Clear Sky Science · en
Functional properties of skeletal myotube-derived extracellular vesicles based on microRNA profiles: a comparative analysis with mesenchymal stem cell-derived extracellular vesicles
Muscle Messages in Tiny Packages
Our muscles do far more than move us—they constantly send molecular "text messages" throughout the body. This study explores how tiny particles released by muscle cells compare with similar particles from widely used stem cells for future therapies. By decoding the genetic signals these particles carry, the authors ask a practical question: for which kinds of diseases might muscle-derived particles be better than stem-cell-derived ones?
Tiny Couriers Traveling Through the Body
All cells release microscopic bubbles called extracellular vesicles, or EVs. These are nano-sized packages wrapped in fat, loaded with molecular cargo. A key cargo type is microRNAs—short RNA snippets that help dial genes up or down, shaping how cells behave. Skeletal muscle, the tissue that powers movement, turns out to be the body’s largest secretory organ and can release large amounts of EVs, especially when stimulated in noninvasive ways such as ultrasound. Mesenchymal stem cells, in contrast, are already a workhorse in clinical trials, with their EVs being tested for tissue repair and calming inflammation.
Reading the Genetic Code of the Cargo
Instead of testing EVs directly in animals or people, the researchers reanalyzed existing sequencing datasets that catalog which microRNAs are present in EVs from mouse skeletal myotubes (lab-grown muscle fibers) and from bone-marrow-derived mesenchymal stem cells. Muscle EVs had a few dominant microRNAs, such as miR-206-3p and miR-378a-3p, making up more than 60 percent of their cargo, while stem cell EVs carried a more even mix, including the broadly acting let-7 family. Because each microRNA can influence many target genes, and many microRNAs can converge on the same pathway, the team built computational methods to understand how whole profiles—not single molecules—might reshape cellular programs.
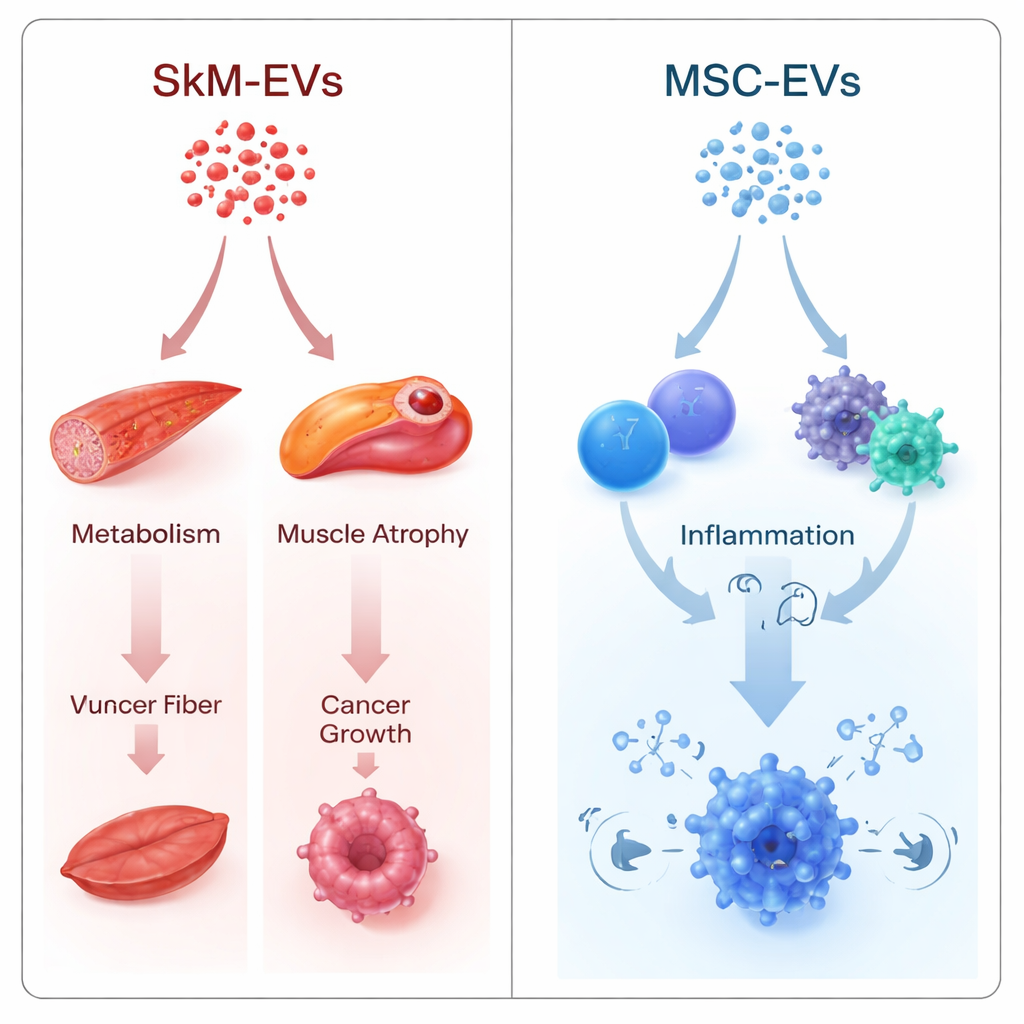
Pathways Linked to Muscle, Metabolism, and Cancer
The authors used two complementary strategies. One compared muscle and stem cell EVs head-to-head, asking where their microRNAs differed. The other treated each EV type separately, converting its microRNA profile into a gene-level "impact score" that estimates how strongly the cargo could damp down particular pathways. Despite different mathematics, both approaches pointed in the same direction. Muscle-derived EVs showed a predicted tendency to suppress pathways tied to muscle wasting (FoxO and TGF-β signaling), blood sugar control and energy balance (FoxO, mTOR, AMPK), and tumor growth pathways (ErbB signaling and a group labeled "microRNAs in cancer"). Many of the most influential targets were central regulators of gluconeogenesis in the liver, key drivers of muscle atrophy, and core cancer-promoting genes.
Stem Cell Vesicles and the Immune System
Stem cell EVs told a different story. Their microRNA cargo was predicted to act more strongly on immune-related routes: NF-κB signaling, cytokine–receptor interactions, B and T cell receptor signaling, and Th17 cell differentiation. These are central to how the body senses infection, turns inflammation on and off, and shapes immune cell identity. This bias lines up with growing experimental evidence that stem cell EVs can broadly modulate immune responses and are being tested in conditions where calming inflammation is crucial, such as autoimmune and inflammatory diseases.
From Computer Predictions to Future Treatments
Overall, the work suggests that muscle-derived EVs may be naturally suited to conditions involving muscle wasting, disturbed metabolism, or unchecked cell growth, while stem cell EVs may be better matched to diseases driven by misfiring immunity. The results are drawn entirely from computational analysis of existing data and do not yet prove benefit in patients, and they do not account for other EV cargo such as proteins or lipids. Still, by turning complex microRNA profiles into pathway-level "fingerprints," this study offers a roadmap for matching EV sources to disease types and helps prioritize which combinations are most promising to test in the lab and, eventually, in the clinic.
Citation: Kawamoto, Y., Yamaguchi, A., Ma, X. et al. Functional properties of skeletal myotube-derived extracellular vesicles based on microRNA profiles: a comparative analysis with mesenchymal stem cell-derived extracellular vesicles. Sci Rep 16, 7436 (2026). https://doi.org/10.1038/s41598-026-38076-8
Keywords: extracellular vesicles, skeletal muscle, microRNA, mesenchymal stem cells, cell signaling pathways