Clear Sky Science · en
SIRT3 suppresses renal cancer progression by regulating IDH2 acetylation
Why this kidney cancer study matters
Kidney cancer, especially a type called renal cell carcinoma, is increasingly common and often hard to treat once it spreads. This study looks under the hood of cancer cells to see how tiny changes in their energy factories, the mitochondria, help tumors grow—and how a natural plant compound, honokiol, might help slow them down. For readers, it offers a glimpse of how understanding cell “housekeeping” could open new options beyond current drugs.
A hidden brake inside kidney cells
Our cells constantly manage energy production and harmful by-products. In healthy kidney cells, a protein called SIRT3 acts like a maintenance supervisor inside mitochondria, keeping other proteins in working order. The authors examined large public cancer databases and patient tissue samples and found that SIRT3 levels were consistently lower in kidney tumors than in nearby normal tissue. Tumors with the least SIRT3 showed gene patterns linked to faster growth and greater ability to move, suggesting that when this cellular brake is lost, cancer cells become more aggressive.
How cancer cells twist their energy balance
To test whether SIRT3 loss actually drives cancer behavior, the researchers reduced SIRT3 in kidney cancer cell lines grown in the lab. Cells with less SIRT3 multiplied faster and formed many more colonies, a sign of heightened survival and growth. When these altered cells were implanted into mice, the resulting tumors were larger and heavier than tumors from control cells. Together, these experiments show that SIRT3 is not just missing in kidney cancer—it actively restrains tumor growth when present.
A key enzyme and the problem of “rust” inside cells
The team then focused on another mitochondrial protein, IDH2, which helps produce NADPH, a molecule that fuels systems that detoxify reactive oxygen species (ROS)—damaging chemicals similar to cellular “rust.” IDH2’s behavior can be altered by a small chemical tag called an acetyl group. SIRT3 is known to remove such tags. In kidney cancer cells, SIRT3 physically bound to IDH2 and stripped off acetyl groups at a specific position (K413). When SIRT3 was reduced, this acetyl mark built up, IDH2 worked less efficiently, NADPH levels fell, and ROS levels rose. Mitochondria lost some of their membrane potential, a sign of impaired function. By using engineered IDH2 variants that mimicked either the acetylated or deacetylated state, the authors showed that the acetylated form could override SIRT3’s growth-slowing effect, tying the tumor brake directly to this single chemical switch.
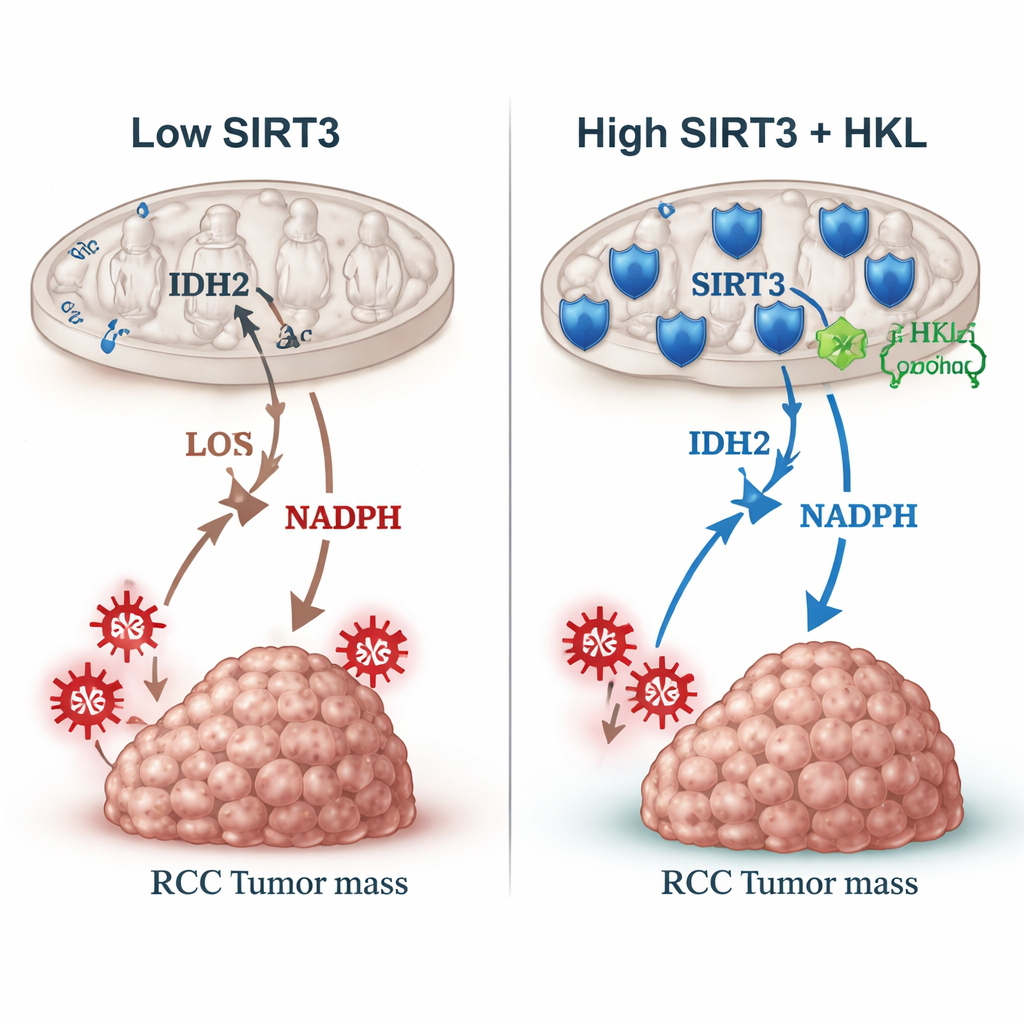
Turning the brake back on with a plant compound
Because SIRT3 seems protective, the authors asked whether it could be boosted with a drug. They tested honokiol, a natural molecule from magnolia bark already known to affect multiple cancer pathways. At a carefully chosen dose that did not simply kill cells outright, honokiol increased SIRT3 levels over time in kidney cancer cells, alongside related regulators (p-AMPK and PGC-1α). As SIRT3 rose, IDH2 lost its acetyl tag, ROS levels dropped, and cells grew and formed colonies more slowly. When honokiol was combined with sunitinib, a standard kidney cancer drug, the duo suppressed growth more than sunitinib alone, in part by blunting the excess ROS that sunitinib itself can induce. Similar effects were seen when the researchers used a classic antioxidant, supporting the idea that controlling oxidative stress can make existing treatments work better.
What this could mean for future treatments
For non-specialists, the take-home message is that this study identifies SIRT3 as a natural tumor brake in kidney cancer, working through fine control of IDH2 and the balance between protective and harmful molecules inside mitochondria. When SIRT3 is low, cells accumulate damage-promoting ROS and gain a growth advantage. Restoring SIRT3 activity—potentially with honokiol or future, more precise drugs—may slow tumor growth and help current therapies perform better. While honokiol itself still faces hurdles, including how it is absorbed and its multiple targets, the work points to a clear pathway, the SIRT3–IDH2–redox axis, that drug developers can now aim at to design smarter, metabolism-based treatments for renal cell carcinoma.
Citation: Li, L., Tian, Y., Chen, S. et al. SIRT3 suppresses renal cancer progression by regulating IDH2 acetylation. Sci Rep 16, 7619 (2026). https://doi.org/10.1038/s41598-026-37783-6
Keywords: kidney cancer, SIRT3, mitochondria, oxidative stress, honokiol