Clear Sky Science · en
Comparative analysis of microRNA expression in serum-derived extracellular vesicles from sudden infant death syndrome cases
Why Tiny Messages in Blood May Hold Clues to Tragic Infant Deaths
Sudden infant death syndrome (SIDS) remains one of the most frightening diagnoses because it often strikes seemingly healthy babies in their sleep, without warning or explanation. This study explores whether microscopic particles circulating in an infant’s blood carry telltale molecular messages from the heart that could help explain why some babies die suddenly—and perhaps one day guide better diagnosis or prevention.
Looking Beyond the Autopsy Table
For decades, doctors and pathologists have searched for reliable signs that distinguish SIDS from other causes of infant death. Classic autopsies rarely reveal a clear culprit, and most proposed risk factors, such as sleeping position or mild infections, are too general to pinpoint which infants are truly vulnerable. The prevailing “triple risk” idea suggests that SIDS occurs when a vulnerable baby, during a sensitive developmental window, encounters an external stressor—such as overheating or smoke exposure—that the body fails to handle. Yet there have been few concrete biological markers to support this model or to reveal what goes wrong inside the body just before death.
Tiny Packets Carry Messages from Cells
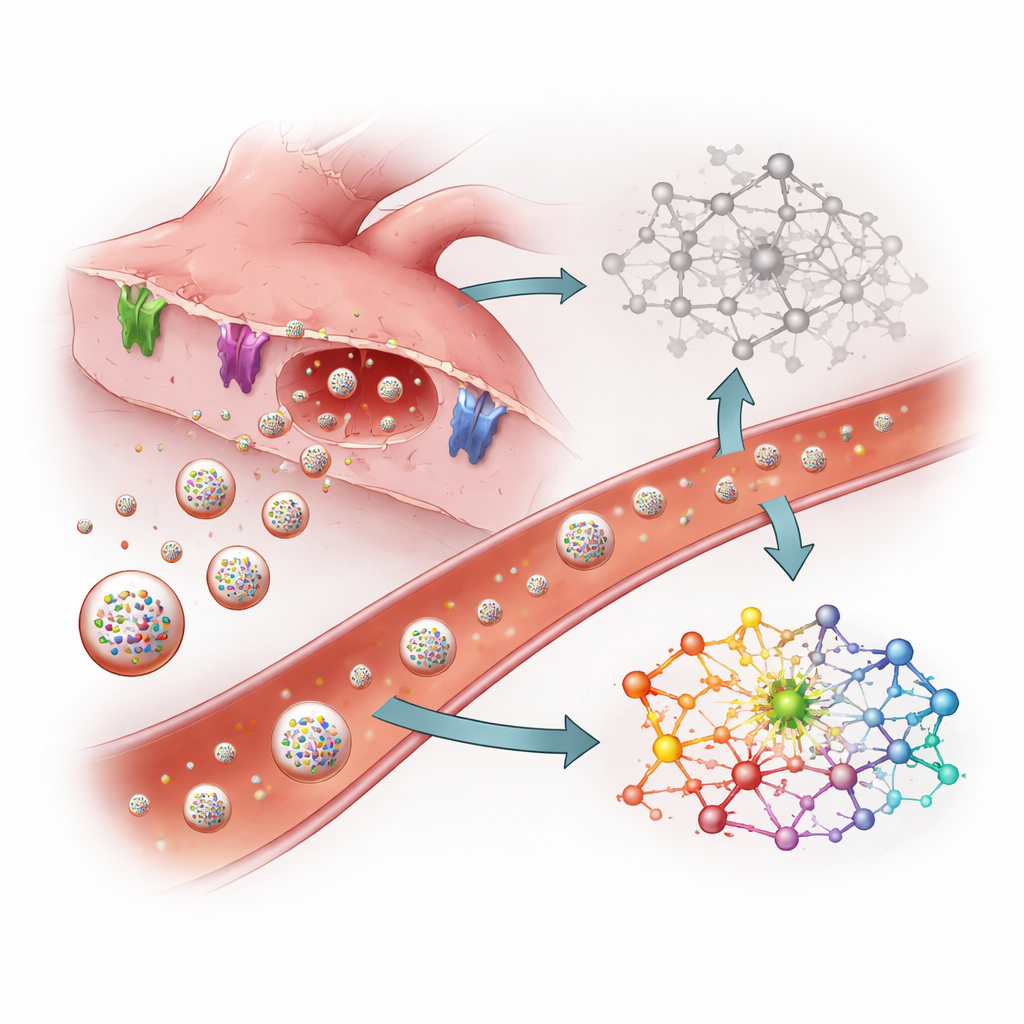
The researchers turned to extracellular vesicles—nano-sized bubbles released by most cells into blood and other body fluids. These vesicles act like sealed envelopes, carrying cargo such as small regulatory RNAs called microRNAs. Because vesicles have a protective membrane, their contents can remain surprisingly stable even after death. The team reasoned that if SIDS involves hidden damage to the heart, heart cells might release a distinctive pattern of microRNAs into vesicles that could still be detected in blood collected at autopsy.
