Clear Sky Science · en
A tool for predicting pH and temperature effects on porcine and human pepsin activity during in vitro gastric digestion
Why Stomach Enzymes Matter to Everyday Eating
Every time we eat, powerful enzymes in our stomach begin breaking food proteins into smaller pieces our bodies can use. One of the most important of these enzymes is pepsin. Food scientists frequently use a pig version of pepsin in lab tests to stand in for the human form, assuming they behave much the same. This study asks a simple but crucial question: under the changing conditions inside a real stomach—where both acidity (pH) and temperature rise and fall—do human and pig pepsin really act alike, and how can we predict what they will do?
How the Stomach’s Changing Conditions Shape Digestion
Inside the stomach, pH and temperature are far from constant. A cold drink can temporarily cool the stomach by more than 10 degrees, and a large or alkaline meal can raise stomach pH several units before it slowly returns to a more acidic state. Because pepsin’s ability to cut up proteins depends strongly on both acidity and warmth, these swings can speed up or slow down digestion. Directly tracking this inside people is hard, so researchers rely on in vitro models—carefully controlled glassware versions of digestion. Those models usually use porcine (pig) pepsin as a stand-in for human pepsin, but until now scientists lacked a clear map of how both enzymes respond when pH and temperature shift together, as they do after a meal.
Measuring Pepsin Performance Across Many Conditions
The authors tested human pepsin taken from gastric fluid and commercial porcine pepsin across 37 combinations of pH (from very acidic to nearly neutral) and temperature (from refrigerator-cold to quite hot). They then used these measurements to build mathematical models that can predict how active each enzyme will be under any stomach-like combination of acidity and warmth. Both enzymes worked best near pH 2 and body temperature, but the human version kept working over a broader range. At pH 3 and 4, human pepsin still retained substantial activity, while pig pepsin’s activity dropped off sharply. Interestingly, even though porcine pepsin was more powerful at its sweet spot, it proved much more sensitive to pH changes, meaning its performance can fall off faster as conditions become less ideal.
What the New Prediction Tool Can Tell Us
