Clear Sky Science · en
Modeling and experimental analyses for Chitosan/Zinc oxide nanocomposite
Why this matters for everyday life
From food packaging that keeps fruit fresh longer to coatings that kill germs and clean up pollution, materials built from natural polymers and tiny inorganic particles are quietly moving into daily products. This study looks at a promising pair: chitosan, a sugar-based material derived from shellfish waste, and zinc oxide, a light-sensitive mineral used in sunscreens and electronics. By understanding, in detail, how these two ingredients interact at the atomic level, the researchers show how to steer their electronic and light-absorbing behavior—knowledge that could help design safer, greener materials for sensors, packaging, and environmental cleanup.
A natural helper meets a tiny mineral
Chitosan is attractive because it is nontoxic, biodegradable, and gentle to living tissue, but it dissolves poorly in water and has limited electronic activity. Zinc oxide, in contrast, is a semiconductor widely used in LEDs, solar cells, and UV detectors, and it also fights bacteria and fungi. When zinc oxide nanoparticles are mixed into thin chitosan films, previous work showed that the films become stronger, better at blocking gases and light, and more effective at stopping microbes and degrading dyes in water. The open question was how, exactly, the zinc oxide particles bind to chitosan’s chemical groups and how those bonds change the way the material handles charge and light.
Peering into the bonds with virtual experiments
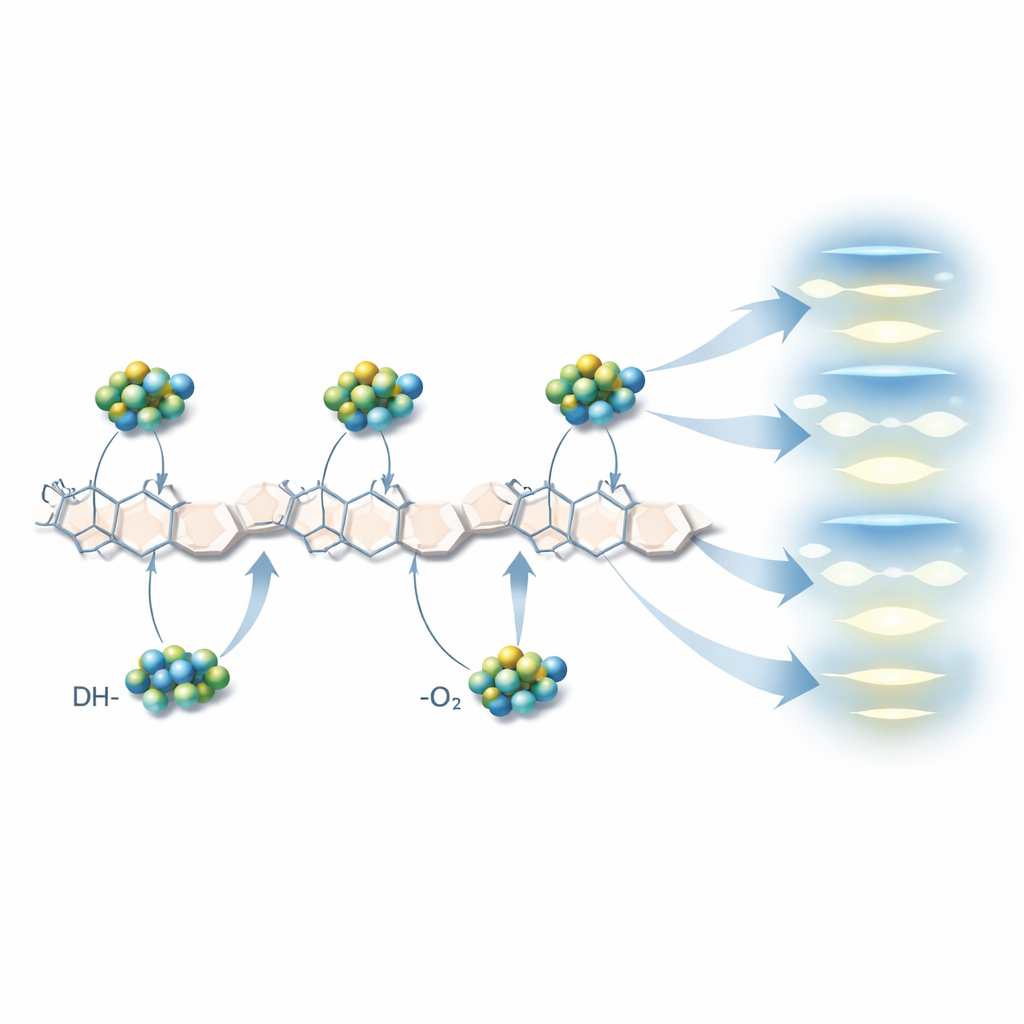
To answer this, the team used quantum-chemical calculations to build simplified models of short chitosan chains interacting with one or two zinc oxide units. They explored three main ways the partners can connect: through the nitrogen-containing amine group, through oxygen atoms that link sugar rings, and through oxygen in alcohol-like groups. These virtual experiments revealed that adding zinc oxide sharply increases the overall polarity of the molecule—the separation of positive and negative charge—and shrinks the gap between its most stable filled and empty electronic states. In some two-zinc configurations, this gap drops to less than half the value found in pure chitosan, signaling a material that can more easily move or reorganize electrons when stimulated by light or electric fields.
How the charge rearranges inside
