Clear Sky Science · en
SARS-CoV-2 nucleocapsid protein forms complexes with soluble complement regulatory proteins that can bind to the virion
Why this hidden viral trick matters
Most of us have heard about the coronavirus “spike” protein, because it is the main target of vaccines. But inside the virus is another protein, called the nucleocapsid or N protein, that turns out to be far more than structural packaging. This study shows that the N protein can latch onto key human immune regulators in the blood and ferry them onto the virus surface. By doing so, SARS-CoV-2 may partly switch off one of our earliest defense systems—the complement system—helping the virus survive and possibly contributing to severe COVID-19.
A closer look at the virus’s inner protein
The N protein is the virus’s genome “wrapper,” helping to package and stabilize its RNA. Doctors have found high levels of N protein circulating in the blood of patients with COVID-19, especially in severe cases, and earlier work suggests it can stir up inflammatory signals. The authors wondered whether N might also interfere with complement, a network of blood proteins that mark invaders for destruction and can punch holes in microbes. Many viruses have evolved clever ways to borrow or imitate the body’s own complement regulators to avoid being attacked. The team asked three questions: does N bind the spike protein and the virus itself, does it bind human complement regulatory proteins in blood, and can it carry these regulators onto the virus surface?
How N and spike meet on the virus
Using several protein-binding tests, the researchers showed that N protein can stick directly to spike protein, whether spike is made in the lab or presented on real SARS-CoV-2 particles. They confirmed this interaction in different experimental setups and measured its strength, finding an affinity similar to that of many antibodies. By trimming the spike protein into fragments and identifying which pieces remained attached to N, they mapped likely contact zones mainly to the spike’s N-terminal domain, a region already known as a hot spot for antibody binding and for docking certain host molecules. This suggests that N can form a tight, biologically meaningful bridge to spike on actual virions.
Hijacking the body’s own safety switches
