Clear Sky Science · en
Generation of TCRγδ + T cells from human embryonic stem cells
New Ways to Arm the Immune System
Cancer treatments increasingly enlist the body’s own immune system to hunt down tumors. One especially promising type of immune cell, called a gamma-delta T cell, can kill cancer cells without needing to recognize the exact tissue type of the patient—making it a strong candidate for “off‑the‑shelf” therapies. But these cells are rare in adult blood, and expanding them in large numbers is difficult. This study describes a way to grow gamma-delta T cells in the lab starting from human embryonic stem cells, potentially opening the door to more accessible cancer immunotherapies.
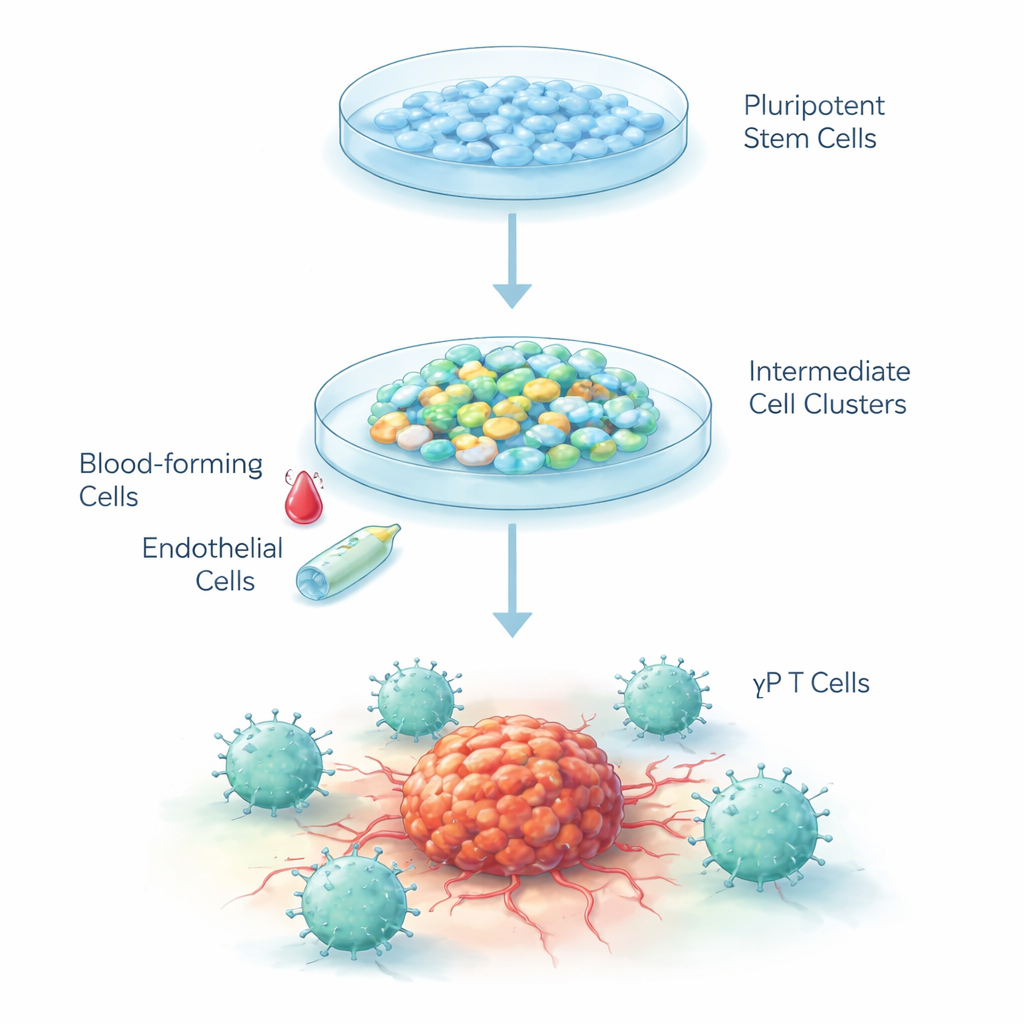
Turning Blank-Slate Cells into Cancer Fighters
Human embryonic stem cells are “blank-slate” cells that can, in principle, become almost any tissue in the body. The researchers designed a step‑by‑step recipe to coax these cells to develop along the same pathway they follow in the embryo when the blood and immune system first form. They used a three‑stage process: first guiding stem cells toward early blood‑vessel–like cells, then into immature blood-forming cells, and finally into T cells of the gamma-delta type. The cells were grown as three‑dimensional clusters in special liquid culture under low‑oxygen conditions that mimic the developing embryo, and then brought back to normal oxygen levels as they matured.
Building the Immune Cell Assembly Line
In the first stage, chemical signals nudged stem cells to become mesoderm, the early layer that gives rise to blood and vessels. Within six days, nearly 40% of the cells in each cluster showed markers of “hematopoietic endothelial cells,” a kind of transitional cell that can give rise to blood. In the second stage, changing the growth factors triggered these endothelial-like cells to undergo a switch into free‑floating blood progenitor cells. Over 99% of these floating cells carried a key early blood marker, and about 90% expressed another marker of blood cells, indicating a remarkably pure population with strong potential to continue down the immune-cell pathway.
