Clear Sky Science · en
Enhancing wound healing with synergistic dual-drug electrospun roflumilast and L-arginine loaded PLA/PVA nanofibers through fabrication, optimization, and in vivo assessment
Why faster healing bandages matter
Anyone who has dealt with a deep cut, surgical incision, or a chronic sore knows that waiting for skin to heal can be slow, painful, and risky. Doctors are searching for dressings that do more than just cover a wound – they want bandages that actively calm inflammation, feed new tissue, and keep the area moist but stable. This study explores a new type of “smart” bandage made from ultra-fine fibers that carry two different medicines at once, designed to help skin close faster and with better-quality repair.
A tiny fiber web that mimics skin support
The researchers built their bandage from an airy mat of nanofibers – strands thousands of times thinner than a human hair. These fibers form a breathable web that resembles the skin’s natural support structure, giving cells something to cling to as they grow. To make this web, the team used a technique called electrospinning: liquid polymer mixtures are drawn out into long, thin fibers by a high-voltage field and collected as a soft sheet. One solution was based on PLA, a sturdy, slowly breaking-down plastic already used in medical devices. The other was PVA, a water-loving material that swells and helps keep wounds moist. 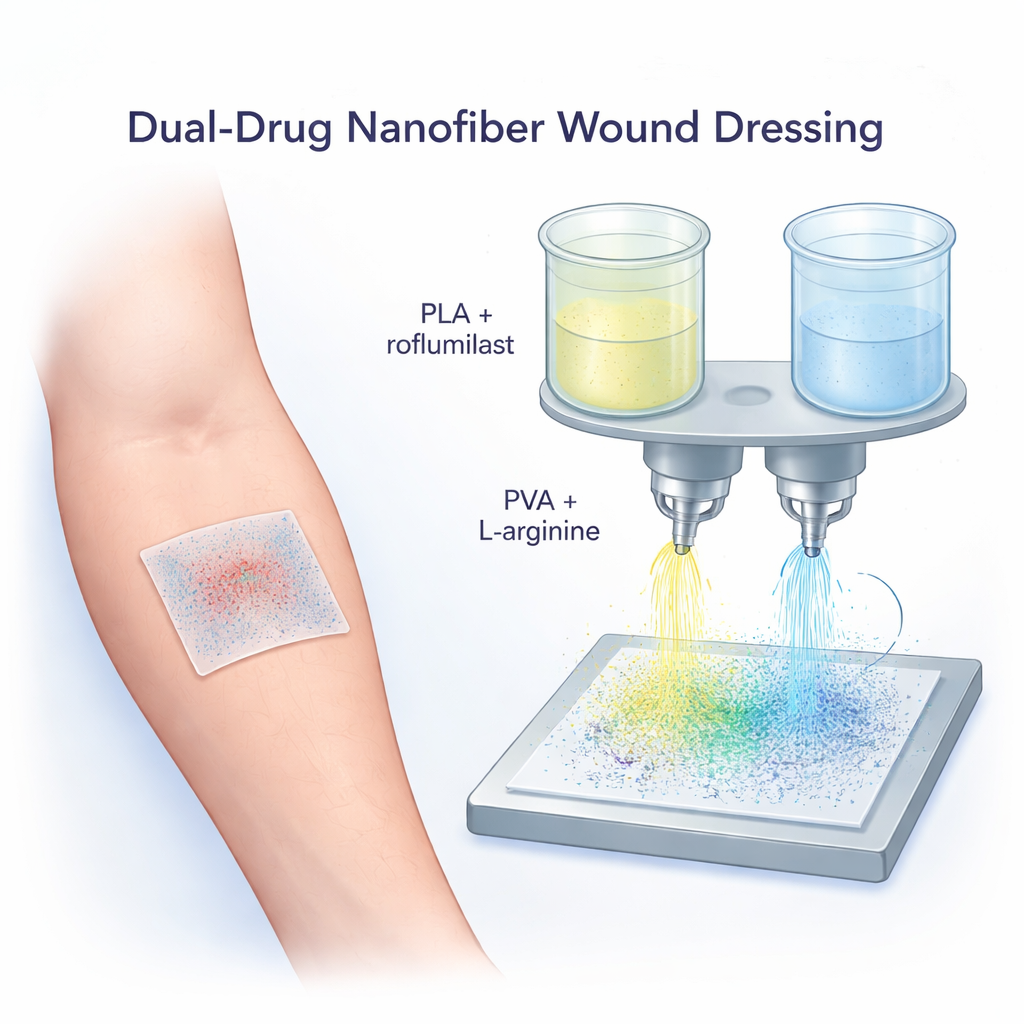
Pairing two drugs for a one-two healing effect
On top of this clever fiber design, the team loaded each polymer with a different medicine. The PLA fibers carried roflumilast, a drug better known for treating lung disease and psoriasis by calming inflammation inside the body. The PVA fibers carried L-arginine, a natural amino acid that the body uses to make nitric oxide, which boosts blood flow, supports immune cells, and helps build collagen – all key steps in skin repair. By spinning both solutions at the same time through two nozzles, they created a single mat where the two drugs sit side by side in separate but interwoven fibers, ready to be released in a coordinated way.
