Clear Sky Science · en
Chronic NH4Cl loading improves glucose tolerance without modifying insulin sensitivity in mice
Why acid–base balance matters for blood sugar
People with chronic kidney disease often develop a condition called metabolic acidosis, where the blood becomes slightly more acidic than normal. Doctors usually view this as harmful, in part because short bursts of acidosis are known to worsen blood sugar control. This study turns that idea on its head: in mice, a long-lasting, mild acidosis actually improved how the body handled sugar, without making the hormone insulin work any better or worse. Understanding this surprising twist could eventually help refine treatments for both kidney disease and type 2 diabetes.
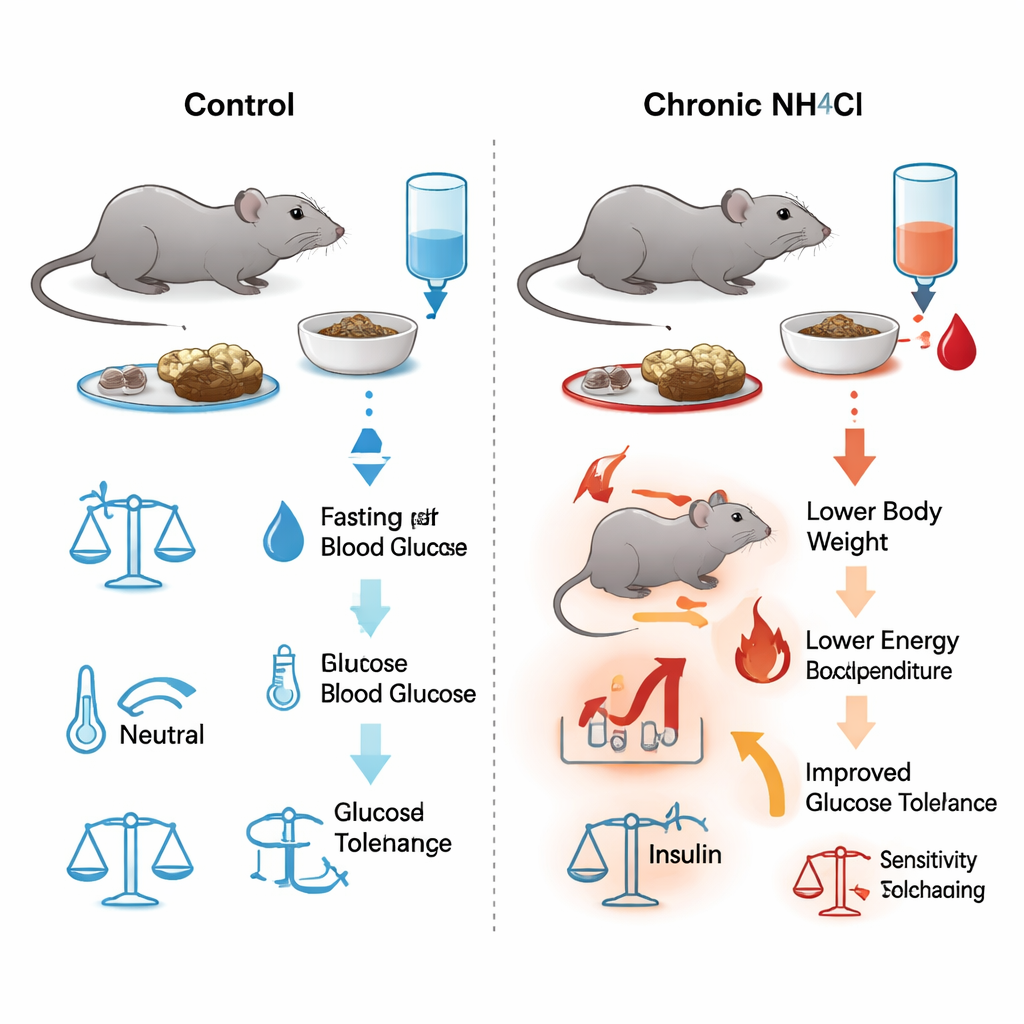
A long-term acid challenge in mice
The researchers gave male mice drinking water containing ammonium chloride, a salt that gently acidifies the blood, for up to six months. This treatment mimics the chronic acid load seen in kidney disease. Blood tests confirmed a persistent, mild metabolic acidosis: lower pH and bicarbonate, higher chloride, and otherwise stable kidney-related measures such as urea and hematocrit. Despite eating and drinking as much or more than untreated mice, the acid-loaded animals stopped gaining weight after about two months. Detailed measurements in metabolic cages showed that these mice were burning more energy overall, had less lean (muscle-rich) mass, and carried relatively more fat, pointing to a higher metabolic cost of living under chronic acidosis.
Better blood sugar control without extra insulin help
The team repeatedly tested how well the mice cleared sugar from their bloodstream by injecting glucose and tracking blood levels over time. As early as one week after starting the acidifying drink, the treated mice lowered their blood glucose faster than controls, and this advantage persisted at many time points up to 180 days. Fasting blood sugar was consistently lower in the acidotic group. Yet, when the scientists measured insulin in the blood during these tests, levels were the same in both groups. Separate tests in which mice were given insulin directly showed that their overall sensitivity to insulin was also unchanged. In other words, chronic acidosis improved glucose tolerance without asking the pancreas to produce more insulin or the tissues to respond more strongly to it.
