Clear Sky Science · en
Preliminary assessment of biodistribution and targeting of the fluorescent molecular probe Cy7-SYL3C in an EpCAM-positive colorectal cancer mouse model
Why this matters for cancer detection
Colorectal cancer is one of the world’s biggest cancer killers, in part because many tumors are found late. Today’s main screening tool, colonoscopy, is effective but invasive and uncomfortable, so many people avoid it. This study explores a new kind of glowing “smart tracer” that could one day help doctors see bowel tumors earlier and more clearly, using light instead of a scalpel or a long tube.
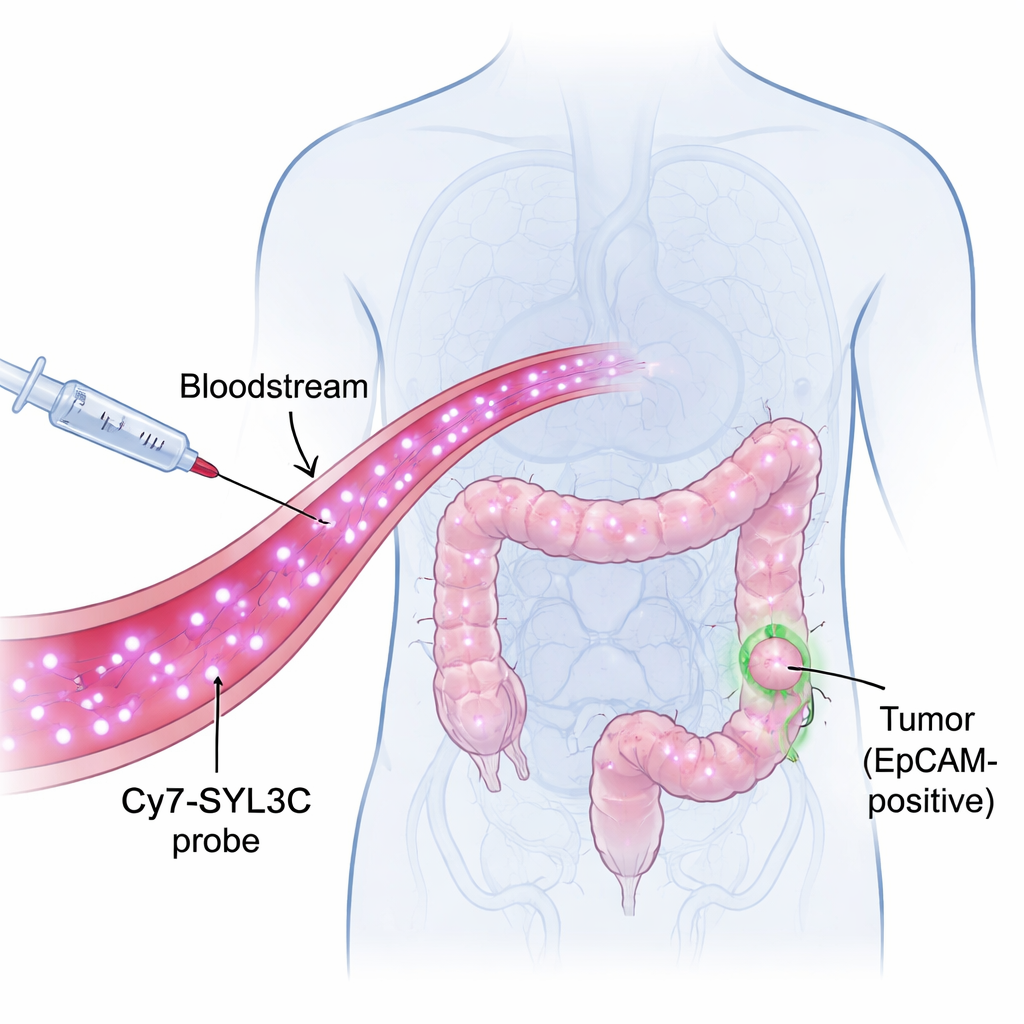
A glowing tag that recognizes tumor cells
The researchers focused on a molecule called EpCAM, which sits on the surface of many colorectal cancer cells but is scarce on healthy tissues. They built a probe named Cy7-SYL3C by attaching a near‑infrared dye (Cy7) to a short strand of DNA called an aptamer (SYL3C) that can recognize EpCAM much like a key fits a lock. Near‑infrared light is invisible to the eye but can pass several millimeters into tissue, allowing cameras to pick up signals from inside the body with high contrast. The idea is simple: inject Cy7-SYL3C into the bloodstream, let it seek out EpCAM‑rich tumor cells, then use a special camera to make those cells light up against a darker background.
Testing safety and stability in the lab
Before trying the probe in living animals, the team checked whether it was stable and safe. They mixed Cy7-SYL3C with blood serum from mice and cattle and found that most of the probe stayed intact for at least eight hours, long enough for imaging during a medical procedure. They then exposed human colorectal cancer cells and normal colon cells to different probe concentrations. Cell growth was essentially unchanged, and when the probe was mixed with red blood cells, it did not cause them to burst. These tests suggest that Cy7-SYL3C is both chemically steady and gentle on cells—key requirements for any imaging agent that might be used in patients.
