Clear Sky Science · en
Chronic early life stress alters the microbial and transcriptional profile of the zebrafish gut
Why early stress in tiny fish matters to us
Stressful experiences in the first weeks of life can leave health scars that last for decades, but how this happens inside the body is still being uncovered. In this study, researchers turned to zebrafish—small, transparent fish that are powerful models for human biology—to ask what chronic early life stress does to the gut, its resident microbes, and future fertility. By following stressed fish across several generations, they show that early stress can ripple forward in time, reshaping the gut ecosystem, gene activity, and even the chances that eggs and offspring survive.
A multigenerational stress experiment
To probe these long-term effects, the team exposed young zebrafish to repeated mild challenges twice a day from about one week after hatching until they were a month old. The stressors mimicked an unpredictable environment: brief bursts of strobe light, water turbulence, or gentle chasing with a net, delivered at random times so the fish could not adapt. This routine was applied not just to one group, but across four successive generations, creating lineages with and without a family history of early stress. In adulthood, the scientists measured how many eggs these fish produced and how many offspring survived, and they collected gut samples to examine both the microbial communities and the activity of thousands of genes. 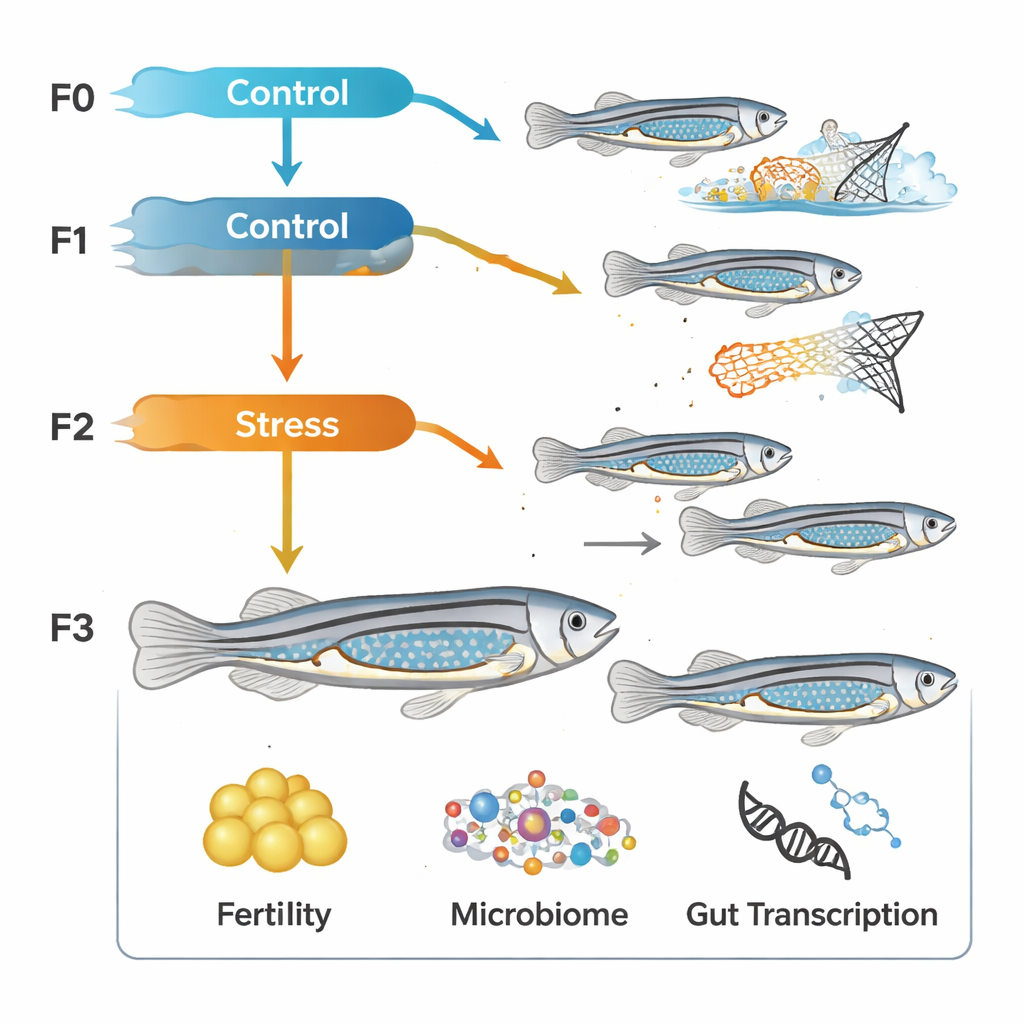
Fewer healthy eggs and lower survival
The first striking outcome was reproductive. Adult fish that had been stressed early in life produced noticeably more dead or abnormal eggs than their unstressed siblings. Even when the researchers started with equal numbers of healthy-looking embryos, fewer stressed-lineage offspring survived the transition from embryo to larva, and many did not make it to adulthood. By the time the surviving fish reached 16 months of age, overall survival in the stressed group was roughly one third that of controls, and the population was skewed toward females. These patterns suggest that early stress can quietly undermine fertility and survival, with effects that emerge long after the stressful period has ended.
