Clear Sky Science · en
Assessment of the durability of polyurea resin coatings against selected aggressive solutions in the sewage infrastructure environment
Why Sewer Coatings Matter to Everyday Life
Hidden beneath our streets, concrete pipes and tanks quietly carry away and clean our wastewater. If these structures fail, repairs are costly, disruptive, and can threaten the environment. This study examines how a modern protective material—polyurea resin coatings—stands up to harsh chemicals commonly found in sewage treatment plants. Understanding which chemicals weaken these coatings, and how fast, helps cities design sewer systems that last longer and stay safer.
A Tough Skin for Concrete Structures
Polyurea coatings act like a seamless, rubbery skin sprayed onto concrete. They cure in seconds and are prized for their flexibility, water tightness, and resistance to many chemicals. Because they can bridge tiny cracks and cling strongly to concrete, engineers increasingly use them to protect tanks, channels, and other components in wastewater treatment plants. In those environments, however, the coatings must survive not just water and grit, but also an ever-changing soup of acids and organic chemicals produced by industry and by the breakdown of household waste.
The Three Trouble-Makers in Sewage
The researchers focused on three chemicals that are particularly important in sewage systems: sulfuric acid, phenol, and urea. Sulfuric acid forms when bacteria convert hydrogen sulfide gas in poorly ventilated pipes, driving the local acidity so low it can eat away at bare concrete. Phenol comes mainly from industrial and pharmaceutical sources and is known to attack many building materials even at low levels. Urea, a major component of urine, breaks down into ammonia and carbon dioxide and has also been linked to material degradation. While typical concentrations in real plants are usually low, the team deliberately used higher, “accelerated test” levels to mimic many years of exposure in just a few weeks.
How the Coatings Were Put to the Test
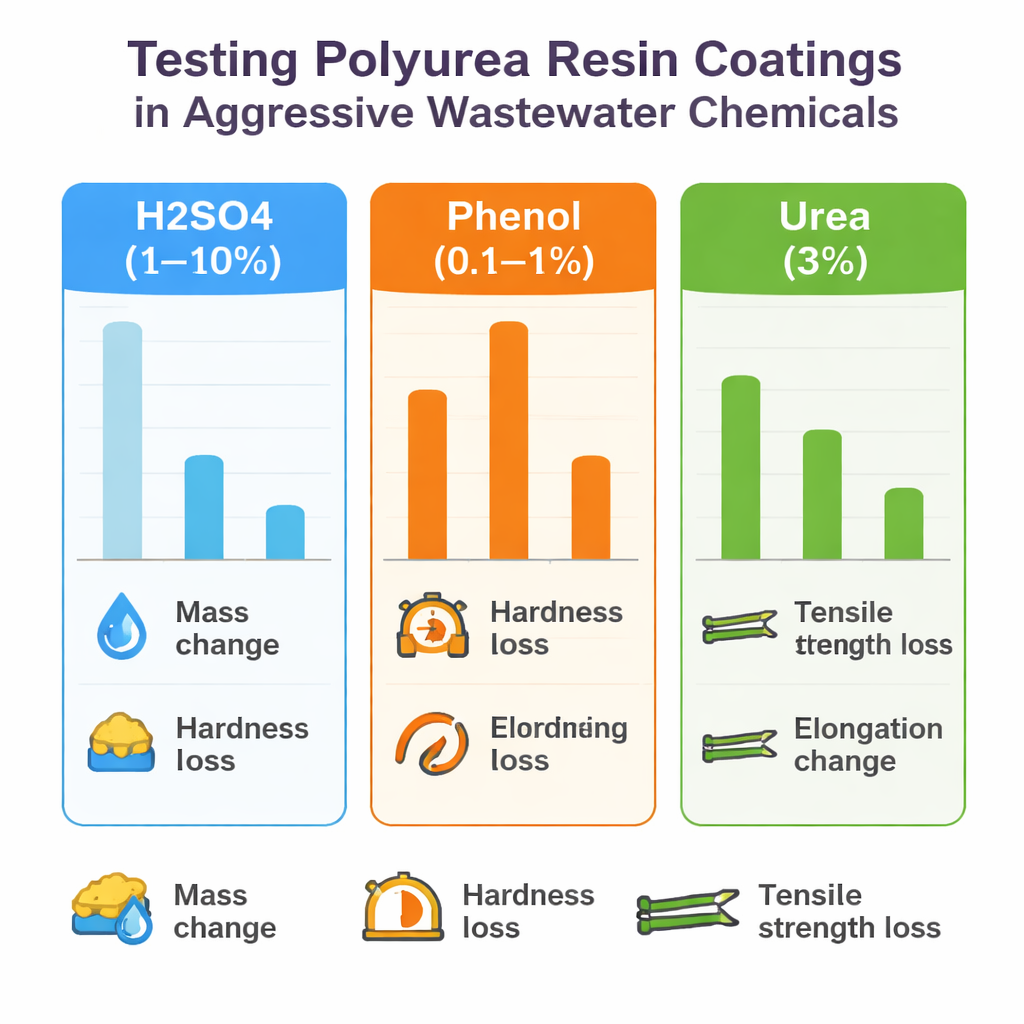
Three commercially available polyurea coatings of different thicknesses and starting strengths were sprayed onto test panels and allowed to cure under controlled conditions. The samples were then fully immersed for 7 or 28 days in one of five solutions: 1% or 10% sulfuric acid, 0.1% or 1% phenol, or 3% urea. After soaking, the team examined the samples with the naked eye for cracking, blistering, and discoloration; weighed them to see how much liquid they had absorbed; measured surface hardness with a standard indentation tool; and stretched strips of coating until they reached their maximum tensile strength to see how much their strength and stretchability had changed.