The complement system is tightly controlled by soluble “brakes” called complement regulatory proteins, which protect our own cells from friendly fire. The team focused on four of these—C1-inhibitor, C4-binding protein, factor H, and vitronectin—because they act at different stages of the complement cascade. In blood-serum–based tests and purified protein assays, the N protein bound all four regulators, whereas spike did not bind any of them under the same conditions. This means N can form N–CRP complexes in plasma. Crucially, when N was pre-mixed with human serum and then exposed to virus particles, the researchers detected these regulatory proteins attached to the virions—but only when N was present. The more N they used, the more complement regulators appeared on the virus, pointing to a dose-dependent recruitment mechanism. 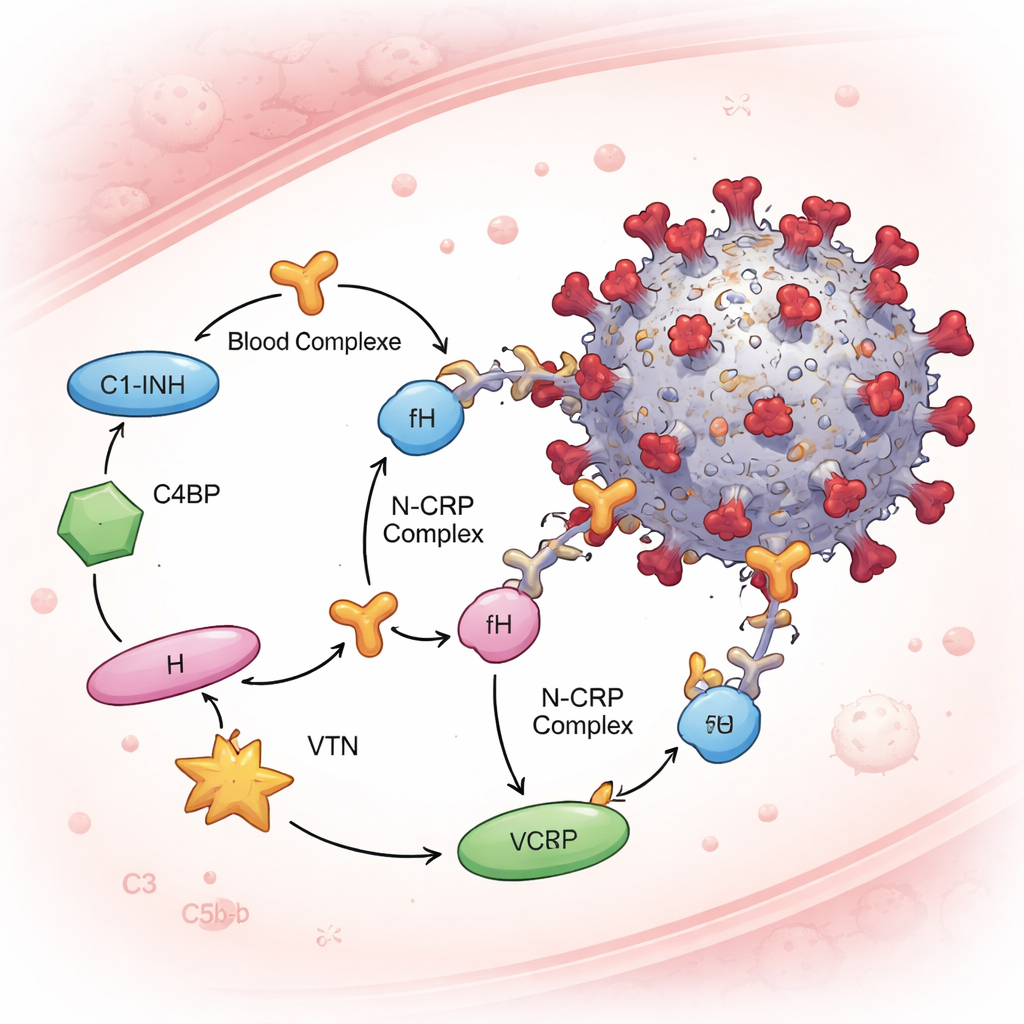
Turning down the final attack step
The ultimate weapon of the complement system is the membrane attack complex (MAC), a ring-like structure that forms pores in cell or viral envelopes. The authors tested whether N could change MAC formation on SARS-CoV-2. They incubated purified virus with human serum, with or without added N protein, and then measured how much of the MAC’s C5b-9 complex ended up on the virions. Without N, the virus accumulated C5b-9, indicating active complement attack. When N was present, MAC deposition dropped in a concentration-dependent way: more N meant less C5b-9 on the virus. This fits the idea that N, by decorating the virion with complement regulators, shields it from being punched full of pores. 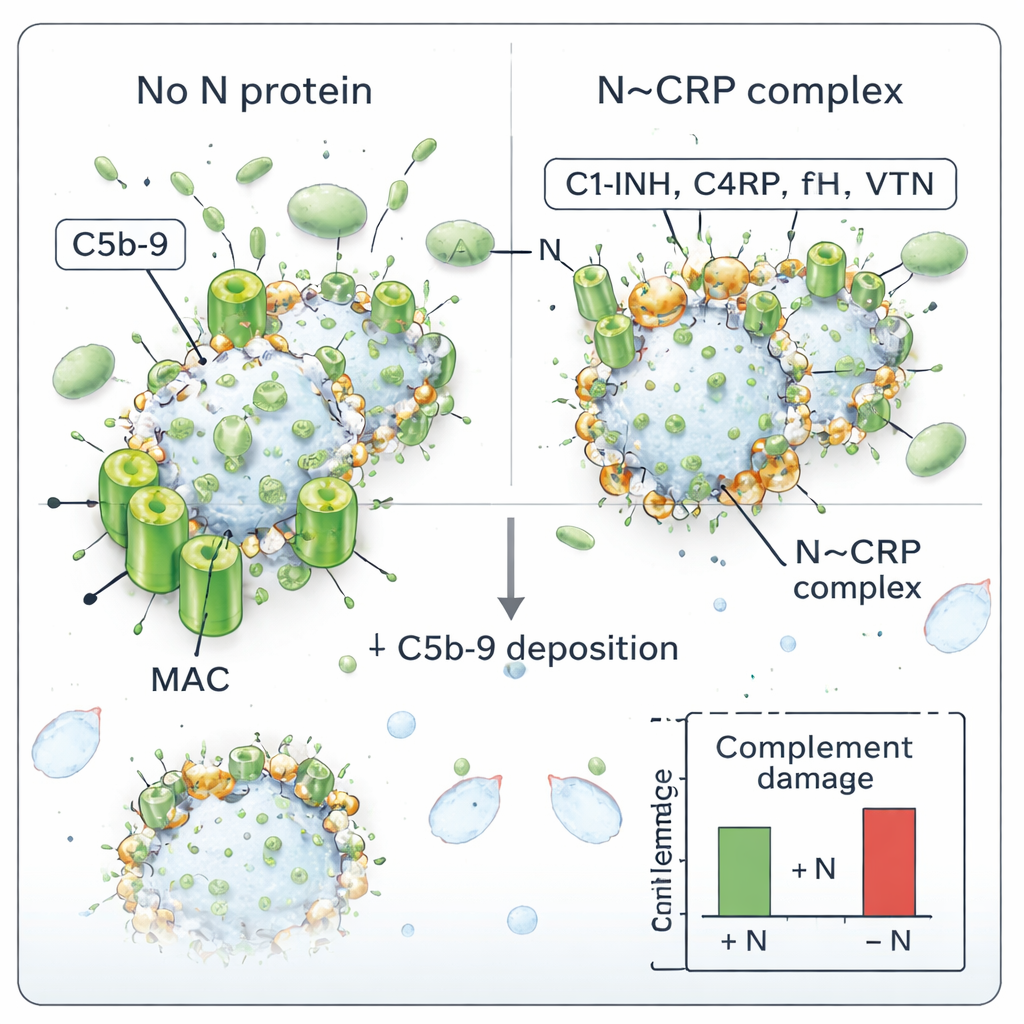
What this means for patients and future therapies
To a non-specialist, the take-home message is that SARS-CoV-2 does not rely only on its famous spike to outsmart the immune system. Its internal N protein can escape into the bloodstream, grab the body’s own complement “brakes,” and carry them back onto the virus surface. There, they may help the virus hide from a powerful arm of innate immunity and reduce direct complement-mediated damage. This could contribute both to persistent infection and to the tangled complement disturbances seen in severe COVID-19. While these experiments were done in controlled laboratory settings, they highlight N–complement interactions as a potential new target: therapies that block N from binding these regulatory proteins, or that restore balanced complement activity, might one day help tip the battle back in favor of the immune system.
Citation: Víglaský, J., Bhide, K., Talpasova, L. et al. SARS-CoV-2 nucleocapsid protein forms complexes with soluble complement regulatory proteins that can bind to the virion. Sci Rep 16, 6599 (2026). https://doi.org/10.1038/s41598-026-37866-4
Keywords: SARS-CoV-2 nucleocapsid, complement system, immune evasion, complement regulatory proteins, COVID-19 severity