Clear Sky Science · en
Exploring the dynamics of chemical species interactions in complex reaction mechanism: classification of fast and slow species and bifurcation analysis
Why the pace of reactions matters
Chemical reactions run everything from car engines to living cells, but not all reacting particles move on the same schedule. Some species appear and disappear in a flash; others change only slowly over time. This paper explores how to sort these fast and slow actors in a key reaction that turns hydrogen and oxygen into water, and how that knowledge can help scientists tame otherwise unwieldy reaction networks in energy, environment, and industrial processes.
Untangling crowded chemical traffic
In complex reactions, dozens of intermediate particles form, react, and vanish long before the final products settle down. Trying to follow every detail quickly becomes overwhelming. The authors focus on a four-step mechanism for hydrogen oxidation, a classic process where hydrogen and oxygen combine to form water. Instead of treating all species alike, they use mathematical tools to separate those that respond almost instantly from those that evolve more gently. This split lets them shrink the full model to a lower-dimensional “backbone” that still captures the overall behavior.
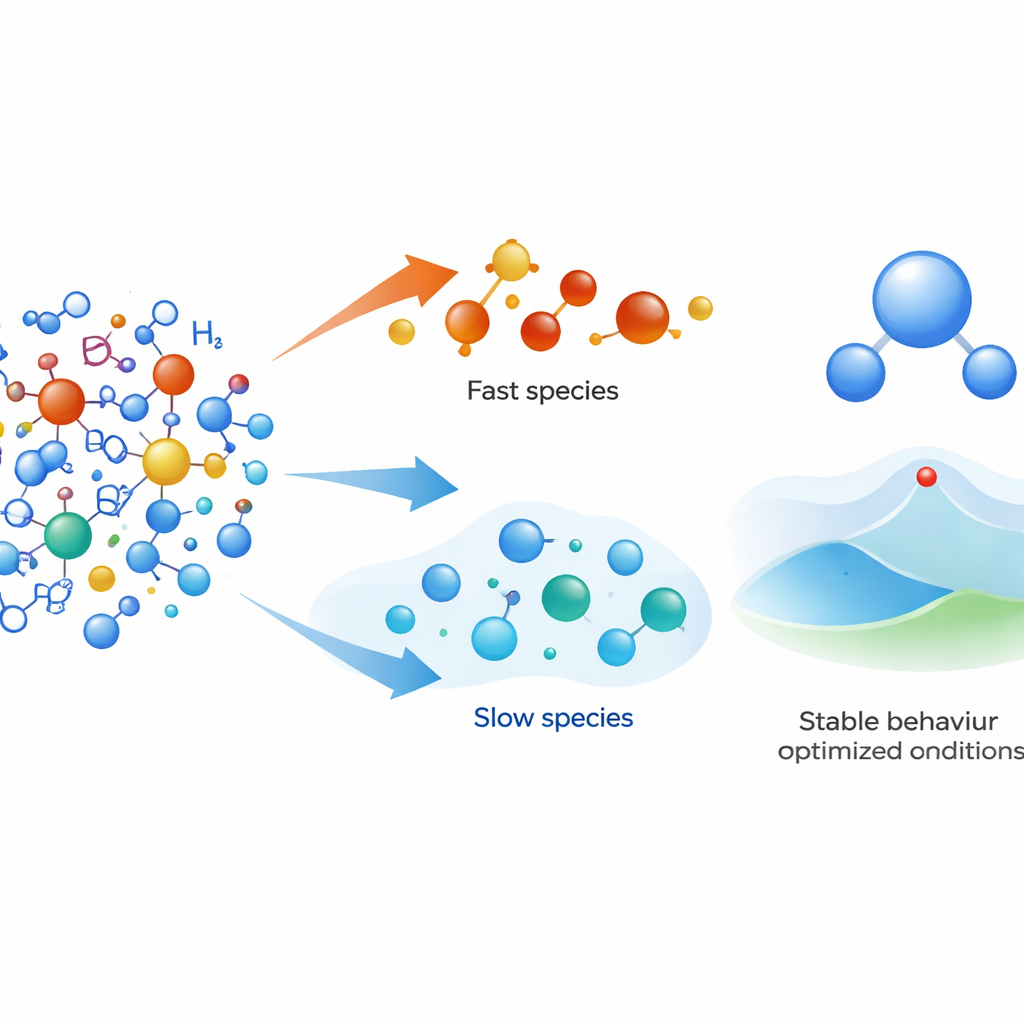
Finding the fast and the slow players
To make this separation precise, the team rewrites the reaction equations in scaled, dimensionless form and applies ideas from singular perturbation theory and quasi steady-state approximations. In simple terms, they look for variables that change so rapidly that, after a brief start-up period, they can be treated as nearly constant in time. Applying this framework to hydrogen, oxygen, hydroxyl, and related species, they show that individual atoms and radicals such as H, O, and OH are the fast movers. By contrast, whole molecules like H2, O2, and H2O behave as slow species, creeping toward their final values on much longer time scales. Time-series plots and numerical data confirm that radicals reach steady levels early, while molecular species keep changing long after.