What Survived, What Suffered
At first glance, all coatings looked surprisingly intact. Apart from slight yellowing in one product after strong acid and a loss of gloss in another after phenol, there were no obvious cracks or peeling. The hidden story emerged in the measurements. Sulfuric acid, even at 10%, turned out to be the gentlest of the three: it caused only small changes in weight, up to about 10% loss of hardness, and a 10–30% drop in tensile strength. Urea had a stronger softening effect, especially after longer soaking. Coatings in 3% urea gained more moisture and could lose up to roughly 13% of their hardness, making them more vulnerable to scratching and abrasion, though their tensile strength still fell only in the 10–30% range.
Phenol: The Silent Coating Killer
Phenol was in a different league. Even at just 0.1%, it caused moderate swelling and noticeable weakening of the coatings, with tensile strength reductions of 40–60%. At 1%, phenol turned from “harmful” to truly destructive: coatings soaked up to 30% more mass, their hardness dropped by up to about a quarter, and their tensile strength plunged by as much as 80%. The ability of the coatings to stretch safely before reaching their maximum strength also declined sharply. A combined damage score, which averaged changes in mass, hardness, strength, and stretch, confirmed phenol as by far the most aggressive chemical, urea as intermediate, and sulfuric acid as the least harmful.
What This Means for Sewage Systems
For non-specialists, the takeaway is clear: polyurea coatings are a promising, durable choice for protecting concrete in most sewage environments, especially where sulfuric acid is the main threat. They withstand even strong acid with only modest loss of performance. In systems where urea levels are significant, engineers should still allow for some softening and the risk of abrasion by grit. However, in areas that may contain even modest amounts of phenol, these coatings can deteriorate quickly and may not provide reliable long-term protection. In such cases, either more robust coating formulations or different materials altogether will be needed to keep the hidden backbone of our wastewater infrastructure sound.
Citation: Francke, B., Michalak, H., Kula, D. et al. Assessment of the durability of polyurea resin coatings against selected aggressive solutions in the sewage infrastructure environment. Sci Rep 16, 6806 (2026). https://doi.org/10.1038/s41598-026-37921-0
Keywords: polyurea coatings, wastewater infrastructure, concrete protection, chemical corrosion, sewage treatment plants