Clear Sky Science · en
Integrative bioinformatics analyses of mitochondrial dysfunction-related genes in human non-obstructive azoospermia
Why this research matters for men and families
Many couples struggling to conceive eventually discover that the problem lies in the complete absence of sperm in the man’s semen, a condition called non-obstructive azoospermia. For these men, options are limited and often involve painful testicular surgery with uncertain results. This study asks a fundamental question: could tiny structures inside cells, the mitochondria—often called the cell’s power plants—hold the key to understanding, diagnosing, and eventually treating this severe form of male infertility?
The most severe kind of male infertility
Non-obstructive azoospermia (NOA) is the harshest diagnosis in male infertility. Unlike obstructive cases, where sperm are made but blocked from leaving the body, men with NOA often do not make functioning sperm at all. Current treatment usually relies on microscopic testicular surgery to search for rare pockets of sperm, yet less than half of patients benefit. Even when sperm are found, the underlying defects may still cause failed treatments or raise concerns about passing problems to future children. Despite its impact, in about half of NOA cases doctors still do not know the true cause. The authors of this paper set out to look beyond hormones and chromosomes and instead examine how the cell’s energy system might be failing in the testes of these men.
Looking for patterns in thousands of genes
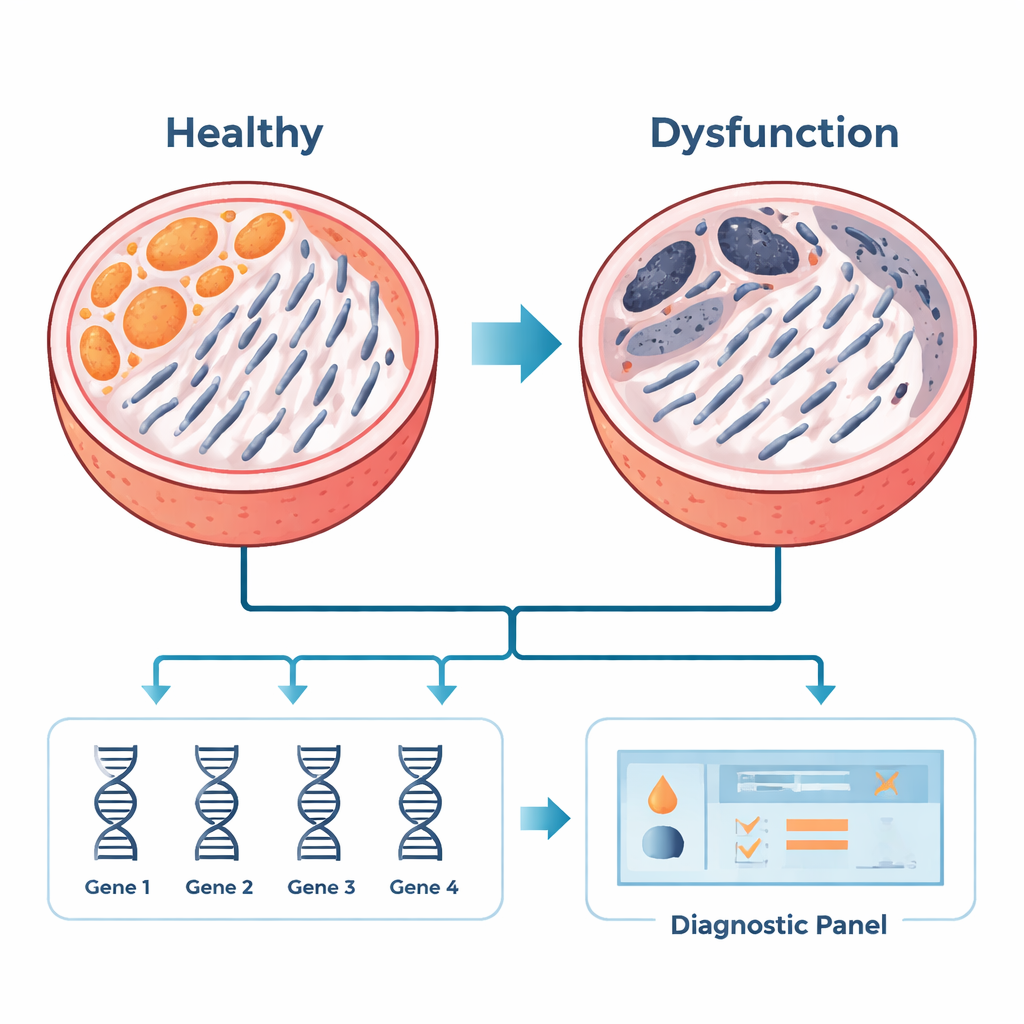
Modern gene chips can measure the activity of thousands of genes at once. The researchers gathered three existing datasets of testicular tissue from men with NOA and from men with normal sperm production. Using bioinformatics—advanced computer analyses of biological data—they compared which genes were turned up or down in NOA. They then focused specifically on genes connected to mitochondria. By cross-referencing their results with a curated mitochondrial gene list, they homed in on 35 genes whose behavior suggested that mitochondrial function is disrupted in NOA. Network analyses, which map how proteins interact with each other, showed that a handful of these genes sit at central “hubs,” coordinating key energy and survival processes in sperm-forming cells.
Six key genes and a potential bloodless test
Among the 35 mitochondrial dysfunction-related genes, six repeatedly stood out as hubs: COX7A1, COX7A2, COX7B2, MRPS15, AURKAIP1, and PDHA2. These genes help mitochondria produce energy, control cell division, and manage stress. In testis samples from additional patients, the team confirmed that one of these genes, COX7A1, was increased in NOA, while the others were reduced. Using four of the strongest candidates—COX7A1, COX7A2, MRPS15, and AURKAIP1—they built a statistical model that could distinguish NOA tissue from normal tissue with high accuracy in existing datasets. Although this work used testicular tissue, the long-term goal is to adapt such gene panels to more accessible samples, like semen-derived cells or vesicles, which could one day help doctors screen patients before resorting to biopsy.
Immune cells and control switches behind the scenes
Beyond genes alone, the study explored how these mitochondrial genes might be regulated and how the immune system might be involved. The authors predicted small regulatory molecules (microRNAs) and transcription factors that could act as on–off switches for the six hub genes, sketching out a complex control network that future lab experiments can test. They also analyzed the mix of immune cells present in testicular tissue. Men with NOA showed higher levels of certain T cells and resting mast cells, and fewer naïve B cells and neutrophils, pointing to subtle immune imbalances in the testis environment. Together, these findings suggest that failing energy production, disturbed cell regulation, and altered local immunity may all converge to disrupt sperm formation.
What this means for patients and future care
To a non-specialist, the core message is that this research highlights mitochondria—the cell’s power plants—as important players in a devastating form of male infertility. By pinpointing a small set of genes linked to mitochondrial health, the study provides promising leads for new diagnostic tools that could, in the future, reduce the need for invasive biopsies and improve how patients are counseled and treated. While the current work is based mainly on computer analysis and a small patient group, it lays essential groundwork. Larger clinical studies and laboratory experiments will be needed to confirm how these genes cause sperm failure and to turn these molecular clues into real-world tests or therapies for men with non-obstructive azoospermia.
Citation: Liu, Q., Wu, H., You, J. et al. Integrative bioinformatics analyses of mitochondrial dysfunction-related genes in human non-obstructive azoospermia. Sci Rep 16, 7295 (2026). https://doi.org/10.1038/s41598-026-38077-7
Keywords: male infertility, azoospermia, mitochondria, biomarkers, spermatogenesis