Testing the structure and water-handling of the bandage
Before testing on animals, the authors needed to show that the materials were safe, stable, and well-mixed. Using electron microscopes, they observed smooth, bead-free fibers with consistent thickness, even after drug loading. Chemical fingerprinting tools confirmed that both drugs were physically trapped inside the PLA/PVA matrix without forming unwanted new compounds. X-ray analysis showed that once inside the fibers, the drugs switched from their usual crystalline form to a more disordered, “amorphous” state, which often dissolves faster and more evenly. The team also measured how the mats soaked up water. The dual-drug version swelled quickly – taking up over six times its dry weight at first – then settled to a steady level, meaning it can absorb wound fluid and stay moist without turning into a mushy, fragile gel. 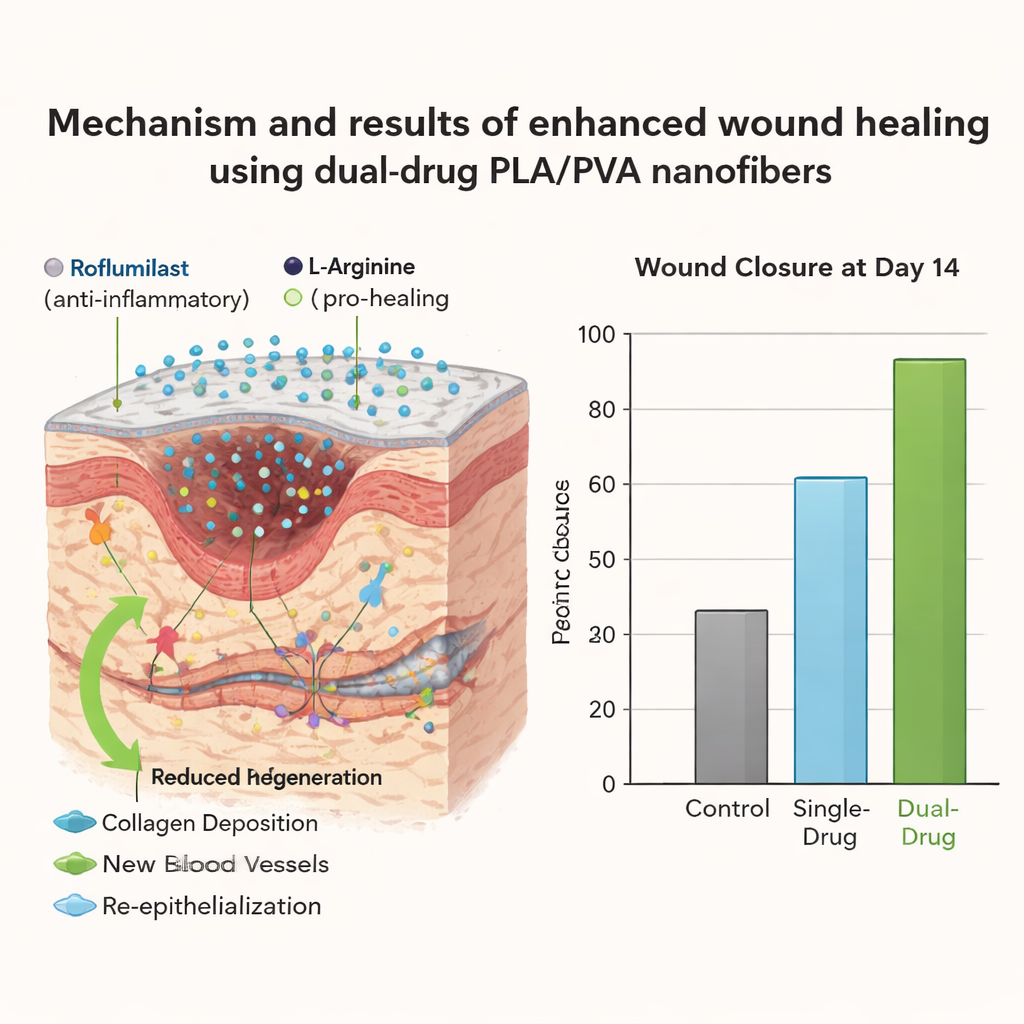
Putting the smart bandage to the test on real wounds
To see whether this design actually improves healing, the scientists treated small, circular skin wounds on rats with different versions of the fiber mats: no drug, roflumilast only, L-arginine only, or both drugs together. Another group received only plain gauze. Over two weeks, they photographed the wounds and measured how much of the original open area had closed. All medicated mats worked better than gauze alone, but the dual-drug bandage clearly stood out. By day 14, wounds covered with the combined roflumilast and L-arginine fibers were almost fully closed, reaching roughly 99.8% healing, compared with much slower closure in untreated and single-drug groups.
Looking inside the skin to judge repair quality
Healing is not just about closing a gap – the quality of the new tissue matters. When the team examined thin slices of skin under the microscope, the plain and some single-drug groups still showed “granulation tissue,” a sign that repair was incomplete, although the surface looked closed. In contrast, skin treated with roflumilast alone showed more mature scar tissue, and the dual-drug treatment performed best of all. These samples displayed dense, organized fibrous tissue, full resurfacing of the outer skin layer, no leftover granulation tissue, and only mild lingering inflammation, suggesting a more complete and robust repair.
What this could mean for future bandages
For non-specialists, the message is straightforward: by combining an anti-inflammatory medicine and a pro-healing nutrient inside a carefully engineered fiber web, this study created a bandage that helped wounds in rats close faster and more completely than standard dressings or single-drug versions. While further testing in humans is needed, the approach shows how next-generation bandages could quietly deliver the right mix of signals directly where they are needed, turning a simple covering into an active partner in wound healing – and potentially improving recovery for patients with difficult-to-heal skin injuries.
Citation: Salim, S.A., Elbadry, A.M.M., Abdelazim, E.B. et al. Enhancing wound healing with synergistic dual-drug electrospun roflumilast and L-arginine loaded PLA/PVA nanofibers through fabrication, optimization, and in vivo assessment. Sci Rep 16, 7481 (2026). https://doi.org/10.1038/s41598-026-38086-6
Keywords: wound healing, nanofiber bandages, dual-drug delivery, roflumilast, L-arginine