Arriving at Gamma-Delta T Cells
For the final stage, the team supplied signals known to encourage T‑cell development, including a Notch‑activating protein that mimics cues normally found in the thymus. After 25 days, more than 40% of the cells had matured into gamma-delta T cells, identified by their characteristic surface receptors and a “double‑negative” profile (lacking the usual CD4 and CD8 tags found on most other T cells). Importantly, very few conventional alpha‑beta T cells were produced, showing that the protocol strongly favors the gamma‑delta branch. Genetic analysis over time revealed that the cells switched on and off groups of genes in an orderly sequence that closely mirrors how blood and T cells form inside the body.
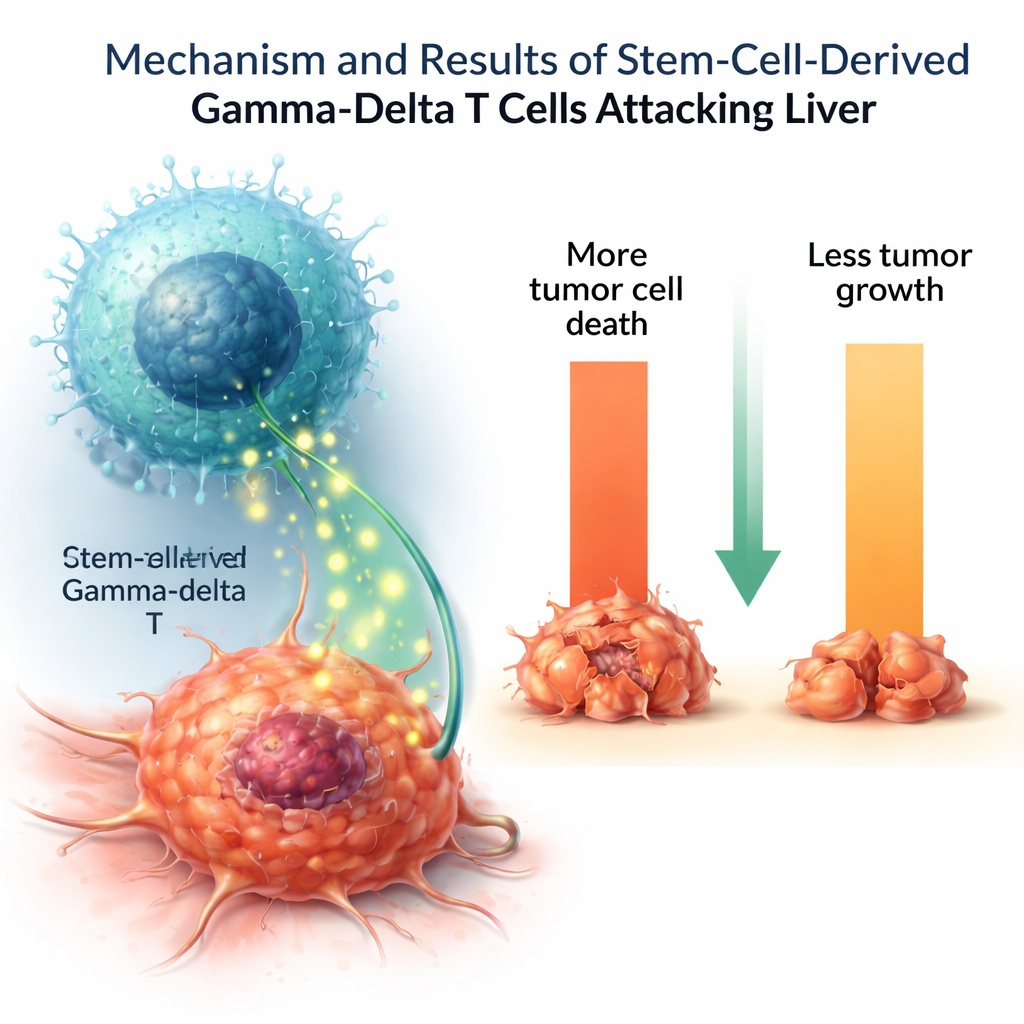
Putting Lab-Grown Cells to the Test
To see whether these lab‑made cells functioned as true cancer fighters, the researchers exposed them to several human tumor cell lines. The gamma-delta T cells killed a substantial fraction of liver cancer cells and slowed their growth, while having little effect on healthy control cells. When stimulated, they secreted important immune messenger molecules such as tumor necrosis factor and interleukin‑2, which help coordinate and amplify immune attacks. Some other killing molecules were present at lower levels than in natural gamma-delta T cells, suggesting that the lab‑grown cells may not be fully mature or may represent a specific functional subtype.
What This Could Mean for Future Cancer Care
By recreating a streamlined version of early blood development in a dish—without using animal feeder cells—the scientists produced large numbers of gamma-delta T cells in under a month, faster and more simply than previous methods. While more work is needed to purify the final cell mix, scale up production, and adapt the recipe to clinically approved, serum‑free conditions, this approach points toward a renewable source of standardized gamma-delta T cells. If these cells prove safe and effective in further testing, they could become a widely available building block for next‑generation, off‑the‑shelf immunotherapies against cancers such as liver tumors.
Citation: Zhang, X., Chen, C., Fu, Y. et al. Generation of TCRγδ + T cells from human embryonic stem cells. Sci Rep 16, 6762 (2026). https://doi.org/10.1038/s41598-026-37941-w
Keywords: gamma-delta T cells, cancer immunotherapy, human embryonic stem cells, cell differentiation, liver cancer