Clear Sky Science · en
Effects of alkali contamination on mechanical properties and microstructure of red clay
Why the Ground Beneath Factories Matters
Across many industrial areas, high‑pH liquids from processes like metal refining and paper making can leak into the soil. When those alkaline liquids meet red clay—a common foundation soil across southern China and many other warm, wet regions—the ground can quietly soften, swell, or even harden and crack. This study asks a deceptively simple question with major safety implications: how does the strength and inner structure of red clay change as more and more alkaline solution soaks in?
Red Clay Under Chemical Stress
Red clay is a weathered, iron‑rich soil that often forms the base for buildings, roads, and slopes. Because it contains minerals that react readily with strong alkalis, it can be both vulnerable to pollution and, intriguingly, a candidate for chemical strengthening. The researchers recreated real‑world leaks from alumina plants by mixing red clay with sodium hydroxide (a common strong alkali) at six concentrations, from none at all up to very strong solutions. After letting the samples cure for ten days, they measured how hard the clay was to shear, and used several microscopic and laboratory techniques to see how its pores, grains, and minerals had changed. 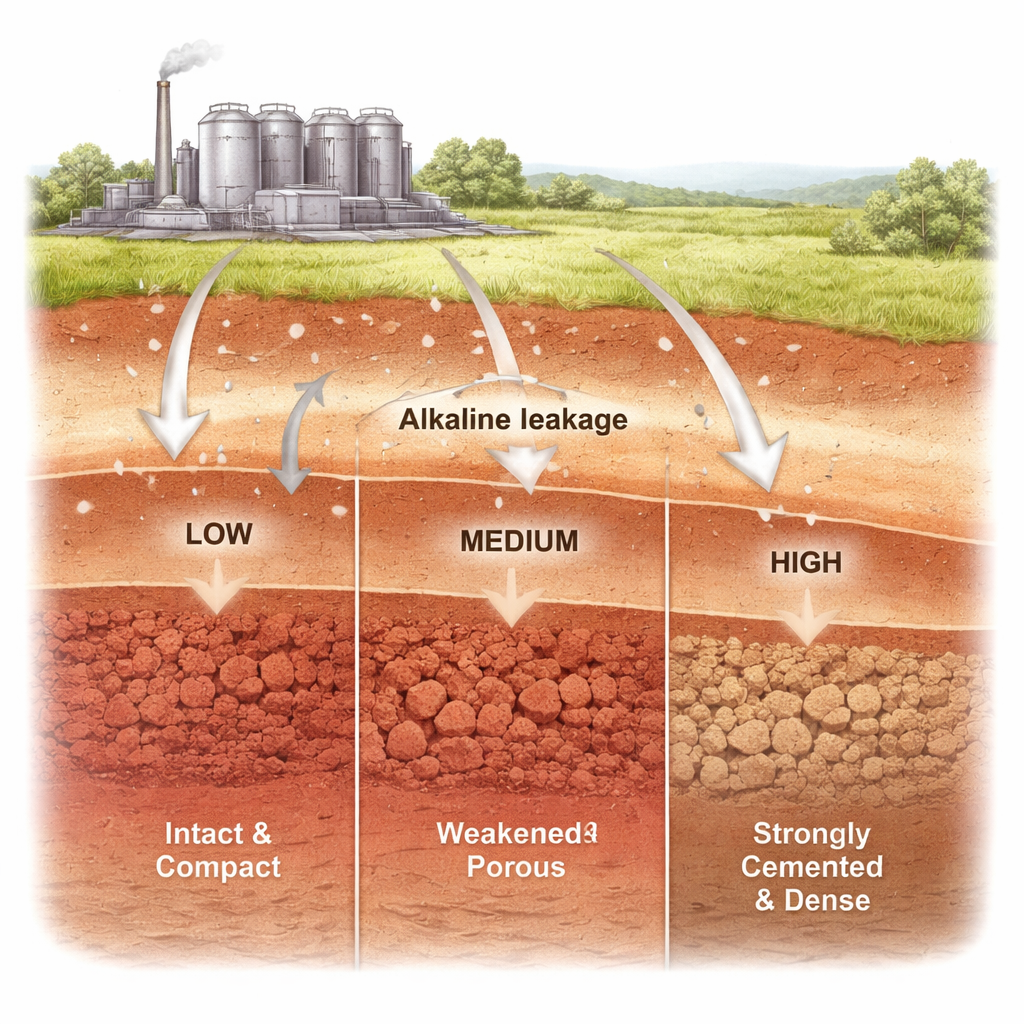
A Surprising Sweet Spot of Weakness
One of the most striking findings is that the clay does not simply get weaker as more alkali is added. Instead, its strength follows a “V‑shaped” curve with clear thresholds. At a modest alkali level, around 3.5 percent by mass, the clay becomes its weakest. Stress–strain tests show that under this contamination, the soil softens considerably: both its cohesion (the “stickiness” that helps hold grains together) and its internal friction (how well grains resist sliding) drop to their lowest values. Microscopic pore measurements reveal why. The total void space shrinks, but the share of large pores grows, and the inner walls of the pores become smoother. Under the microscope, once‑interlocked plate‑like particles are broken down into finer pieces coated in soft, gel‑like material, creating a more deformable, silty mass that is easy to shear apart.
