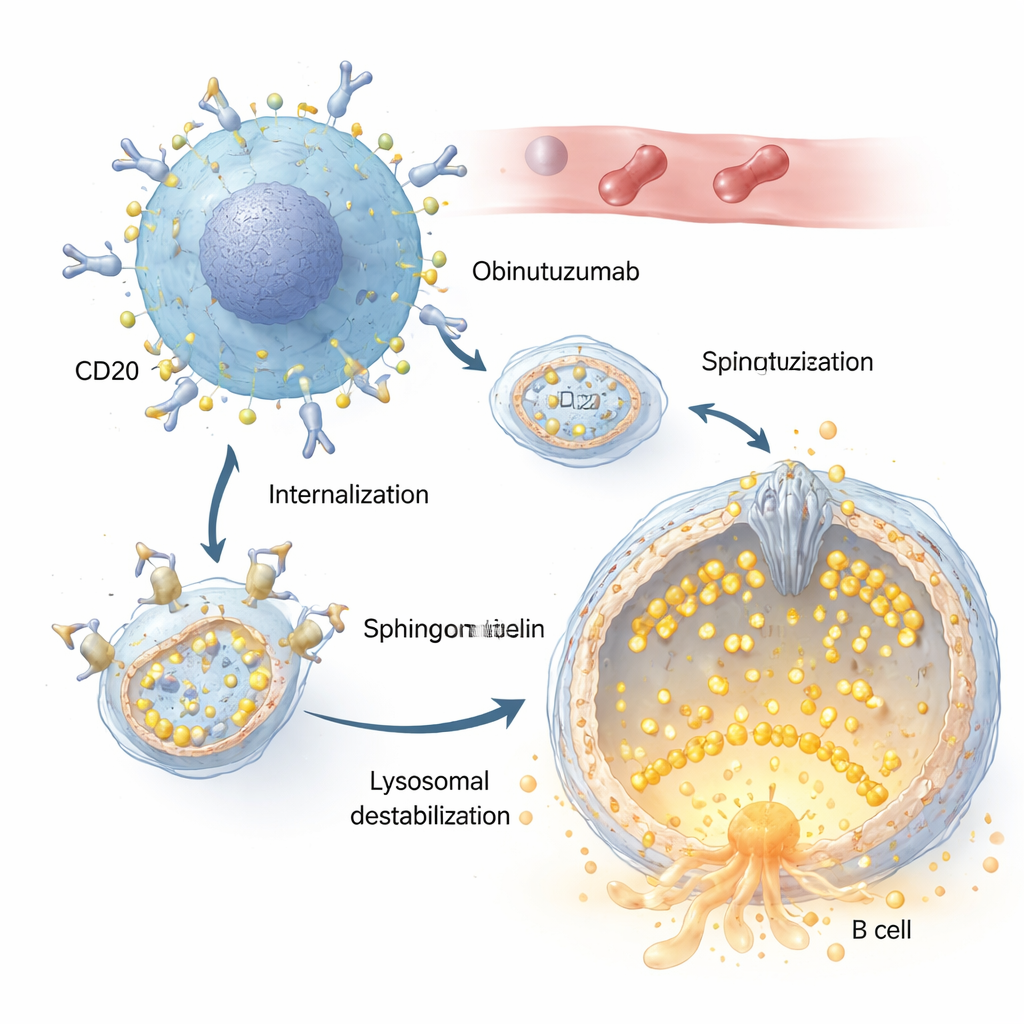
The role of a tiny gatekeeper channel
Central to this story is TRPML2, a calcium channel embedded in the lysosomal membrane. Under normal conditions, TRPML2 helps lysosomes cope with mechanical and osmotic stress by releasing calcium, which supports membrane repair, trafficking, and volume control. Using a combination of fluorescent calcium dyes and engineered calcium‑sensing versions of TRPML2, the researchers show that obinutuzumab rapidly shuts down this channel: within minutes of treatment, a drug that normally opens TRPML channels no longer produces a calcium burst. Genetic approaches that reduce TRPML2 levels, or small‑molecule inhibitors that block TRPML activity, both make lymphoma cells more vulnerable to obinutuzumab, leading to more lysosomal leakage and higher rates of direct cell death.
How membrane fats tip the balance
The study then asks what links antibody uptake to channel shutdown. Attention turns to sphingomyelin, a lipid (fat) that is abundant in cell membranes and is known to interfere with TRPML channels. Using a fluorescent sphingomyelin‑binding probe, the authors find that obinutuzumab‑containing vesicles are rich in sphingomyelin once they reach acidic compartments, much more so than vesicles containing rituximab. When cells are treated with sphingomyelinase, an enzyme that trims sphingomyelin, TRPML2’s calcium‑releasing activity is restored even in the presence of obinutuzumab. Under these conditions, lysosomes are less prone to leak, and fewer cells die, strongly suggesting that accumulated sphingomyelin disables TRPML2 and primes lysosomes for rupture.