Where the probe travels in the body
The scientists next followed the path of Cy7-SYL3C in healthy mice using a small‑animal imaging system. Soon after injection, the strongest signal appeared in the liver and kidneys, organs that filter and clear substances from the blood. The liver signal peaked at about one hour and then faded, while the kidneys continued to glow for longer, pointing to urine as a main exit route for the probe. After two days, only a small fraction of the original signal remained, and microscopic examination of major organs showed no obvious tissue damage. This means the probe is cleared reasonably quickly and does not visibly harm vital organs in this short‑term study.
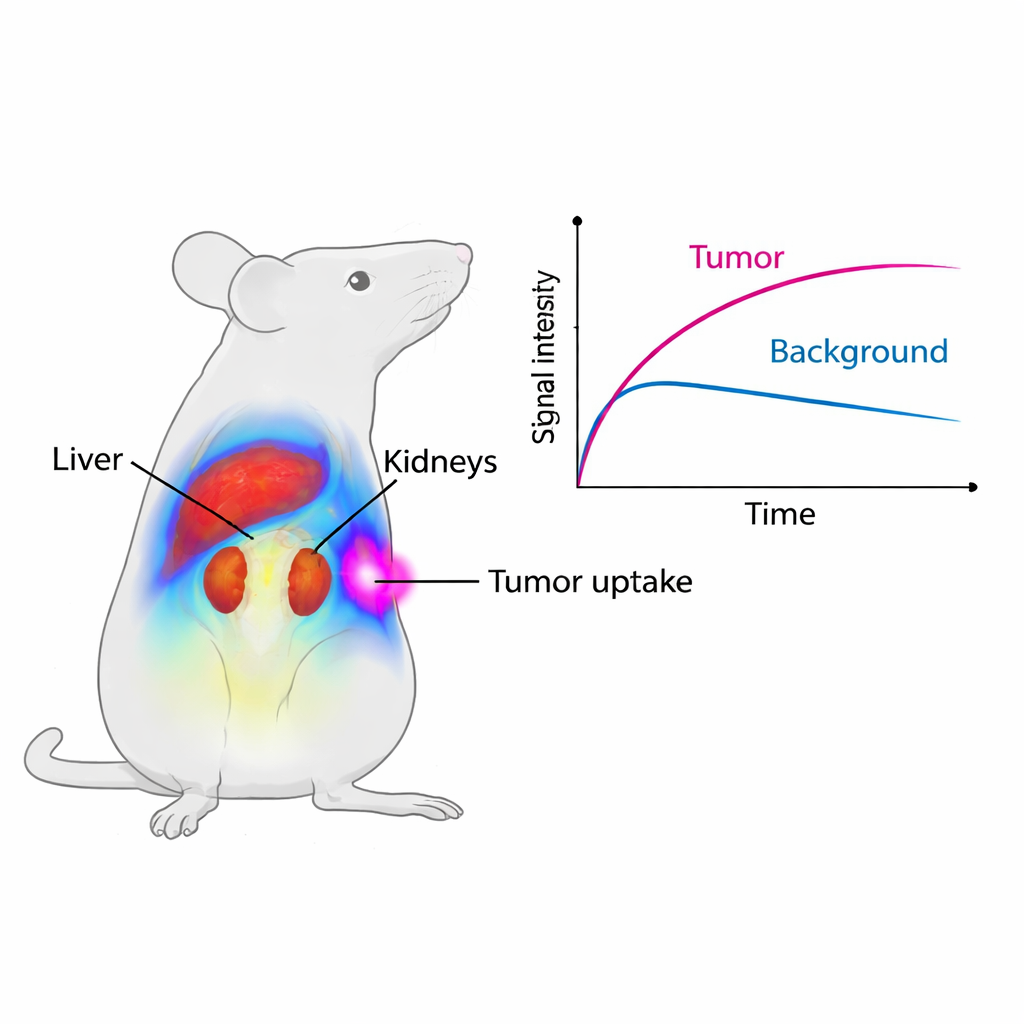
Zeroing in on colorectal tumors
To see if Cy7-SYL3C truly homes in on cancer, the team used mice implanted with human colorectal tumors that produce EpCAM. Within just five minutes of injection, the tumors began to shine on the imaging system. Over the first four hours, the average signal in tumors was nearly twice as strong as in mice where the tumors had been “pre‑blocked” with an excess of unlabeled SYL3C, which temporarily occupied EpCAM sites and prevented the glowing probe from attaching. The ratio of tumor signal to nearby muscle rose above one and peaked around eight hours, meaning tumors were clearly brighter than their surroundings for a useful imaging window. Detailed microscope studies confirmed that the fluorescent probe overlapped with EpCAM staining on tumor cells, while normal organs showed little or no EpCAM.
How this could guide future surgery and diagnosis
To a non‑specialist, the main message is that Cy7-SYL3C behaves like a smart dye that seeks out colorectal cancer cells and then largely washes out through the kidneys. It appears stable, gentle to cells, and able to make tumors glow quickly and for several hours, which could help surgeons see tumor edges more clearly in real time or help radiologists spot suspicious areas during minimally invasive procedures. Although these results are from early mouse experiments and more work is needed before use in people, the study lays important groundwork for a new class of light‑based tracers that could make colorectal cancer detection and surgery more precise and less invasive.
Citation: Li, Y., Li, M., Li, P. et al. Preliminary assessment of biodistribution and targeting of the fluorescent molecular probe Cy7-SYL3C in an EpCAM-positive colorectal cancer mouse model. Sci Rep 16, 6589 (2026). https://doi.org/10.1038/s41598-026-37787-2
Keywords: colorectal cancer imaging, near infrared fluorescence, EpCAM targeting, aptamer probe, tumor surgery guidance