Clear Sky Science · en
Identification of ubiquitination-related signature genes for predicting kidney transplant rejection
Why early warning for new kidneys matters
For people with kidney failure, a transplanted kidney can be life-changing—but only if the body accepts it. Even with modern anti-rejection drugs, the immune system can quietly damage a new kidney before standard tests notice anything is wrong. This study explores a new way to spot rejection earlier and more precisely by reading patterns of gene activity linked to a basic cell process called protein tagging, potentially helping doctors protect transplanted kidneys for longer.
A hidden tug-of-war inside the transplanted kidney
After a kidney transplant, the recipient’s immune system constantly scans the donor organ, trying to decide whether to tolerate it or attack it. Doctors typically classify rejection into two main types: one driven mostly by immune cells called T cells, and another driven by antibodies circulating in the blood. In reality, many patients show a confusing mix of both, making diagnosis and treatment decisions difficult. Current tools—blood tests like creatinine or needle biopsies—either pick up damage late or are too invasive for frequent use, leaving a gap in early, reliable detection.
The cell’s quality-control tags as an early signal
Cells maintain order by attaching tiny molecular “tags” to proteins, marking them for recycling or changing how they behave. This tagging system, called ubiquitination, also helps control immune responses and inflammation. Because it sits high up in the control chain, disturbances in this system may appear early when the immune system begins to attack a transplanted kidney. The researchers reasoned that measuring the activity of genes involved in this tagging process could provide a sensitive readout of impending rejection, even before obvious tissue damage appears under the microscope.
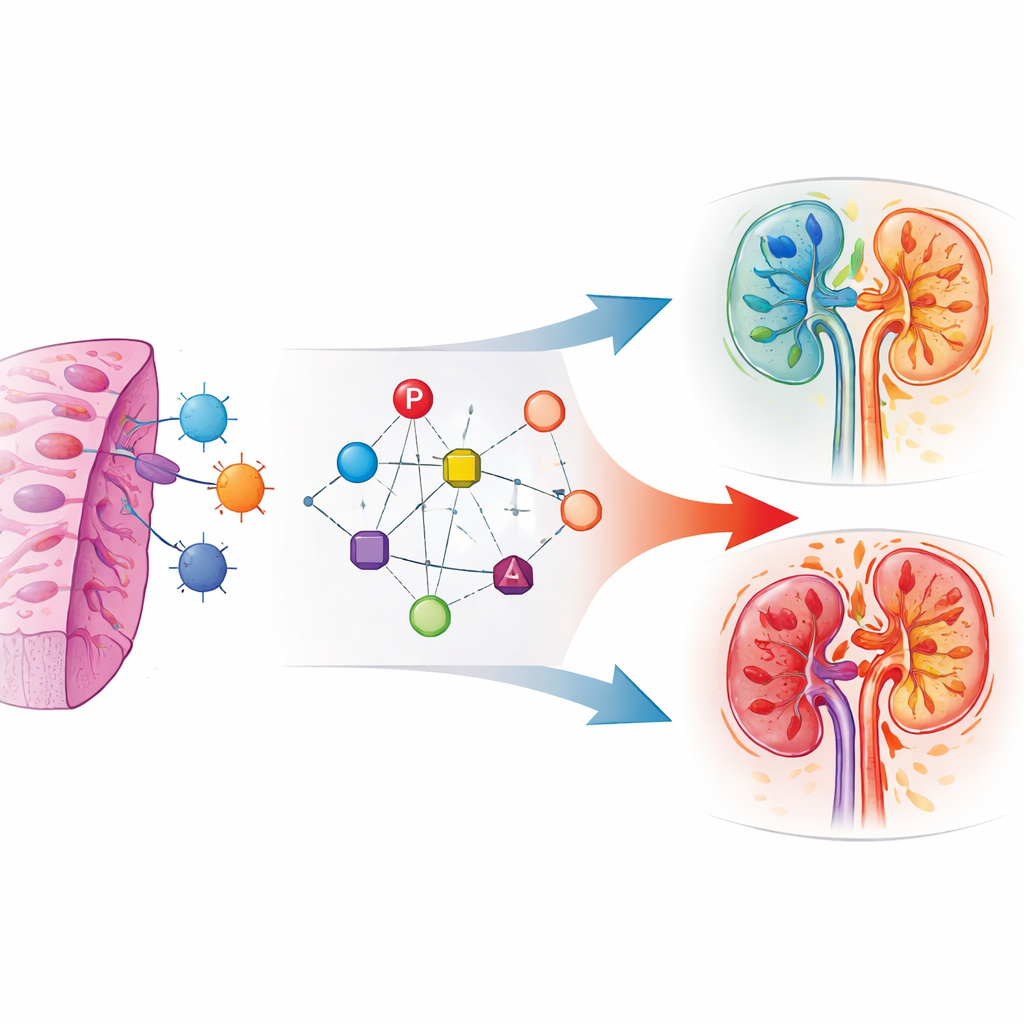
Turning gene activity into a risk score
The team analyzed thousands of biopsy samples from kidney transplant recipients stored in public gene-expression databases. They first looked for genes that behaved differently in rejection versus non-rejection biopsies, then focused on those tied to the protein-tagging machinery. Six genes showed the strongest and most consistent differences, especially in samples with clear rejection. The scientists combined the activity of these genes into a single “score” that could be calculated for each biopsy. Higher scores tended to appear in patients with rejection, and the score performed well at separating rejection from stable cases in multiple independent groups of patients.
Linking gene patterns to specific types of immune attack
When the researchers compared different rejection types, they found a striking pattern: levels of the six key genes rose stepwise from no rejection to antibody-driven rejection, were higher still in T-cell–driven rejection, and peaked when both mechanisms were present together. This suggests the gene pattern acts like a dial for the overall intensity and complexity of the immune attack, especially when T cells are heavily involved. The score also tracked with shifts in immune cell populations inside the kidney, including regulatory T cells and other specialized cells that can both damage and protect the organ, reflecting a dynamic struggle between injury and repair.
From lab model to potential bedside tool
To move toward clinical use, the authors built a simple chart that translates the six-gene pattern from a biopsy into an estimated risk of rejection. They then cross-checked the genes in a small, real-world group of transplant patients using a standard lab method. In the two patients with mixed rejection, two of the genes stood out as particularly high, matching the patterns seen in the larger datasets. One patient with a different kidney disease but no formal rejection also showed high levels of all six genes, hinting that this signature may be a sensitive alarm for strong immune activity in the kidney, not just classic rejection. This means the tool could help flag patients who need closer monitoring, while still requiring doctors to interpret results in the context of other clinical information.
What this could mean for transplant patients
In everyday terms, this study suggests that reading a small set of “protein-tag” genes in a kidney biopsy can reveal how fiercely the immune system is attacking the graft, often before standard tests make the problem obvious. While larger, prospective studies are still needed and the method does not replace a doctor’s judgment, such a gene-based score could one day help tailor drug doses, time follow-up biopsies more wisely, and catch trouble early enough to save more transplanted kidneys.
Citation: Shan, Z., Yu, S., Wang, J. et al. Identification of ubiquitination-related signature genes for predicting kidney transplant rejection. Sci Rep 16, 8102 (2026). https://doi.org/10.1038/s41598-026-38022-8
Keywords: kidney transplant rejection, gene expression, ubiquitination, biomarkers, immune response