Clear Sky Science · en
Process modeling and sludge characterization of electrocoagulation for the removal of oil-in-water emulsions and calcium from petroleum refinery wastewater
Why cleaning refinery water matters
Modern oil refineries turn crude oil into the fuels and lubricants we rely on every day, but they also generate vast amounts of dirty water loaded with oil droplets and dissolved minerals such as calcium. If this water is not cleaned properly, it can foul equipment, waste valuable water resources, and pollute rivers and seas. This study explores a promising electrical treatment method that can tackle two major problems at once—oily pollution and hard-water minerals—while keeping energy use and cost in check.
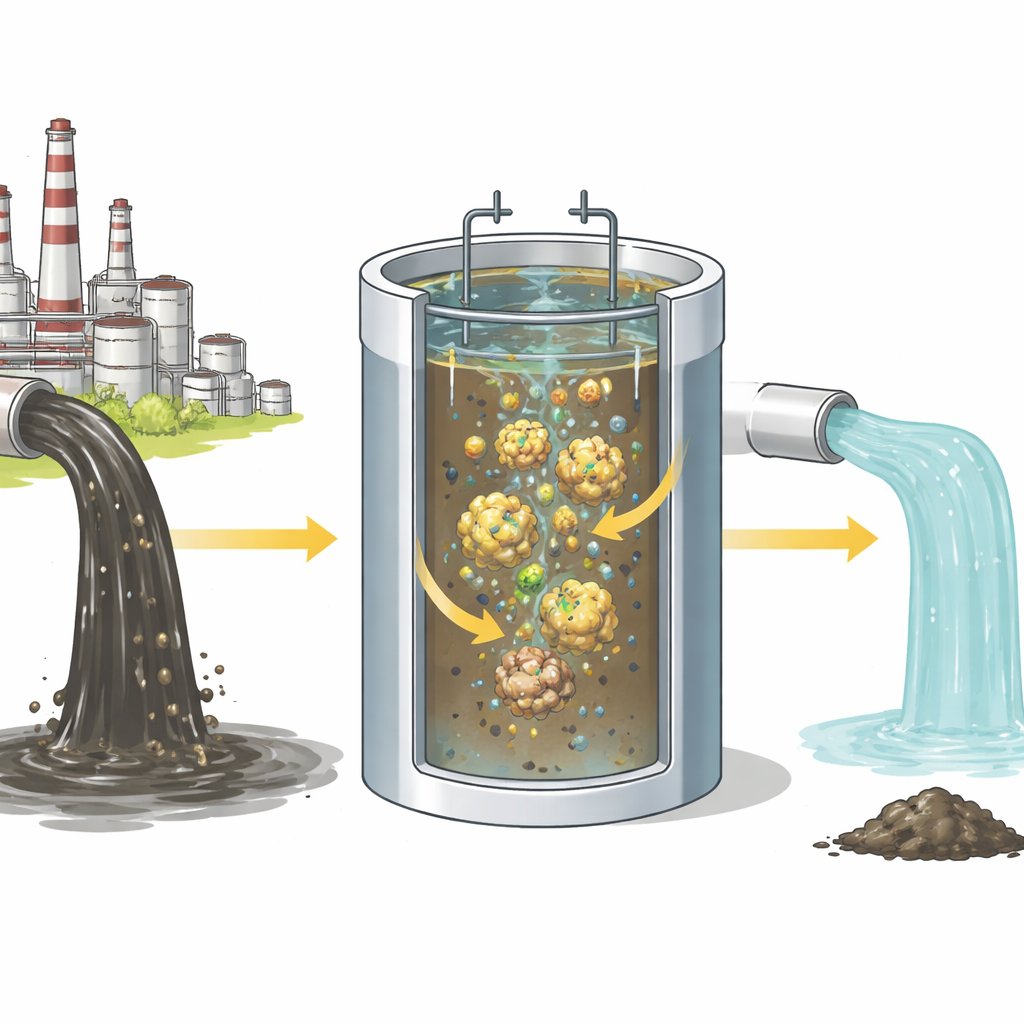
An electrical way to trap dirt in water
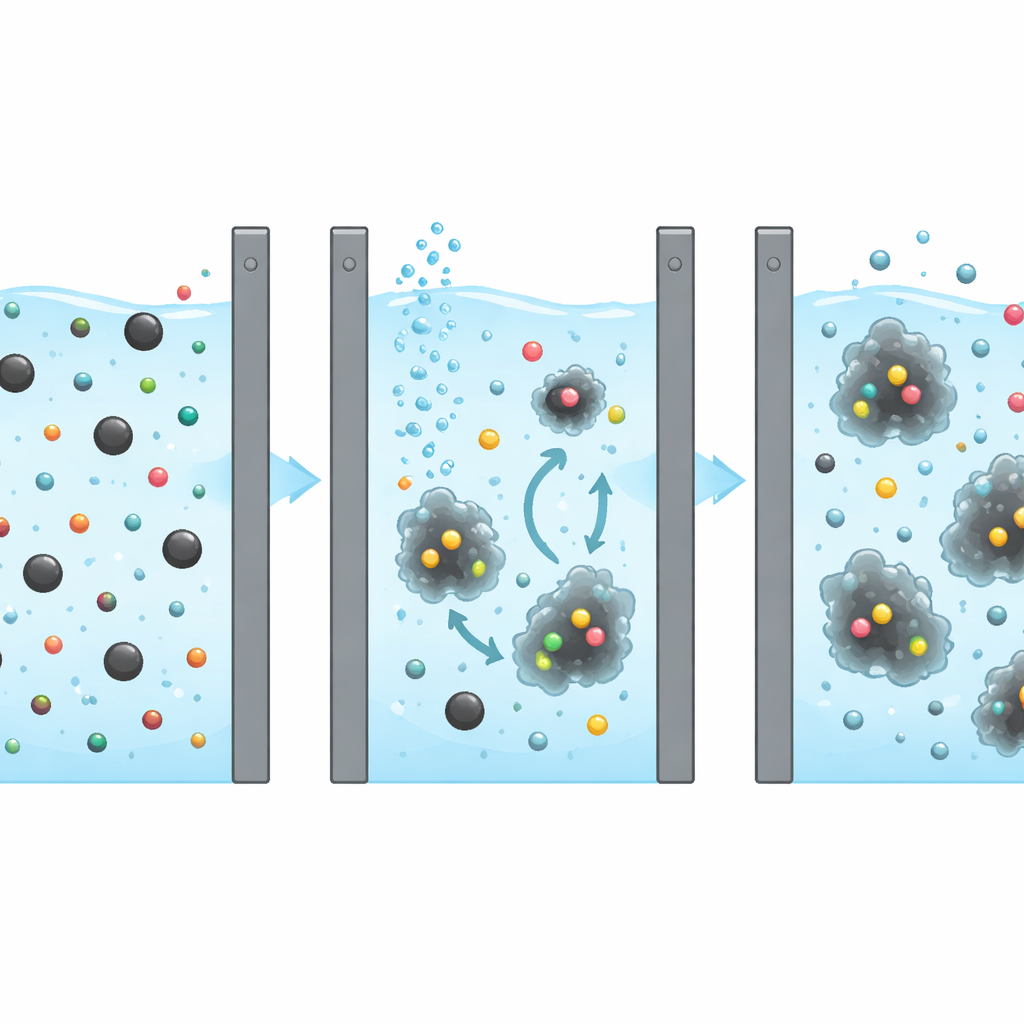
The researchers focused on a process called electrocoagulation, in which metal plates inside a reactor are connected to a direct current power supply. When electricity passes through the water, tiny amounts of aluminum dissolve from one plate and react with water to form fluffy, sticky particles. These particles can grab onto oil droplets and dissolved calcium, clumping them together into larger bits that either float to the surface as scum or sink as sludge. Unlike traditional chemical treatment, this method generates its own “cleaning chemicals” from the metal plates, reducing the need to add external reagents.