Comparing Lost Infants, One Molecule at a Time
The study analyzed serum (the liquid part of blood) from eight infants who had died and undergone autopsy: four from SIDS and four from clearly explained causes such as dehydration or airway blockage. The scientists first isolated vesicles from each serum sample and confirmed their size and structure. They then used next-generation sequencing to catalogue which microRNAs were present and in what amounts. Comparing SIDS and non-SIDS cases, they found 15 microRNAs that were more abundant and 38 that were less abundant in the SIDS group, with many changes large enough to suggest a meaningful biological shift rather than random noise.
Signals Pointing to Heart Stress
Among the microRNAs elevated in SIDS were several known to be heavily concentrated in heart muscle, including miR-1, miR-208, and miR-499. These molecules are already recognized as early blood markers of heart attacks and other cardiac injuries in adults; they are typically released when heart muscle cells are stressed or damaged. Their increase in vesicles from SIDS cases suggests that, even if the heart looks normal under a microscope, it may have suffered subtle injury or rhythm disturbance before death. Computational analyses of the genes controlled by these microRNAs indicated that key pathways involved in heart muscle contraction, calcium handling, and electrical signaling were affected. Many genes that help move calcium in and out of cells, maintain ion channels, or transmit mechanical signals were predicted to be dialed down.
How Disrupted Cell Machinery Can Stop a Tiny Heart
The gene and pathway patterns painted a picture of a heart whose internal machinery is mis-tuned. Proteins that form the scaffolding and motors of heart muscle, as well as those that regulate how calcium ions trigger each heartbeat, appeared to be compromised in SIDS cases. Important signaling routes, including MAP kinase and related pathways that help heart cells sense and respond to mechanical load and stress, also seemed suppressed. Together, these shifts could make an infant’s heart more vulnerable to dangerous rhythm disturbances or an inability to respond to stressors such as overheating, mild infection, or low oxygen—consistent with the triple risk model.
What This Could Mean for Families
In simple terms, the study suggests that some SIDS deaths may involve hidden heart injury or malfunction that leaves few visible traces at autopsy but leaves a molecular fingerprint in the blood. Heart-related microRNAs carried in extracellular vesicles rose sharply in SIDS cases, while many of their target genes tied to heart rhythm and contraction appeared to be dampened. Although the work is based on a small number of cases and cannot yet be used as a clinical test, it highlights a promising path: minimally invasive analysis of vesicle-borne microRNAs could one day help distinguish SIDS from other causes of infant death and provide deeper insight into how to identify and protect at-risk babies.
Citation: Kanno, S., Fukuta, M., Kato, H. et al. Comparative analysis of microRNA expression in serum-derived extracellular vesicles from sudden infant death syndrome cases. Sci Rep 16, 8168 (2026). https://doi.org/10.1038/s41598-026-38034-4
Keywords: sudden infant death syndrome, microRNA, extracellular vesicles, cardiac injury, biomarkers