Gut microbes shift toward opportunists
Because the gut microbiome is known to influence both brain and reproductive health, the team next asked how stress reshaped the bacterial communities in the zebrafish gut. Using DNA sequencing to profile microbes from pooled gut contents, they found that fish exposed to early stress—either in their own lives or across generations—carried very different microbial communities from unstressed controls. While overall diversity within each gut changed only modestly, the mix of species shifted dramatically. Control fish tended to harbor more Pseudomonas bacteria, whereas stressed and stress-lineage fish showed expansions of Vibrio, Aeromonas, and Shewanella. These microbes are common in water and can live harmlessly in fish, but they are also known as “opportunistic pathogens” that take advantage when defenses are weakened. The distinct microbial signatures seen in stressed lineages suggest that early adversity can tilt the gut ecosystem toward a more infection-prone state that partly persists across generations.
Stress-primed gut genes and immune defenses
To understand how the host tissue itself responded, the researchers examined which genes were turned on or off in the guts of young fish that had experienced early stress. By sequencing RNA from whole guts, they identified more than 800 genes whose activity levels changed. Many of the genes turned up by stress were involved in immune defenses, especially pathways triggered by interferons—molecular alarms that help cells fight viruses and other invaders. In contrast, several genes linked to fat processing, T cell signaling, and nerve cell development were dialed down. Network analyses highlighted families of antiviral and antimicrobial genes, suggesting that the gut of stressed fish is in a heightened defensive state, perhaps reacting to the altered microbial community or directly to stress hormones. 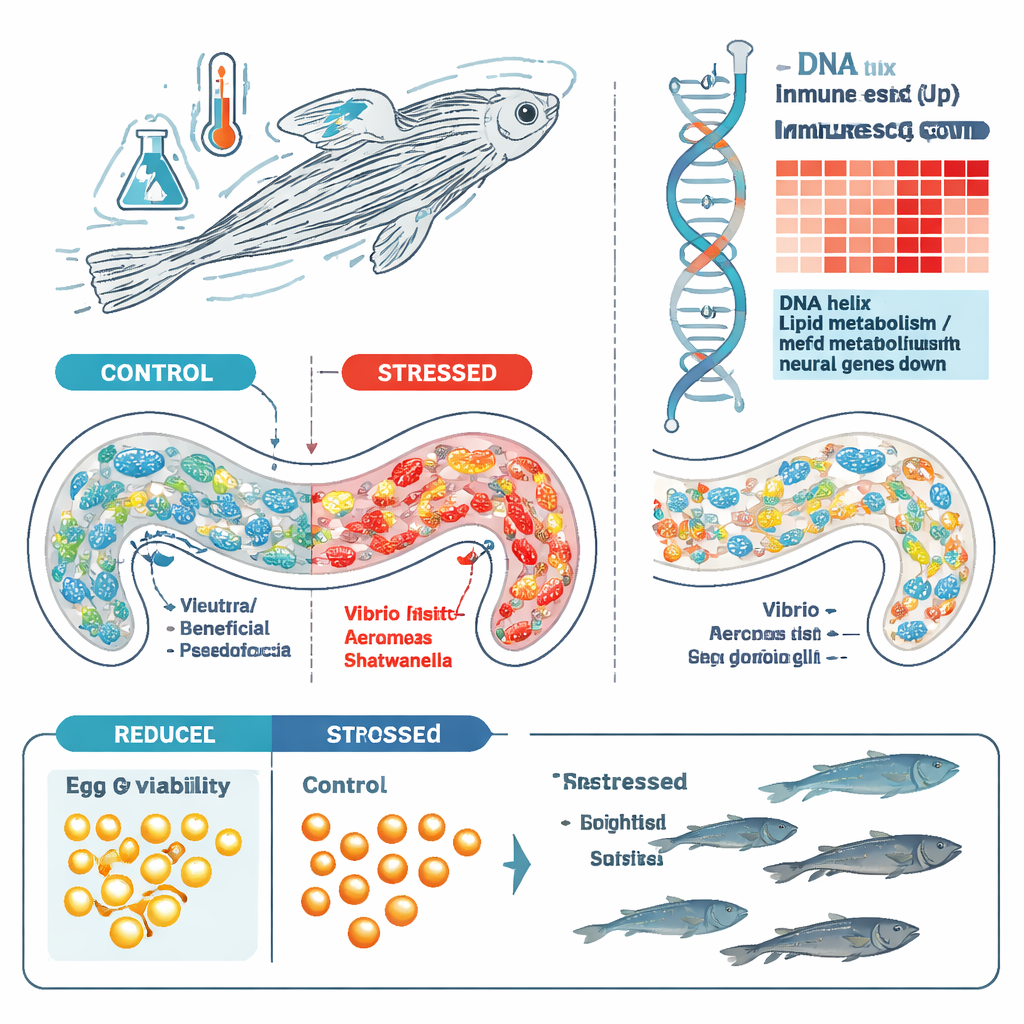
What this means for health and future research
Together, these findings paint a picture of early life stress as a body-wide programmer, capable of reducing egg quality and survival while nudging gut microbes toward riskier species and rewiring gut gene activity toward chronic alert. Although this work was done in zebrafish, the basic biology of stress hormones, immune defenses, and gut microbes is shared across vertebrates, including humans. The study strengthens the idea that the gut–brain–gonadal axis—linking mental state, digestion, microbes, and reproduction—is a key route by which childhood adversity can shape lifelong health. It also positions zebrafish as a powerful system for future experiments that can move beyond correlation to test exactly how early stress, microbes, and immune pathways interact to influence fertility, resilience, and disease risk.
Citation: Norloff, E., Coker, K., Tusneem, S. et al. Chronic early life stress alters the microbial and transcriptional profile of the zebrafish gut. Sci Rep 16, 6949 (2026). https://doi.org/10.1038/s41598-026-37978-x
Keywords: early life stress, zebrafish, gut microbiome, fertility, mucosal immunity