Shifting where and how sugar is made and lost
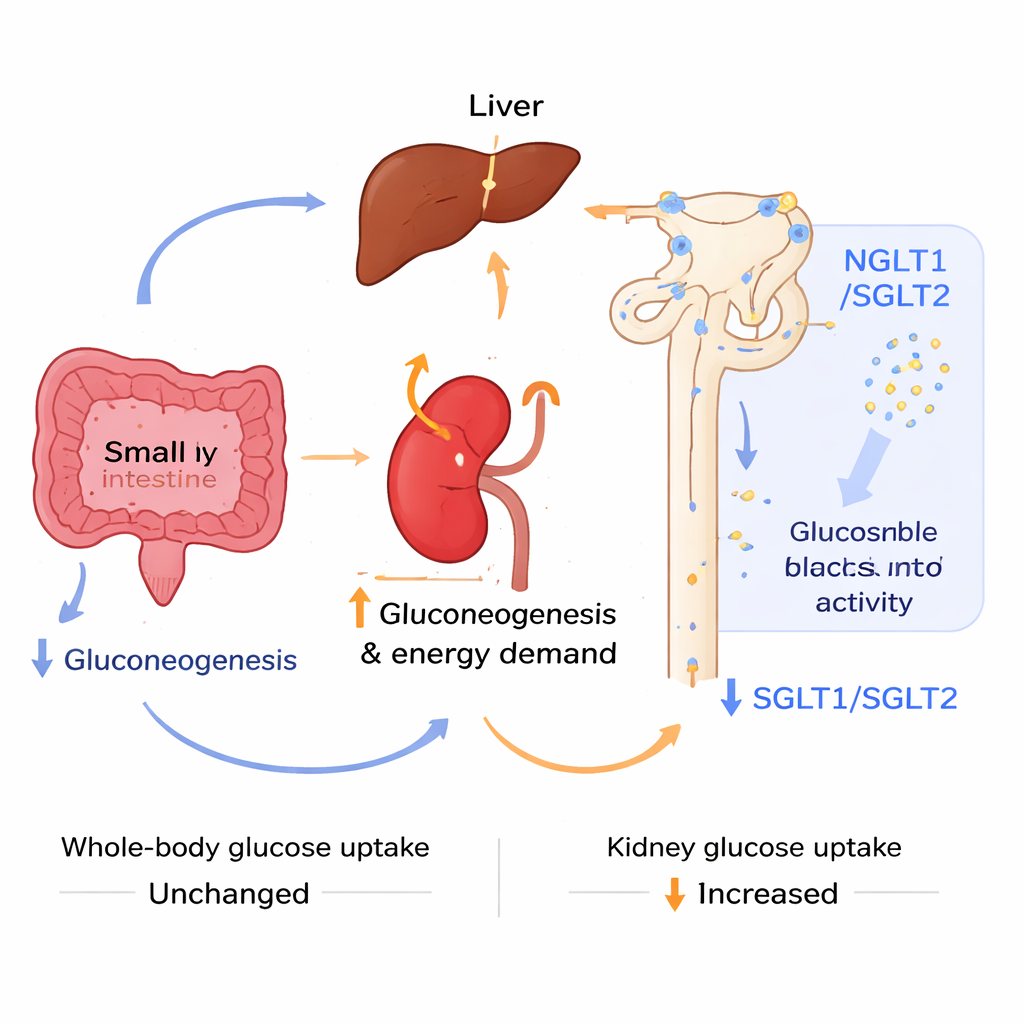
To find out where the extra sugar control came from, the researchers probed how different organs produced glucose. They used specific substances—pyruvate, alanine, and glutamine—that feed into sugar-making pathways in liver, kidney, and intestine. Over time, the liver and parts of the intestine of acidotic mice became less active at making new glucose, especially from alanine and glutamine. By contrast, the kidney showed signs of working harder: key genes involved in glucose production and energy supply were turned up, and imaging with a radioactive glucose mimic revealed higher sugar uptake in the kidneys and bladder, even though total body uptake was unchanged. At the same time, more glucose was lost in the urine after a sugar load, because the kidney’s main sodium–glucose transporters, which normally reclaim filtered sugar, were dialed down at both the RNA and protein level. This combination—less sugar made by liver and gut, more sugar burned and spilled by the kidney—helped keep blood levels in check.
Deep changes inside kidney cells
Gene-activity snapshots of the kidneys over the first two months of treatment showed broad remodeling of cellular machinery. Pathways involved in moving charged particles (anions), reclaiming bicarbonate, and running mitochondria and oxidative phosphorylation—the cell’s main energy generators—were boosted. Genes tied to glucose and fat metabolism also shifted upward, while some immune and detoxification pathways, including parts of the complement system and cytochrome P450 family, were dampened. These patterns fit with a kidney that is working harder to excrete acid, consuming more fuel, and at the same time reprogramming how it handles sugar and lipids to meet the extra energy demand.
What this means for people with kidney and sugar problems
In summary, long-term, mild metabolic acidosis in mice did not harm blood sugar control—in fact, it improved it—by reducing sugar production in the liver and intestine, lowering sugar reabsorption in the kidney, and increasing the kidney’s own energy use. Insulin itself played no bigger role than usual. This does not mean that acidosis is good or that patients should remain acidotic; acidosis still has many harmful effects. But the findings reveal that the body can adapt in unexpected ways, and they highlight the kidney as a powerful regulator of blood sugar. Future work may be able to harness some of these kidney-based mechanisms—such as increased urinary glucose loss and altered glucose production—without exposing patients to the risks of chronic acidosis.
Citation: Zaibi, N., Montaigne, J., Baraka-Vidot, J. et al. Chronic NH4Cl loading improves glucose tolerance without modifying insulin sensitivity in mice. Sci Rep 16, 7048 (2026). https://doi.org/10.1038/s41598-026-38007-7
Keywords: metabolic acidosis, glucose tolerance, kidney function, gluconeogenesis, type 2 diabetes risk