Designing the best recipe for cleaner water
Cleaning performance depends on many knobs that engineers can turn: how long the current is applied, how strong that current is, how salty the water is, how acidic or basic it is, and how much oil and calcium it contains. Instead of trial-and-error, the team used a structured statistical approach to explore this multi-dimensional space. They created synthetic refinery wastewater with controlled amounts of lubricating oil and calcium salts, then systematically varied six key conditions: treatment time, pH, current density, salt (sodium chloride) level, and starting concentrations of oil and calcium. Specialized software helped them plan 84 experiments and fit mathematical models that relate these inputs to removal of oil and calcium, along with energy use and operating cost.
What the experiments and models revealed
The analysis showed that treatment time was the single most important factor for removing both oil and calcium: more time generally gave the aluminum-based flocs longer to form and capture pollutants. Current density and salt level also played strong roles, but in more complex ways. Higher current helped once enough time had passed, improving removal by generating more flocs and gas bubbles, but at short times it could disturb floc formation. Moderate amounts of calcium and salt improved electrical conductivity, but too much led to hard mineral layers on the electrodes and unwanted side reactions that wasted aluminum and reduced efficiency. The pH of the water mattered as well: slightly alkaline conditions around pH 9 favored the formation of aluminum species that are especially good at breaking oil emulsions and binding calcium.
Finding a sweet spot for performance and cost
By combining the experimental data with response surface modeling, the team identified a set of operating conditions that jointly optimize oil removal, calcium removal, and cost. Under these conditions—pH 9, a moderate-to-high current density, specific starting oil and calcium levels, modest added salt, and a treatment time of about an hour and a half—the system removed over 91 percent of oil and nearly 73 percent of calcium. At the same time, it used about 12 kilowatt-hours of electricity per cubic meter of water and achieved a total operating cost of about 0.21 US dollars per cubic meter, lower than some earlier electrocoagulation studies. Computer simulations using COMSOL software confirmed that at these settings, the electric field inside the cylindrical reactor is distributed more uniformly, helping reactions proceed efficiently throughout the water volume.
What happens to the captured waste
After treatment, the trapped pollutants appear as a mixture of sludge and floating scum. The authors examined this material with infrared spectroscopy, X-ray elemental analysis, and electron microscopy. They found that it contained aluminum hydroxide structures along with oil-derived carbon and calcium and sodium salts, forming porous, irregular particles with large surface area. These characteristics suggest that the sludge might be reused rather than simply discarded—for example, as a soil amendment where its mineral content could improve soil properties, or as a source of aluminum that could be recovered and recycled into new treatment chemicals.
Cleaner water from a simpler system
Overall, the study demonstrates that a relatively simple electrocoagulation reactor, powered by modest electrical currents and using readily available aluminum electrodes, can simultaneously strip out emulsified oil and calcium from refinery wastewater at competitive cost. By carefully tuning operating conditions and backing experiments with statistical modeling and computer simulation, the authors show that this technology can turn heavily contaminated industrial water into a much cleaner stream, while producing a manageable sludge that may even have secondary uses. For communities and industries facing water scarcity and strict discharge limits, such optimized electrical treatment offers a practical path toward safer and more sustainable water reuse.
Citation: Mohamed, Y.E., El-Gayar, D.A., Amin, N.K. et al. Process modeling and sludge characterization of electrocoagulation for the removal of oil-in-water emulsions and calcium from petroleum refinery wastewater. Sci Rep 16, 7954 (2026). https://doi.org/10.1038/s41598-026-37854-8
Keywords: petroleum refinery wastewater, electrocoagulation, oil-in-water emulsion, water hardness removal, wastewater treatment optimization