Clear Sky Science · en
Early prediction of colonization by carbapenemase-producing enterobacterales at ICU admission using machine learning
Why hidden hospital germs matter
Many of the sickest patients in a hospital are treated in intensive care units (ICUs), where powerful antibiotics and invasive devices are common. In this environment, a dangerous group of gut bacteria—carbapenemase‑producing Enterobacterales, or CPE—can quietly take hold. People who carry these germs may not feel sick, but they can spread them to others or later develop life‑threatening infections that are hard to treat. This study asks a practical question: can we predict, at the moment someone enters the ICU, who is likely already carrying CPE so that staff can protect other patients more effectively?
A quiet threat in the ICU
CPE are gut bacteria that have learned to resist carbapenems, some of the strongest antibiotics used when other drugs fail. In South Korea, CPE infections have surged in recent years, echoing a global trend. ICU patients are especially at risk because they often stay in the hospital longer, undergo more procedures and receive more antibiotics than other patients. Hospitals can use rectal swabs to detect CPE, but results take time, and it is unrealistic to place every new ICU patient in isolation until the lab report arrives. The authors set out to build a tool that uses information already in the medical record at ICU admission to estimate which patients are likely CPE carriers.
Mining hospital records for clues
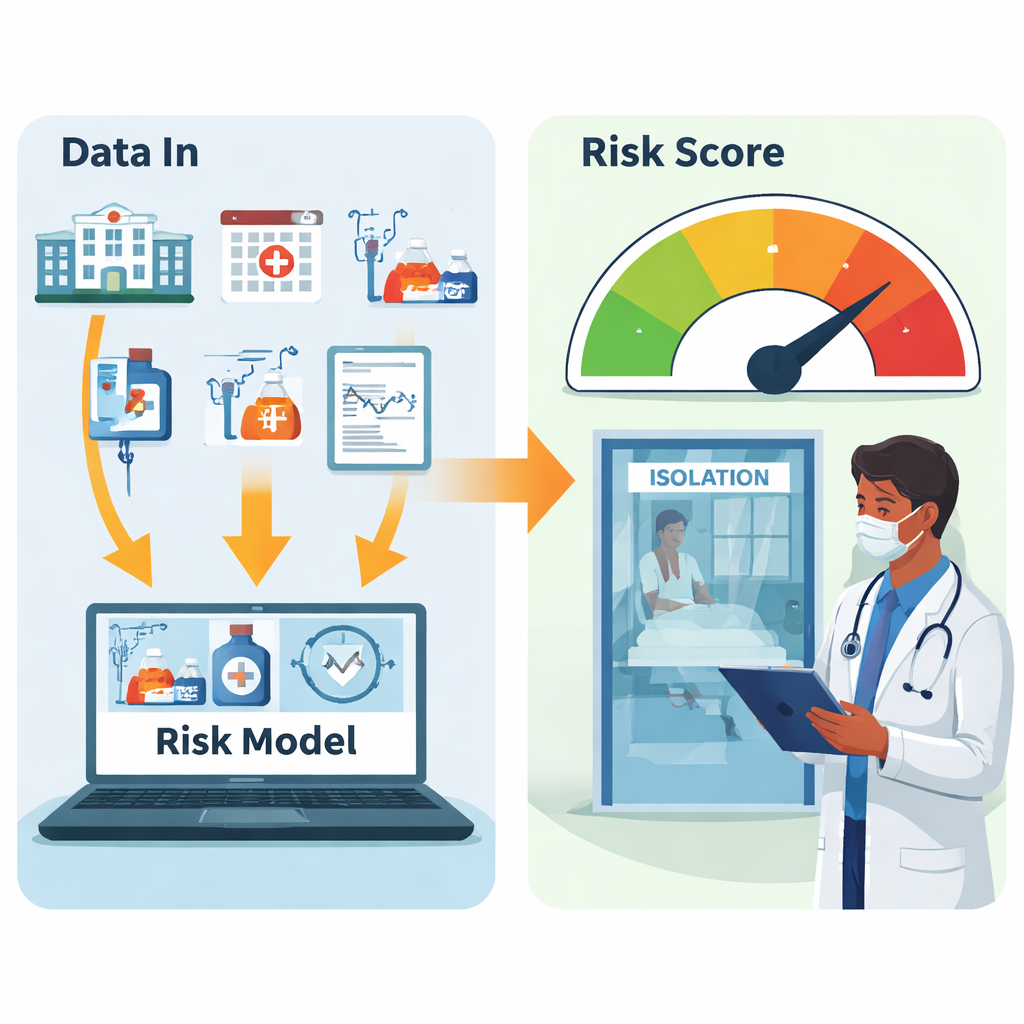
The researchers examined 4,915 adult ICU admissions at a large South Korean hospital between 2022 and 2023. All of these patients had rectal swabs taken within 48 hours of arriving in the ICU. About 9.2 percent—453 people—were found to be colonized with CPE. From the electronic medical records, the team pulled 42 pieces of information available at admission, including age, recent hospital and long‑term care stays, past surgeries, underlying illnesses, prior antibiotic use, and whether the patient had tubes or catheters in place. Using these variables, they compared ten different machine‑learning approaches to see which one best separated CPE carriers from non‑carriers.
A simple model with strong ruling‑out power
Rather than favoring the most complex algorithm, the study found that a relatively straightforward method—logistic regression—struck the best balance for real‑world use. With a chosen risk cut‑off, the model correctly identified about 73 percent of carriers and classified 96 percent of predicted non‑carriers as truly negative. In practical terms, when the tool says a patient is unlikely to carry CPE, it is almost always right. This is crucial for infection control teams that must decide who really needs scarce isolation rooms. Other, more sophisticated models were more specific but missed many true carriers, making them less safe for this purpose.
Who is most at risk?
To keep the tool understandable for clinicians, the authors focused on 12 key predictors. Having a biliary drain—a tube that drains bile from the liver—was linked to the highest odds of CPE carriage. Other strong signals included recent residence in a long‑term care facility, the presence of a nasogastric feeding tube or a central venous catheter, recent steroid treatment, prior use of multiple antibiotics, and more days spent in the hospital before entering the ICU. A history of colonization or infection with another hardy germ, vancomycin‑resistant Enterococci, also raised the odds. The team used SHAP (Shapley Additive Explanations), a method that shows how each factor pushes a patient’s risk up or down, so individual predictions are not a mysterious “black box.”
From numbers to bedside decisions
To make the research usable beyond the data set, the team built a free web‑based calculator (www.cpepredictor.com). Clinicians can enter answers to 14 simple questions at ICU admission—for example, whether the patient recently took certain antibiotics or has particular tubes in place—and the tool instantly estimates the chance of CPE colonization. The authors stress that the model is best used to rule out low‑risk patients, not to definitively label people as carriers. A positive result should prompt early isolation or rapid molecular testing, not replace standard lab cultures. While the study took place at a single hospital and will need validation elsewhere, it shows how carefully designed, interpretable machine‑learning tools can help hospitals target infection‑control resources where they are needed most, reducing the spread of highly resistant bacteria without overwhelming already stretched ICUs.
Citation: Kim, J.H., Yang, E., Lee, Y.W. et al. Early prediction of colonization by carbapenemase-producing enterobacterales at ICU admission using machine learning. Sci Rep 16, 6705 (2026). https://doi.org/10.1038/s41598-026-37927-8
Keywords: antibiotic resistance, intensive care unit, infection control, machine learning in medicine, hospital-acquired infections