Further analysis showed that electrons tend to flow from chitosan toward the zinc centers, especially when bonding occurs through bridging oxygen atoms. Maps of electrostatic potential and bond topology indicated that zinc forms partly covalent links with oxygen and, in some cases, nitrogen in the chitosan chain, reinforced by hydrogen bonds. These mixed interactions create a stable interface where charge is unevenly distributed, which explains the increased dipole moment and the material’s greater tendency to accept electrons. In other words, the hybrid becomes “softer” and more reactive than the natural polymer alone, a desirable trait for applications like photocatalysis and sensing, where charge transfer drives performance.
Matching computer predictions with real measurements
The researchers then made actual chitosan films containing different amounts of zinc oxide nanoparticles and probed them with infrared light and ultraviolet–visible diffuse reflectance. In the infrared spectra, the signature bending motion of certain nitrogen–hydrogen bonds in chitosan systematically shifted to lower frequency as more zinc oxide was added, and new bands associated with zinc–oxygen vibrations appeared and grew. These changes match the calculated picture of stronger bonding at specific sites. Optical measurements showed that the energy needed to promote electrons with light—the optical bandgap—decreased as zinc oxide content rose. Both “direct” and “indirect” bandgaps moved to lower energies, and the absorption edge became more smeared out, consistent with new defect states and band “tails” created by the embedded nanoparticles.
What this means for future devices and products
Taken together, the modeling and experiments paint a coherent story: when zinc oxide nanoparticles latch onto chitosan at the right chemical sites, they reshape the electronic landscape of the biopolymer, making it more polar, more accepting of electrons, and more responsive to light. By tuning how many particles are added and how they bind, one can adjust the material’s bandgap and therefore the colors of light it absorbs and how it conducts charge. For lay applications, this means that a single biodegradable film could be engineered to act as an active food wrapper, a light-driven pollutant remover, or part of a low-cost optical sensor—simply by tweaking its microscopic chemistry rather than replacing its ingredients.
Citation: Elhaes, H., Amin, K.S., El Desouky, F.G. et al. Modeling and experimental analyses for Chitosan/Zinc oxide nanocomposite. Sci Rep 16, 8942 (2026). https://doi.org/10.1038/s41598-026-38013-9
Keywords: chitosan zinc oxide nanocomposite, biodegradable functional materials, tunable optical bandgap, photocatalytic food packaging, bioinspired sensors