When Pollution Starts Acting Like Glue
As alkali levels rise further, the balance flips. At about 14 percent, the clay is no longer at its weakest but at its strongest. Here, the dissolved ingredients from the clay minerals reorganize into new, rigid binding phases. X‑ray tests detect fresh crystals of a sodium aluminosilicate, evidence of a geopolymer‑type cement forming between particles. Pore measurements show many more tiny pores and far fewer large ones, while a fractal analysis of the pore surfaces indicates they have become rougher and more intricately textured. Electron microscope images confirm that particles now clump into larger, interlocked aggregates stitched together by fine cementing material. In mechanical tests, this heavily “re‑cemented” clay reaches its peak shear strength, although it behaves in a brittle way: it can carry high loads, but once it cracks, its strength falls off quickly. 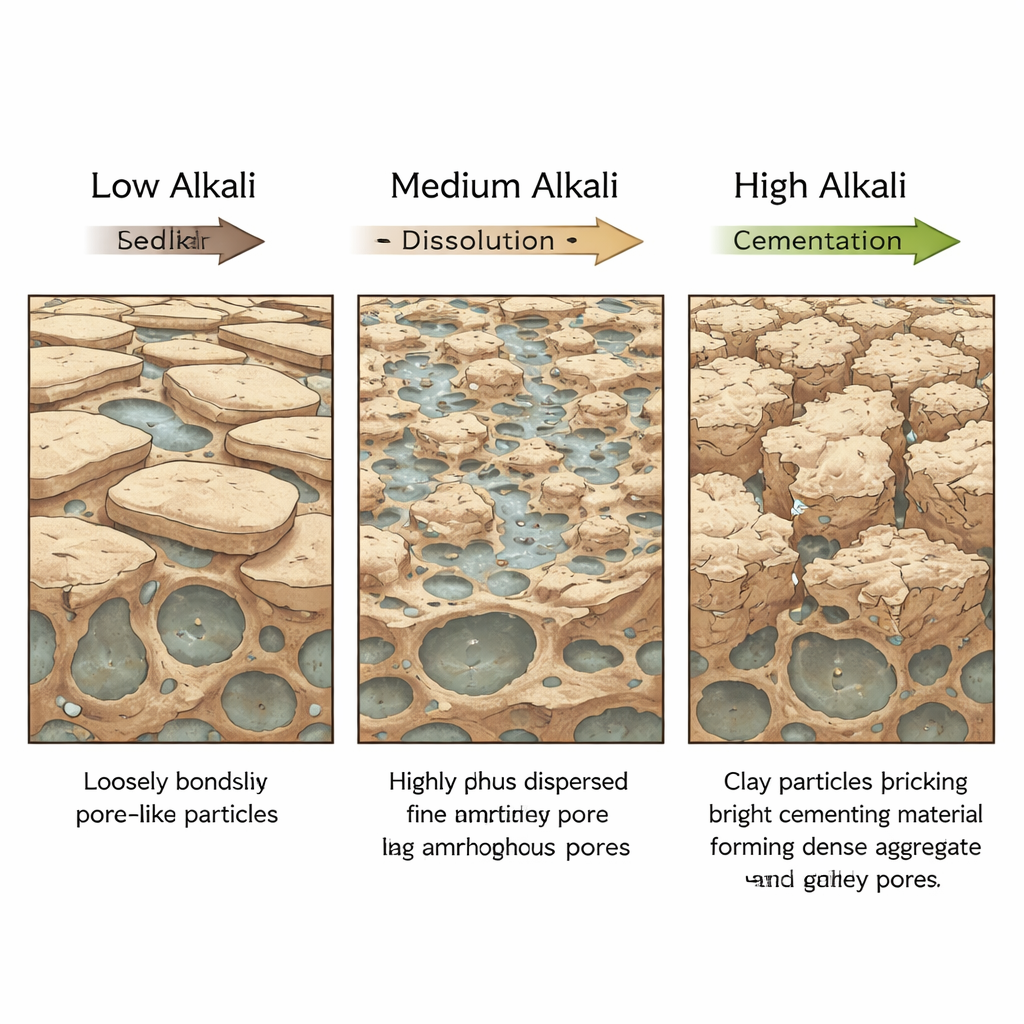
Too Much of a Good Thing
At the highest concentration tested, 21 percent, the trend reverses again. The extra free alkali does not simply strengthen the clay further. Instead, it resumes an aggressive dissolving role, attacking both the original minerals and the newly formed cement. The aggregate structure begins to break down, particle sizes drop, and larger pores re‑emerge. The clay still carries more load than the untreated soil, but its strength is clearly lower than at 14 percent. This suggests there is an upper chemical limit beyond which the new cement network is no longer stable and begins to erode.
What This Means for Safety and Design
For non‑specialists, the key message is that strong alkaline leaks can either destroy or rebuild the structure of red clay, depending on how concentrated they are. At modest contamination levels, the clay quietly weakens and becomes more deformable, undermining foundations or slopes. At a carefully controlled higher level, similar chemistry can be harnessed to create new mineral “glue” that binds grains and fills pores, significantly stiffening the soil—albeit in a brittle, crack‑prone form. Push the concentration too far, and the glue itself is eaten away. These insights help engineers assess the risks of alkali pollution beneath industrial sites and point to how alkali‑based treatments might one day be tuned to deliberately strengthen red clay foundations, provided long‑term durability and environmental side effects are thoroughly managed.
Citation: Wang, L., Chen, J., Liu, D. et al. Effects of alkali contamination on mechanical properties and microstructure of red clay. Sci Rep 16, 6715 (2026). https://doi.org/10.1038/s41598-026-37873-5
Keywords: red clay, alkali contamination, soil strength, geopolymerization, ground stability