Clear Sky Science · en
The mTOR signaling pathway regulates key steps of mammary gland organoid genesis in a temporal manner
Why tiny breast models matter
Our breasts are among the few organs that repeatedly rebuild themselves: they grow during puberty and pregnancy, produce milk, then shrink back after nursing. Understanding how this complex remodeling is controlled is crucial for women’s health, from breastfeeding success to cancer risk. This study uses miniature three-dimensional “mini-breasts,” called organoids, grown from mouse cells to reveal how a master growth switch inside cells, known as the mTOR pathway, times and shapes the development of mammary tissue.
A cellular traffic light for growth
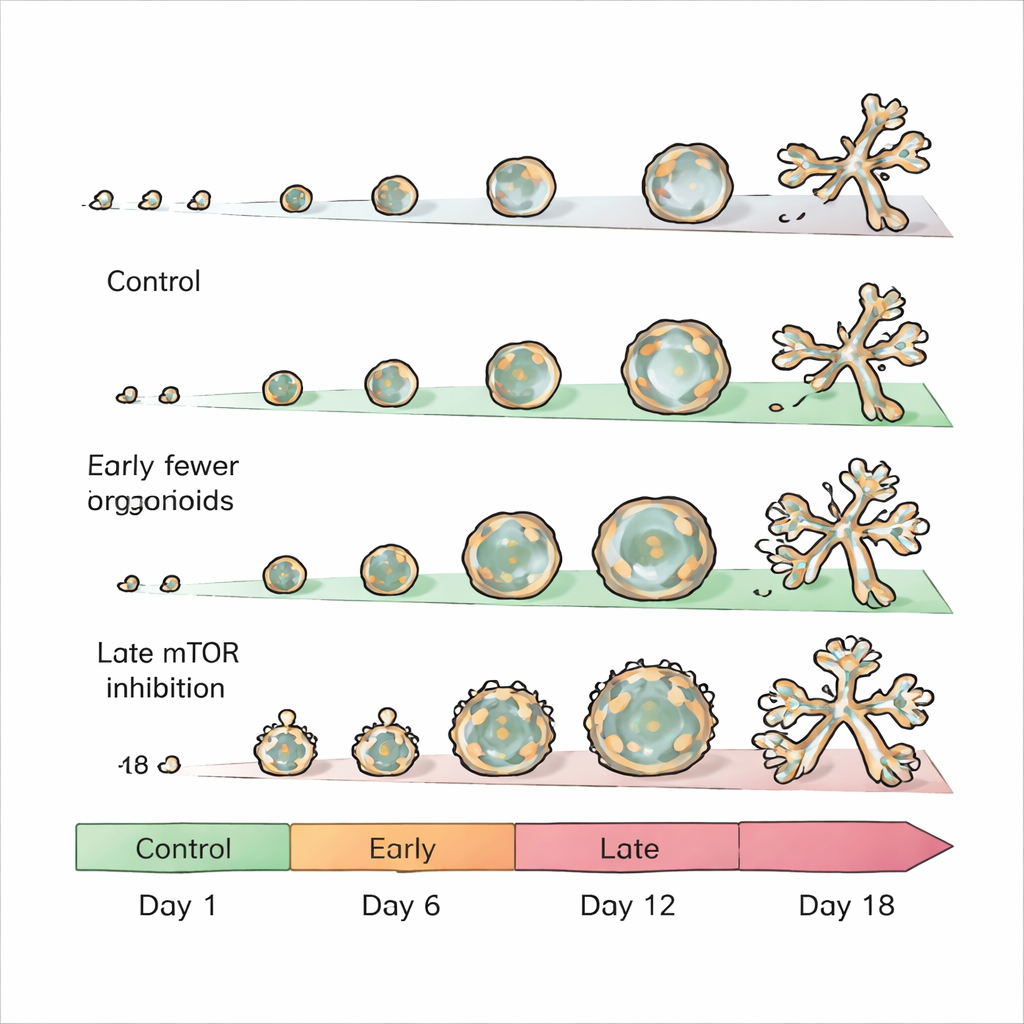
mTOR is a central controller that helps cells decide when to grow, divide, and change their metabolism. The researchers asked how turning this switch off at different moments would affect organoid development. They grew mouse mammary epithelial cells in a gel that lets them self-organize into structures resembling tiny breast ducts with a hollow center and, sometimes, branched “grape-like” clusters. They then added well-known mTOR-blocking drugs, rapamycin and torin 1, either very early, in the middle of growth, or later, when branching usually begins.
Early interference: mini-organs that never quite form
When mTOR was blocked from the very first day, organoids hardly grew at all. Their diameter was cut by roughly two-thirds or more, showing that active mTOR is essential for the early expansion of mammary tissue. This is consistent with mTOR’s reputation as a driver of cell growth and protein production. Because these organoids remained so small and underdeveloped, the team focused on later treatment windows, where the effects were more nuanced and revealing about how the tissue’s internal architecture is controlled.
Mid-course change: hollow spheres and lost flexibility
Blocking mTOR starting around day 6—after initial structures had formed but before branching—produced unexpectedly large, mostly hollow organoids. These were dominated by luminal cells, the inner cells that would normally line milk-secreting ducts, while the outer basal cell layer was almost completely lost. In healthy development, individual luminal or basal cells can each give rise to organoids containing both cell types, a property known as lineage plasticity. mTOR inhibition at this mid-stage essentially froze this flexibility, forcing cells into a luminal identity and preventing the usual balance of layers. Molecular analyses showed widespread changes in gene and protein levels, particularly reductions in components of the cell’s metabolic machinery and in proteins that help remodel the surrounding support matrix—changes that help explain the altered shape and cell makeup.
