Clear Sky Science · en
Synergistic combinatorial anticancer potential of Tamoxifen with Naringin and Diosmetin in MCF-7 breast cancer cells and their liposomal delivery
Why pairing a common drug with citrus compounds matters
Breast cancer is often treated with tamoxifen, a hormone-blocking pill that has saved many lives. But over time, some tumors outsmart the drug, and treatment side effects can be difficult to bear. This study explores an appealing idea: can we make tamoxifen work better and perhaps at lower doses by pairing it with natural substances found in citrus fruits, then packaging them together in tiny fat-based bubbles called liposomes so they reach cancer cells more effectively?
Citrus helpers for a long-standing cancer drug
The researchers focused on two plant compounds, naringin and diosmetin, which are flavonoids commonly found in grapefruits, oranges, and other citrus fruits. In laboratory tests using estrogen-sensitive breast cancer cells (MCF-7) and another breast cancer line (T47D), they first measured how strongly each compound alone could slow or kill cancer cells. Tamoxifen was the most potent, but both naringin and diosmetin also reduced cancer cell survival, especially in MCF-7 cells, hinting that these natural molecules have their own anticancer punch. This set the stage to test whether combining them with tamoxifen would yield more than the sum of their parts.
Stronger together: testing drug combinations
To understand how the drugs interact, the team treated breast cancer cells with tamoxifen plus naringin, tamoxifen plus diosmetin, or all three together, at different fixed ratios. Using a standard mathematical approach for drug pairs, they showed that several combinations were synergistic – meaning the mixture killed more cancer cells than expected from each drug alone. A ratio with roughly twice as much flavonoid as tamoxifen (1:2) was especially effective, particularly for tamoxifen plus naringin. In these settings, the same cancer-killing effect could be achieved with lower amounts of tamoxifen, a key advantage if this translates into fewer side effects in patients.
Triggering cancer cell self-destruction
Next, the scientists asked what was happening inside the cancer cells. They looked at oxidative stress – a buildup of reactive oxygen species – and at signs of programmed cell death, or apoptosis. The combinations, especially the triple mix of tamoxifen, naringin, and diosmetin, led to higher oxidative stress and more pronounced features of apoptosis than tamoxifen alone. Under the microscope, more cells showed the shrunken, brightly stained appearance typical of late-stage cell death. Protein analyses backed this up: the combinations lowered levels of Bcl-2 and Bcl-xL, proteins that normally help cancer cells resist death, and increased activated caspase-3, a key executioner of apoptosis. In simple terms, the citrus compounds appeared to help push tamoxifen-treated cells over the edge into self-destruction.
Packaging drugs into tiny carriers
Although the combinations looked promising, naringin and diosmetin are not very water-soluble and can be poorly absorbed. To tackle this, the team created liposomes – nanometer-scale spheres made from natural fats – that could carry tamoxifen and the flavonoids together. They optimized the lipid composition and processing so that the resulting particles were about 150–210 nanometers across, fairly uniform, and carried a surface charge that helps keep them stable in suspension. Imaging confirmed the liposomes were smooth and roughly spherical. Tests showed that a high fraction of each drug could be encapsulated, and that release into a surrounding liquid occurred slowly over 24 hours, following diffusion-driven kinetics. This kind of controlled release is desirable because it can maintain steady drug levels for longer periods.