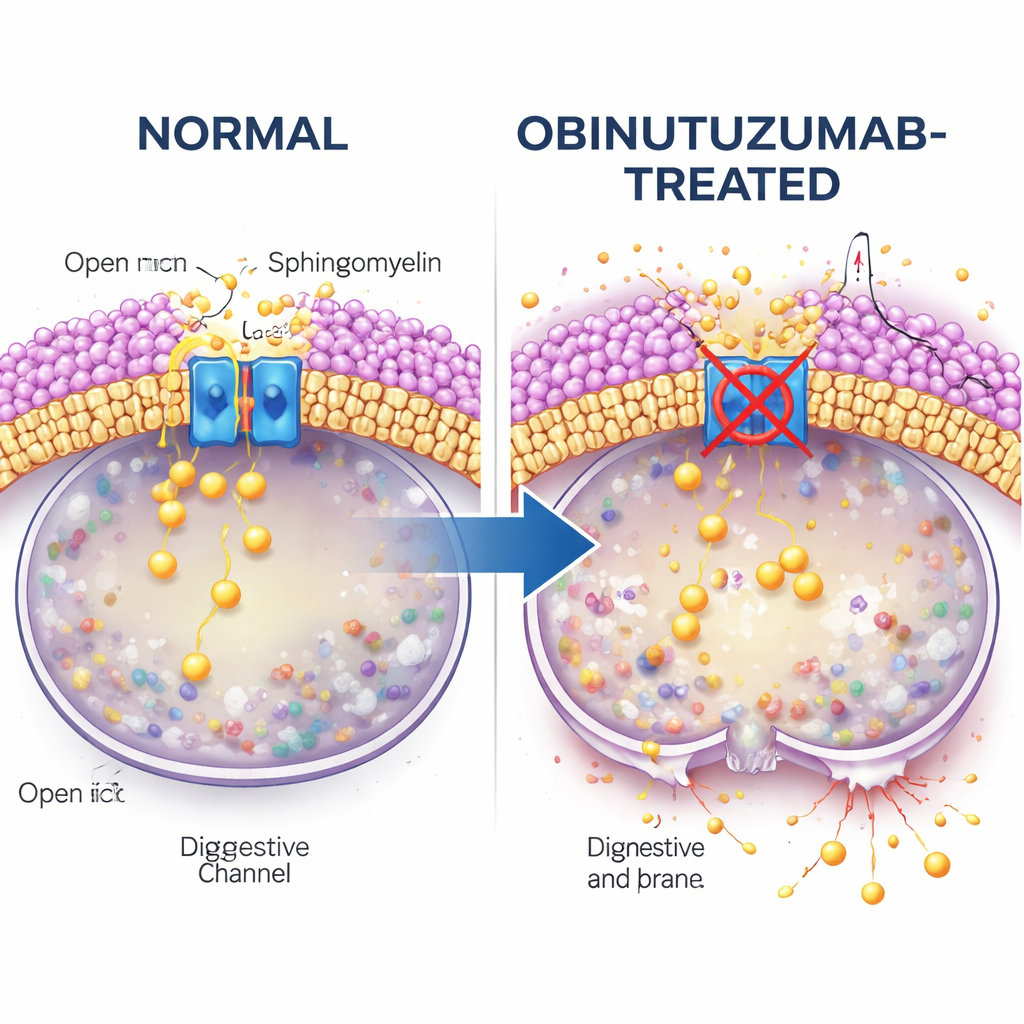
Traffic routes and cholesterol checkpoints
The path obinutuzumab takes into the cell also matters. Electron microscopy and pH‑sensitive antibody labeling reveal that obinutuzumab leaves the cell surface and enters intracellular vesicles more quickly than rituximab. When the researchers interfere with specific uptake routes, they find that blocking a cholesterol‑dependent form of endocytosis (with a compound called filipin) prevents obinutuzumab from turning off TRPML2, and partially reduces lysosomal damage and cell death. Other endocytosis inhibitors do not have this protective effect. This points to a specialized, cholesterol‑ and sphingomyelin‑rich pathway that delivers obinutuzumab to lysosomes in a way that sets up TRPML2 inhibition and subsequent membrane destabilization.
What this means for future cancer treatments
In plain terms, this work shows that obinutuzumab kills B‑cell lymphomas so efficiently because it forces their lysosomes into a fragile state. By driving antibody–CD20 complexes into sphingomyelin‑rich compartments, the drug indirectly switches off a protective calcium channel, TRPML2. Once that safeguard is gone, lysosomes are more likely to burst under stress, spilling corrosive enzymes that dismantle the cancer cell from within. Understanding this lipid–ion channel axis does more than explain why obinutuzumab outperforms rituximab; it also suggests new strategies to make antibody therapies stronger—for example, by pairing them with drugs that alter sphingolipids or boost TRPML2 activity to fine‑tune how and when tumor cell lysosomes self‑destruct.
Citation: Oh, J., Jin, N., Kwon, S. et al. Obinutuzumab induces lysosomal destabilization via sphingomyelin-dependent inhibition of TRPML2. Sci Rep 16, 7079 (2026). https://doi.org/10.1038/s41598-026-38087-5
Keywords: obinutuzumab, B-cell lymphoma, lysosome, sphingomyelin, TRPML2