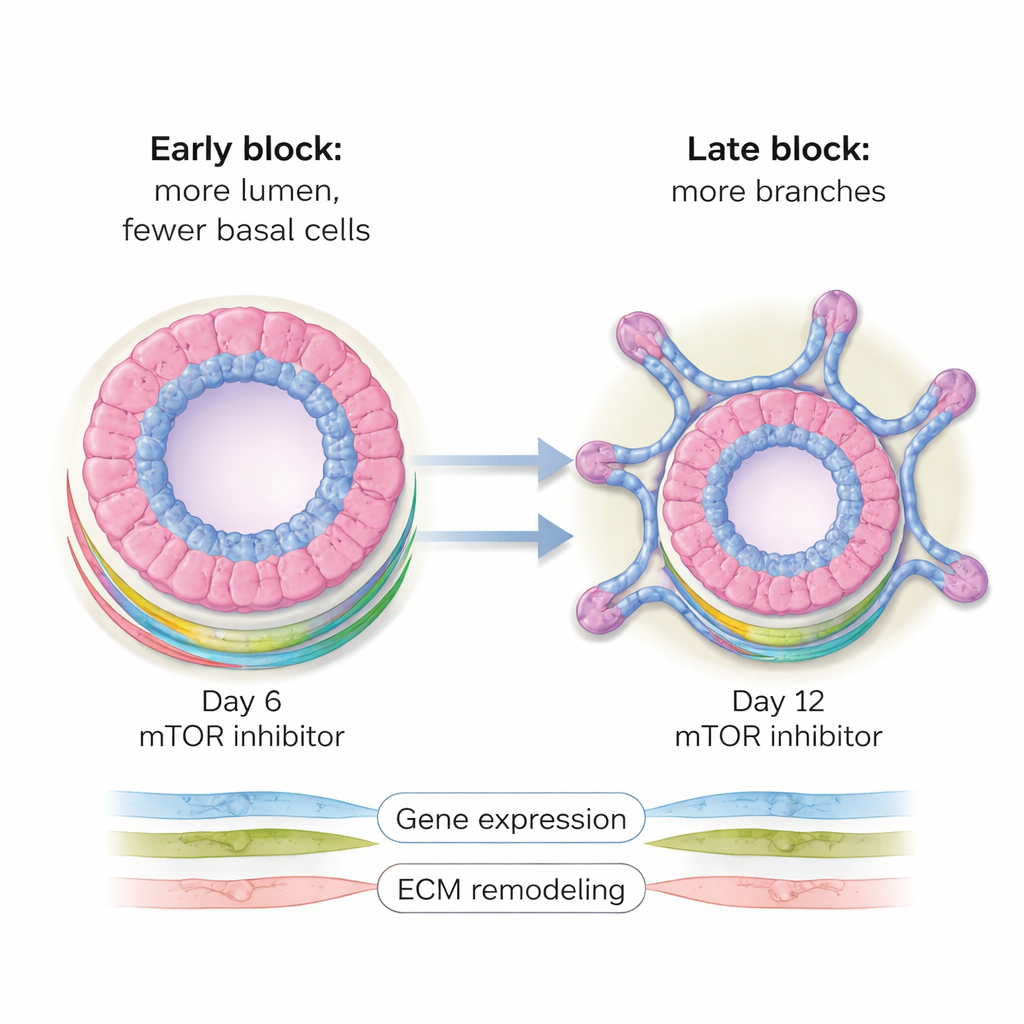
Late interruption: more branches, different wiring
When the same drugs were added later, around day 12, just as branching typically begins, the outcome flipped. Instead of enlarged hollow spheres, organoids now formed more—and more complex—branched, bud-like structures that resembled the small sacs where milk is produced. In this setting, the balance between luminal and basal cells was largely preserved, but the number of branches and buds increased substantially compared with untreated controls. Gene activity patterns reflected this shift: many genes linked to tissue shaping and branching were turned up, while many involved in metabolism were turned down. Protein-level measurements echoed these findings, highlighting changes in components of the extracellular matrix and in metabolic pathways, especially amino acid and energy processing.
What this means for real breasts
Taken together, the work shows that mTOR is not a simple on–off growth switch but a time-sensitive coordinator of both cell identity and tissue architecture in the mammary gland. Early on, active mTOR is required for cells to grow and to maintain the flexibility to become either inner luminal or outer basal cells. Later, changing mTOR activity reshapes how cells interact with their surroundings to either blunt or enhance the formation of branched milk-producing structures. For a layperson, the take-home message is that the same molecular pathway can have very different jobs depending on when it acts—first helping build the right kinds of cells, then helping sculpt them into the right three-dimensional form. This nuanced view will be important for understanding normal breast development, the impact of drugs that target mTOR, and possibly how early changes in this pathway might contribute to breast disease.
Citation: Lacouture, A., Sylla, M.S., Germain, L. et al. The mTOR signaling pathway regulates key steps of mammary gland organoid genesis in a temporal manner. Sci Rep 16, 6751 (2026). https://doi.org/10.1038/s41598-026-37825-z
Keywords: mammary gland development, mTOR signaling, organoids, branching morphogenesis, breast biology