Clear Sky Science · en
Efficient removal of Cs+ and Sr2+ from water using titanate nanotubes embedded in alginate macromolecules
Why cleaning up radioactive water matters
After nuclear accidents, medical procedures, or routine power plant operations, tiny amounts of long-lived radioactive metals can end up in water. Two of the most worrying are cesium and strontium, which can build up in soft tissues and bones if they enter the food chain. This study explores a promising way to trap these risky metals quickly and efficiently using tailor‑made mineral nanostructures wrapped in a biodegradable, jelly‑like polymer.
Tiny tubes that act like metal sponges
The researchers focused on titanate nanotubes—hollow, needle‑like particles made from a titanium‑based mineral. Because these tubes are extremely small and have a very large surface area, they offer many places for metal ions to stick. The team made the nanotubes in a high‑temperature alkaline solution starting from common titanium dioxide powder. Tests showed that the resulting tubes were stable, uniform in size, and covered with chemical groups that can grab positively charged ions such as cesium (Cs⁺) and strontium (Sr²⁺).
From loose powder to easy‑to‑handle beads
Although bare nanotubes are excellent at capturing metals, they are hard to collect from treated water because they are so fine. To solve this, the scientists embedded the nanotubes inside beads made from alginate, a natural polymer extracted from brown seaweed and already used in foods and medical products. When alginate meets calcium ions in water, it turns into firm gel beads. By mixing the nanotubes with alginate before this step, the team formed a composite material (called T/G) in which the nanotubes are trapped inside millimeter‑scale spheres that can be easily scooped out or packed into filters.
How well the new materials clean water
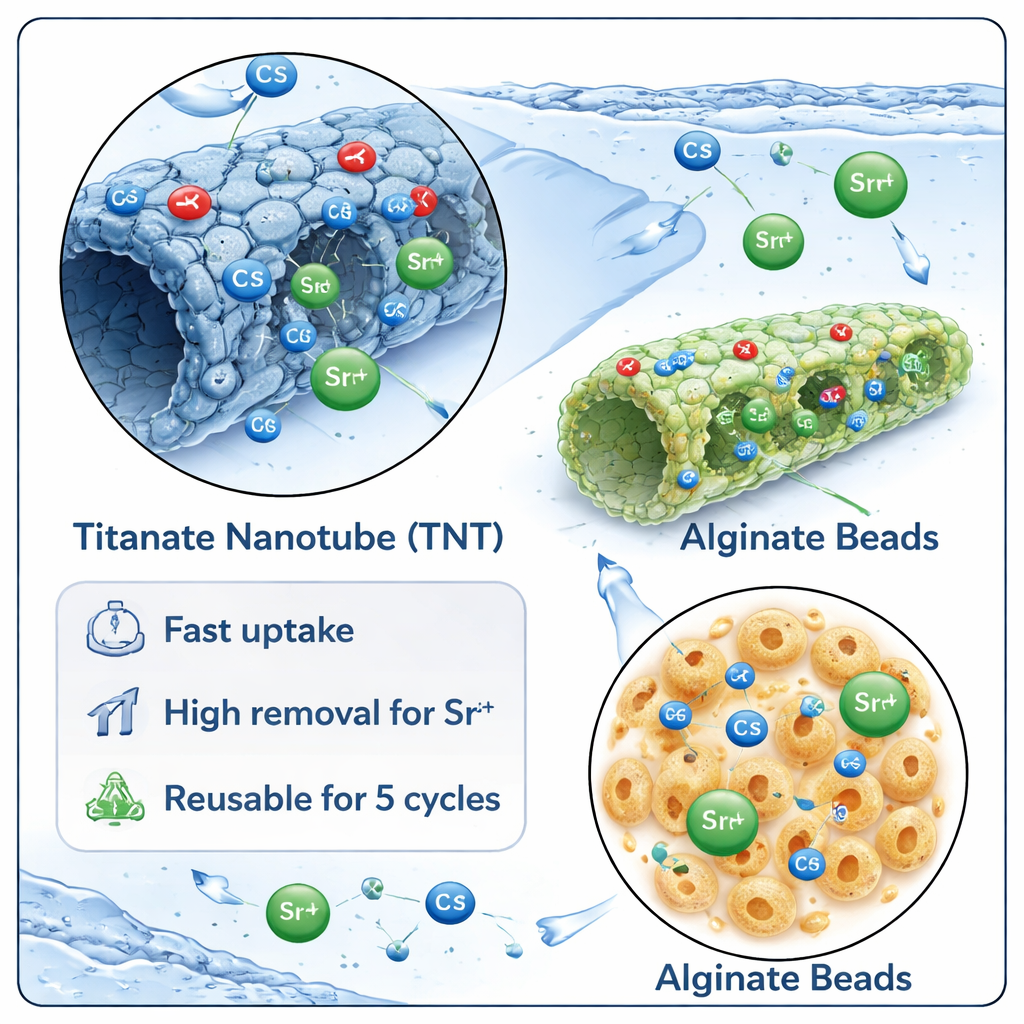
In laboratory tests, the nanotube powder removed cesium and strontium from water very rapidly, reaching near‑maximum uptake in as little as 15–30 minutes. Under mildly alkaline conditions (around pH 8) and at a modest dosage, the nanotubes took out about 90% of cesium and 97% of strontium from dilute solutions. Detailed modeling of how much metal the material could hold showed that the tubes provide a mix of surface types, allowing several layers of ions to build up, especially for strontium. When the nanotubes were locked inside alginate beads, overall removal dropped to roughly 45–70% for cesium and 70–90% for strontium, mainly because each bead contains less active nanotube surface than the loose powder. However, the beads became much easier to handle and separate from the water.
What happens at the atomic scale
Measurements of the materials before and after use revealed a multi‑step trapping process. First, the surfaces of the nanotubes carry negatively polarized oxygen groups that attract the positively charged cesium and strontium ions. Next, these ions form stronger bonds with oxygen‑rich sites on the surface, creating stable complexes. Finally, some of the incoming cesium and strontium push out sodium ions that are naturally present inside the titanate structure, effectively swapping places. This combination of electrostatic attraction, surface bonding, and ion exchange explains both the fast action and the high capacity of the nanotubes, especially for strontium.
Reusing the filters and looking ahead
A key question for any cleanup technology is whether it can be used more than once. The team showed that both the pure nanotubes and the alginate‑based beads could be regenerated by washing them with a mild acid to release the trapped metals, then rinsing and reconditioning them. After five such cycles, the nanotubes still kept over 90% of their original performance, and the beads retained more than 85%, while remaining structurally intact. For real‑world use, the authors note that the ratio of nanotubes to alginate in the beads must be optimized, and the materials need to be tested in genuine wastewaters that contain many competing ions. Still, the work suggests that titanate nanotubes, especially when combined with simple biopolymer beads, are strong candidates for scalable, reusable systems to clean radioactive cesium and strontium from water.
Citation: Farouk, E., Zaki, A.H., Eldek, S.I. et al. Efficient removal of Cs+ and Sr2+ from water using titanate nanotubes embedded in alginate macromolecules. Sci Rep 16, 7483 (2026). https://doi.org/10.1038/s41598-026-38030-8
Keywords: radioactive water treatment, cesium removal, strontium removal, titanate nanotubes, alginate beads