By turning these measurements into user-friendly equations embedded in spreadsheet tools, the researchers created a way for others to estimate pepsin activity during laboratory digestion experiments. They ran several case studies to show what the tool can do. In a semi-dynamic model of milk digestion, simulated human and porcine pepsin showed similar overall patterns, but human pepsin maintained useful activity for longer as pH shifted, leading to a larger total “area under the curve” for activity over time. The tool also compared traditional static digestion setups—where pH is held constant—to semi-dynamic ones that gradually acidify, and simulated how pepsin activity might differ in younger versus older adults, who typically secrete less pepsin. These examples highlight how the same meal and protocol can produce different levels of protein breakdown depending on which enzyme type and digestion model is used.
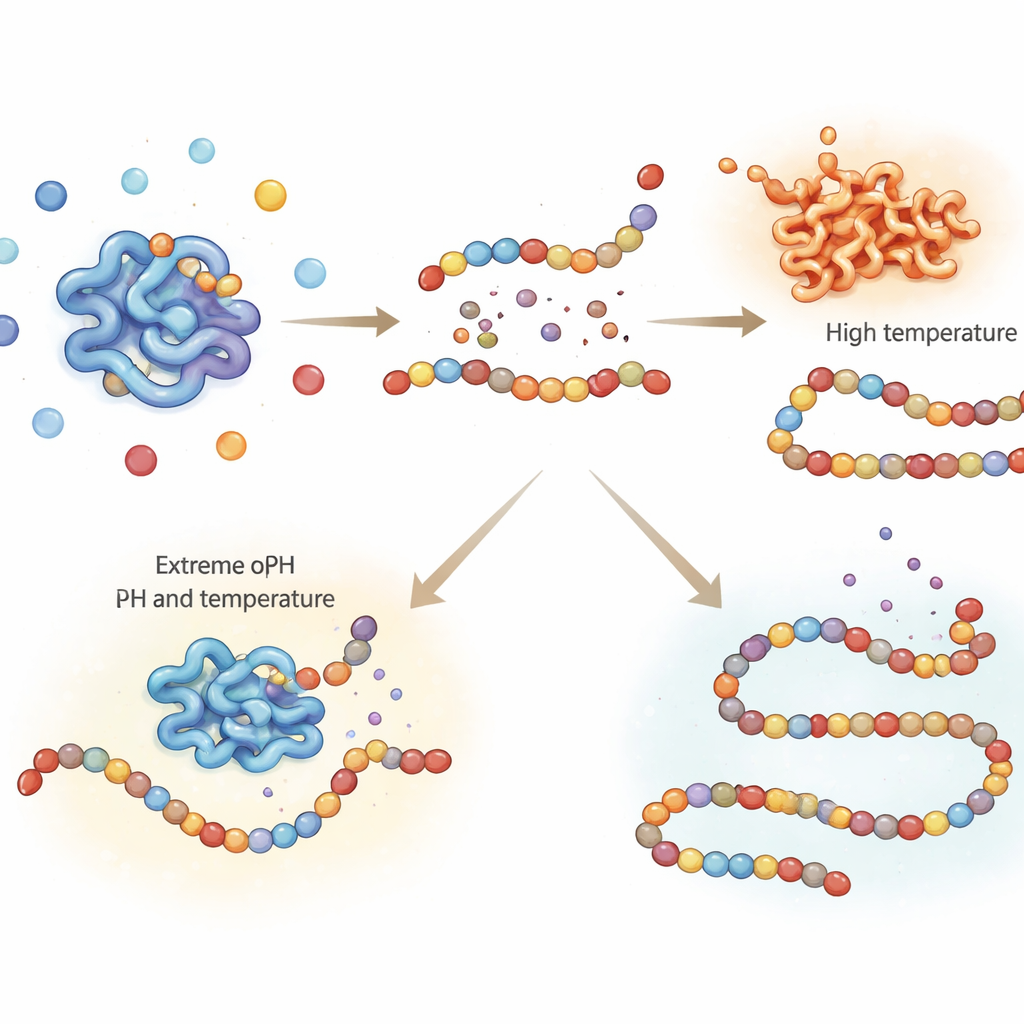
Finding the Gentle Point Where Heat Turns Pepsin Off
In digestion experiments, it is essential to stop pepsin at specific time points so later measurements reflect what happened in the “stomach,” not continued breakdown in the test tube. A common way to switch off enzymes is heating, but too much heat can damage other food components and skew results. This study carefully tested how long and how hot porcine pepsin must be heated to be permanently inactivated. Heating at 65 °C for up to 15 minutes only partially reduced its activity, meaning the enzyme could still keep working afterwards. In contrast, heating at 75 °C or higher for just 5 minutes fully and irreversibly shut down pepsin. Infrared spectroscopy of the enzyme’s structure showed that these higher temperatures caused lasting unfolding and aggregation of the protein, matching the loss of activity.
What This Means for Food Research and Health
For non-specialists, the key message is that the pig pepsin commonly used in lab models does not behave exactly like human pepsin once real-world stomach conditions—shifting acidity and temperature—are taken into account. The new prediction tool helps researchers estimate how much protein-cutting power is present at each moment of a digestion experiment and to judge whether their setup truly reflects what might happen in a human stomach, including that of older adults. At the same time, the work pinpoints a gentle but reliable heat treatment—five minutes at 75 °C—to switch off pepsin without unnecessary damage to other food components. Together, these advances should make lab-based digestion studies more accurate and easier to compare, improving our understanding of how different foods and processing methods affect protein digestion in everyday life.
Citation: C. J., F., D., F., I., C. et al. A tool for predicting pH and temperature effects on porcine and human pepsin activity during in vitro gastric digestion. Sci Rep 16, 9176 (2026). https://doi.org/10.1038/s41598-026-38033-5
Keywords: pepsin, gastric digestion, pH and temperature, in vitro models, protein digestion