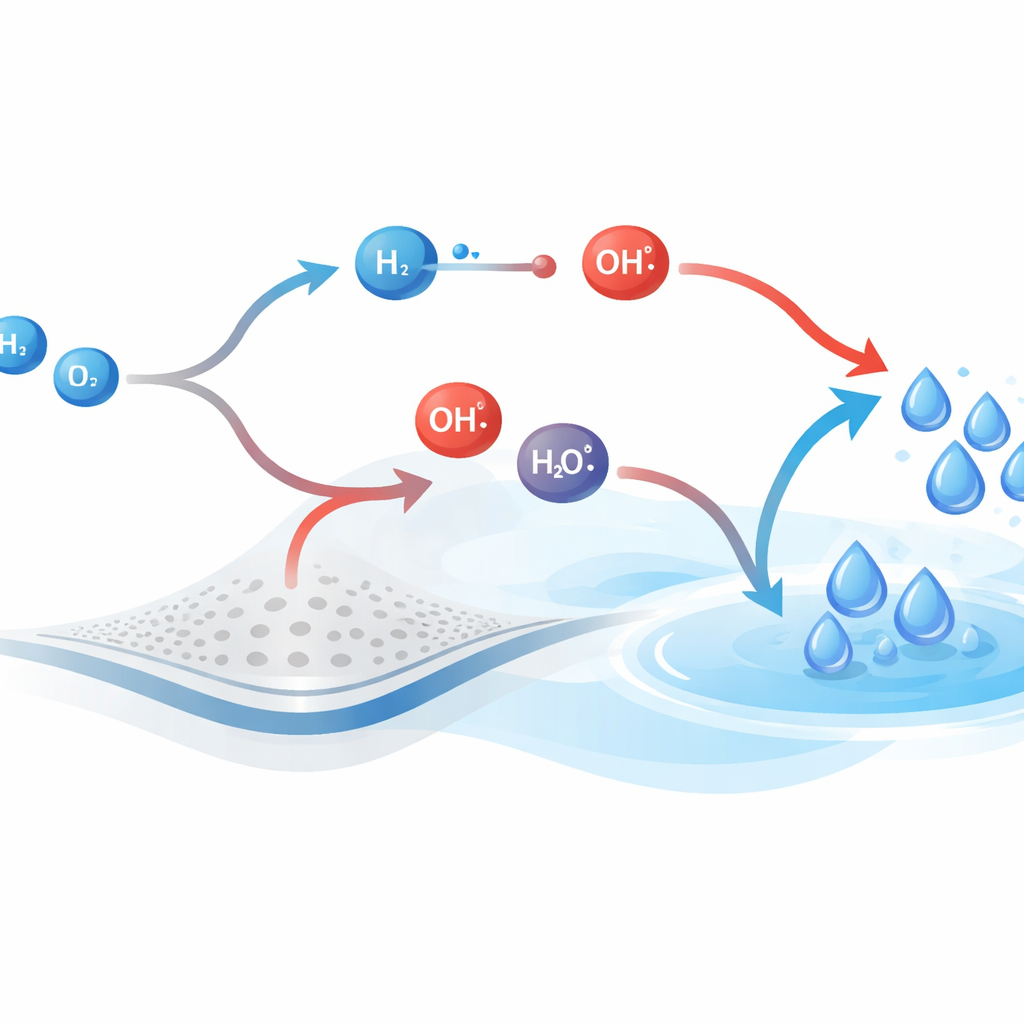
Seeing which pathway fills the pool
Water in this mechanism can form through two competing routes. In one route, hydroxyl (OH) reacts with molecular hydrogen (H2) to produce water and regenerate a hydrogen atom; in the other, OH combines directly with H to make water in a single radical-recombination step. By calculating the instantaneous net flow along each route, the authors track which path contributes more at every moment. Under the chosen conditions, the hydrogen-assisted route carries almost all of the effective traffic toward water, while the direct radical route hovers near balance, with forward and reverse flows nearly canceling out. A time-dependent “dominance ratio” shows the hydrogen-assisted path controlling water formation early and remaining the main contributor as the system approaches steady operation.
Testing which knobs matter most
Knowing who is fast and who is slow is only part of the story; engineers also need to know which parameters are worth tuning. The authors apply both local and global sensitivity analyses to answer this. Local methods probe how small changes in individual rate constants or starting amounts affect specific species, revealing that the hydrogen atom is especially sensitive to variations in certain reaction rates. Global methods, based on Sobol indices, explore the entire range of parameter uncertainty and show that rate constants involving OH and H2 exert the strongest control over how much water is ultimately produced. Together, these tools highlight a small subset of influential parameters hidden inside a large kinetic model.
Mapping where behavior can suddenly change
Finally, the study uses bifurcation surface analysis to explore how steady-state concentrations respond when key rate constants are varied over broad ranges. Visualizing the results as smooth surfaces, the authors find that radicals such as H, O, and OH are highly sensitive, showing curved landscapes that reveal regions of potential multistability or sharp shifts in behavior. In contrast, the slow, stable products H2 and H2O lie on gentler surfaces that change more gradually. This contrast reinforces the idea that time-scale separation is not just a mathematical trick but a structural feature of the reaction network.
What this means for real-world reactions
By combining theory, computer simulations, sensitivity tests, and bifurcation analysis, the authors present a practical recipe for simplifying complex reaction systems without losing their essential physics. For hydrogen oxidation, the work shows that fast radicals steer the early dynamics, a hydrogen-assisted pathway dominates water production, and only a handful of rate constants truly control the outcome. For non-specialists, the key message is that even very complicated chemical networks can be reduced to a manageable core if we learn to identify fast versus slow species and focus on the few parameters that matter most—a strategy that can guide cleaner combustion, better catalysts, and more efficient industrial processes.
Citation: Khatoon, A., Shahzad, M., Elmasry, Y. et al. Exploring the dynamics of chemical species interactions in complex reaction mechanism: classification of fast and slow species and bifurcation analysis. Sci Rep 16, 9486 (2026). https://doi.org/10.1038/s41598-026-37965-2
Keywords: hydrogen oxidation, reaction kinetics, model reduction, sensitivity analysis, bifurcation