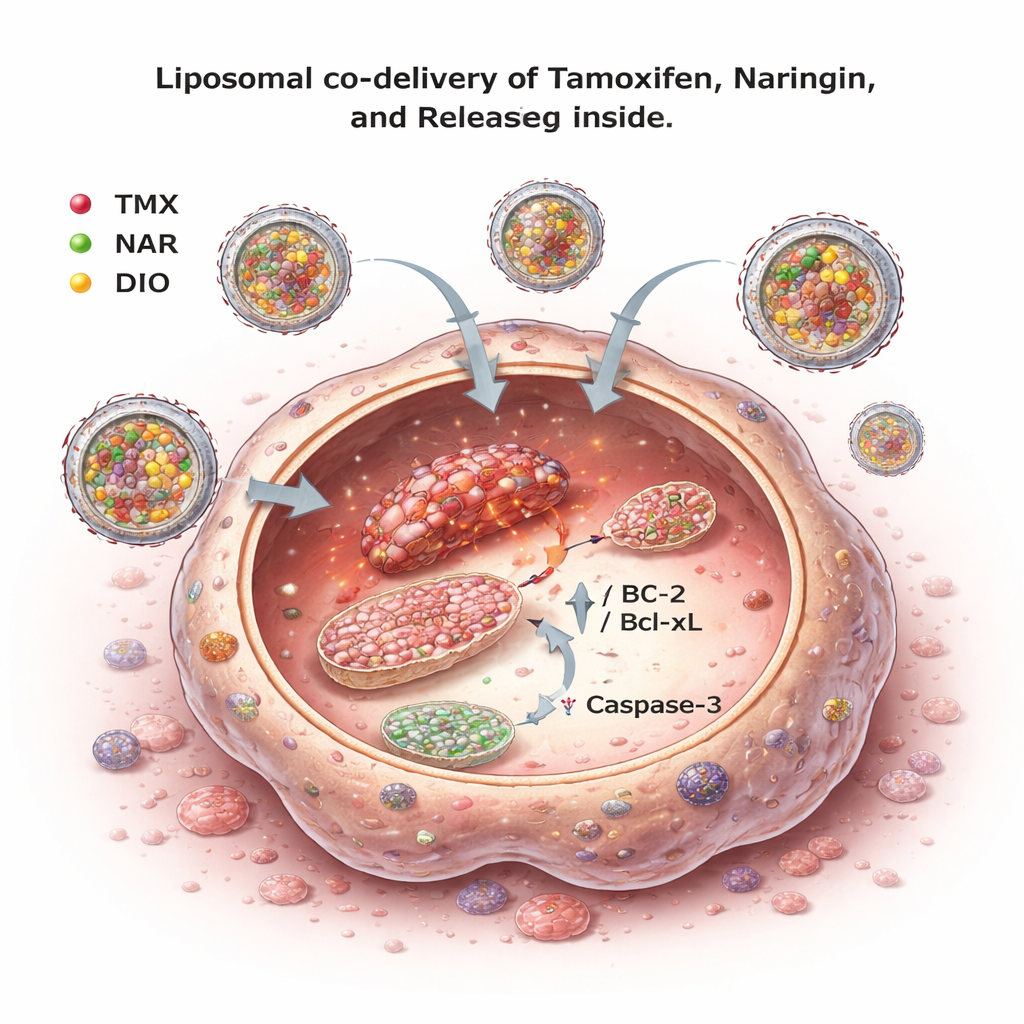
Hitting tumors harder while sparing healthy cells
When these liposomal formulations were tested again on breast cancer cells, the triple-drug liposomes (tamoxifen, naringin, and diosmetin together) were more effective at reducing cancer cell viability than liposomes containing only tamoxifen or only two of the drugs. Importantly, when the same formulations were applied to a non-cancerous human cell line, there was little toxicity within the tested dose range, suggesting a degree of selectivity for cancer cells. Overall, the results indicate that combining tamoxifen with citrus-derived flavonoids and delivering them in liposomes can enhance cancer cell killing while potentially allowing for lower and safer doses of tamoxifen.
What this could mean for future breast cancer care
For a layperson, the take-home message is that pairing a standard breast cancer drug with well-chosen natural compounds, then bundling them into smart nano-sized carriers, may make treatment more effective and possibly gentler. The study shows, in cell cultures, that tamoxifen works better together with naringin and diosmetin: they help switch cancer cells from survival mode into self-destruct mode and do so efficiently when co-delivered in liposomes. While this work is still at the laboratory stage and needs to be confirmed in animals and eventually in clinical trials, it outlines a promising strategy: using combination therapy plus nanotechnology to outsmart resistant breast tumors and improve long-term outcomes.
Citation: Uniyal, P., Das Pramanik, S., Pandey, S. et al. Synergistic combinatorial anticancer potential of Tamoxifen with Naringin and Diosmetin in MCF-7 breast cancer cells and their liposomal delivery. Sci Rep 16, 7646 (2026). https://doi.org/10.1038/s41598-026-37954-5
Keywords: breast cancer, tamoxifen, flavonoids, liposomal delivery, combination therapy