Clear Sky Science · en
Advancing ZnO nanostructures through strategic transition metal doping
Powering gadgets from the world around us
From fitness trackers to wireless sensors, modern electronics increasingly rely on tiny devices that can run for long periods without bulky batteries. One promising route is to harvest small amounts of energy from light, heat, or even the bend of a joint. This study explores how a common material, zinc oxide (ZnO), can be carefully modified at the atomic level to become a more efficient building block for such self-powered technologies, potentially improving solar cells, wearable generators, and sensitive detectors.
Why tinker with a familiar material?
ZnO is already a workhorse in electronics: it is transparent, inexpensive, chemically robust, and works well in nanowire form for sensing and energy harvesting. Yet in its pure state it has two big drawbacks. It does not conduct electricity particularly well, and it mainly responds to ultraviolet light, ignoring much of the visible spectrum. The authors set out to see how adding small amounts of two different metals—yttrium (Y) and vanadium (V)—into the ZnO crystal could overcome these weaknesses, while keeping the structure stable enough for real devices.
Designing atomic-level substitutions
Instead of running many trial‑and‑error lab experiments, the researchers used powerful computer simulations based on quantum mechanics (density functional theory). They built virtual ZnO crystals and then replaced some zinc atoms with Y or V at two concentration levels. These models allowed them to calculate how the atoms rearrange, how strongly the lattice resists deformation, and how easily electrons can move. The work also included simulated X‑ray diffraction patterns—essentially virtual fingerprints—to check that the doped crystals keep the same overall structure as pure ZnO. 
Shaping how electrons flow and light is absorbed
At the heart of the study is how doping reshapes ZnO’s electronic bands—the energy levels electrons are allowed to occupy. For pure ZnO, there is a clear gap between filled and empty levels, which limits conductivity. When Y or V atoms are added, new donor states appear near this gap and push the effective energy threshold upward. In practical terms, more electrons become available to carry current, and the material starts to behave like a highly conductive n‑type semiconductor rather than a poor conductor. The team also examined the “density of states,” which showed strong increases in electronic states near the active energy range, confirming that doping can dramatically boost electrical performance.
From better conductivity to brighter optical response
The same atomic substitutions also re‑shape how ZnO interacts with light. The simulations reveal that Y‑ and V‑doped ZnO absorb more light at lower energies, meaning the material becomes sensitive deeper into the visible spectrum rather than just in the ultraviolet. Measures such as refractive index, reflectivity, optical conductivity, and dielectric response all rise when the right amount of dopant is added. 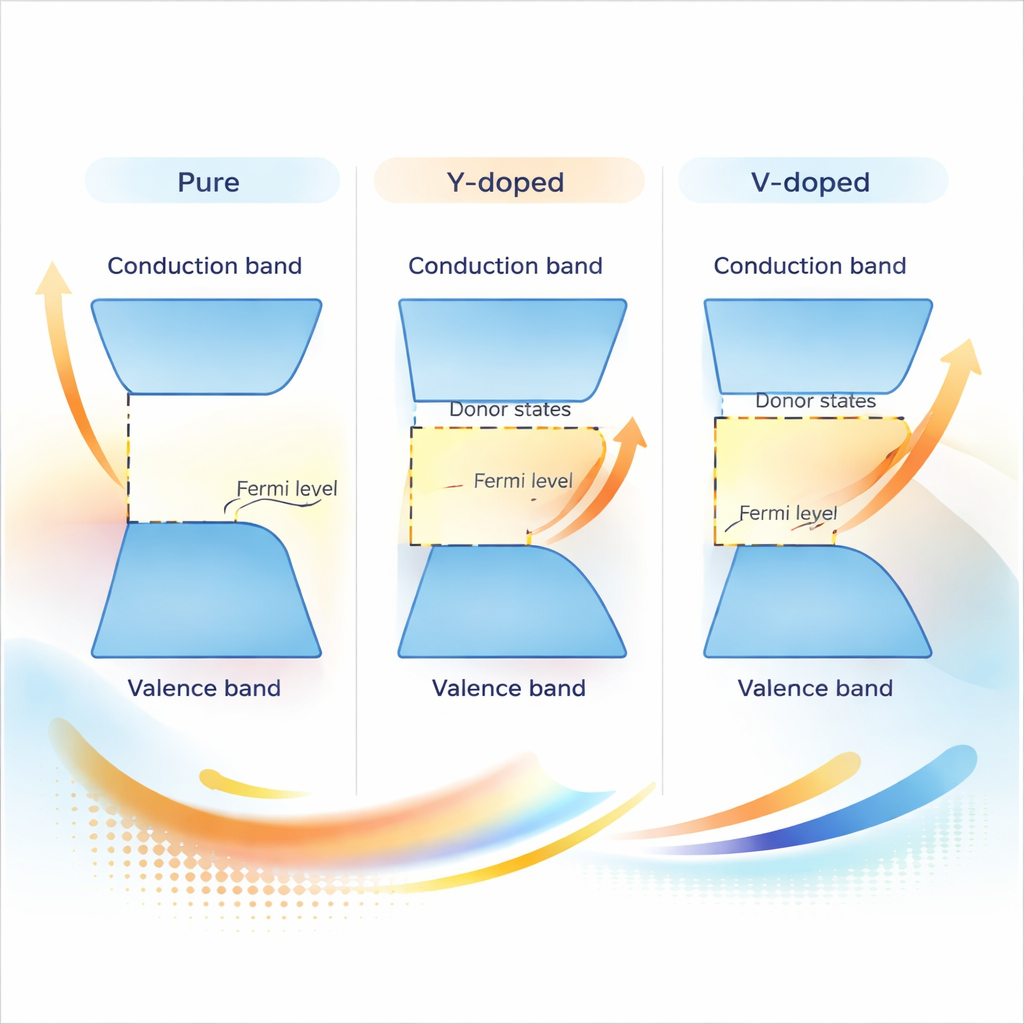
Finding the limits of how much is too much
The study also clarifies that more dopant is not always better. When the researchers pushed vanadium content to a higher level, the simulated crystal showed signs of mechanical instability: one of its key elastic constants became negative, signaling that the lattice would deform under shear. This overly doped version also showed distorted X‑ray patterns, a warning that such compositions may crack or lose long‑range order in real devices. Yttrium, by contrast, could be added more generously without breaking the structure, but its overall balance of properties did not match that of the optimally doped vanadium case.
What this means for future tiny power plants
Put simply, the work shows that carefully choosing and tuning dopants can turn ordinary ZnO into a far more capable material for optoelectronics and energy harvesting. Moderate vanadium doping, in particular, offers a sweet spot where the crystal stays robust, conducts electricity well, and strongly interacts with visible light. While the study is purely computational, it gives experimental teams a precise map of which compositions are most promising to synthesize and test in next‑generation solar cells, transparent conductors, wearable generators, and miniaturized sensors.
Citation: Osama, R.A.A., Siddiqui, K.A., Wang, H. et al. Advancing ZnO nanostructures through strategic transition metal doping. Sci Rep 16, 7443 (2026). https://doi.org/10.1038/s41598-026-37977-y
Keywords: zinc oxide, transition metal doping, optoelectronics, energy harvesting, nanomaterials