Clear Sky Science · en
Removal of carmineusing red mud-supported ZVI materials
Turning Toxic Waste into a Helpful Tool
Huge piles of reddish, powdery waste called red mud are left over when factories make aluminum. These mountains of waste take up land and can leak harmful chemicals, yet they also contain useful metals. In this study, researchers found a way to transform red mud into a powerful cleaning material that can strip stubborn red dyes from water almost completely, offering a fresh strategy for tackling both industrial waste and water pollution at the same time.
The Problem with Red Mud and Red Dyes
Aluminum is essential for cars, planes, electronics, and packaging, but every ton of aluminum oxide produced leaves behind about one to two tons of red mud. Globally, this waste has built up to billions of tons, especially in countries like China. At the same time, textile and dyeing factories discharge colored wastewater containing complex dyes such as carmine, a bright red colorant used in food, cosmetics, and fabrics. These dyes can be toxic, difficult to break down, and visually polluting, making them hard to remove with simple filters or basic treatment.
Cooking Up a New Cleaning Material
The team used red mud as a cheap iron source and mixed it with anthracite, an inexpensive form of coal, to create a new material. They formed the mixture into small pellets and heated them in a furnace at very high temperatures. This process, called carbothermal reduction, converts iron minerals in the red mud into tiny particles of “zero‑valent” iron—iron in its metallic form, which is very reactive. After heating at about 1000 °C for one hour with the right amount of anthracite, they ground the pellets into a fine powder known as RA@ZVI, ready to be tested in colored water.
How Well the New Material Cleans Water
To test performance, the researchers added small amounts of RA@ZVI to water containing carmine dye. Under optimized conditions—about 0.5 grams of material per liter of water, a starting dye level of 50 milligrams per liter, mildly warm room temperature, and an acidic pH around 3—the dye removal was close to 100% within just 30 minutes. They explored how different factors changed the results: higher amounts of RA@ZVI generally improved cleaning until the effect leveled off, lower initial dye concentrations were easier to treat, and the material worked well across a range of temperatures. However, acidic water made a big difference; in neutral or alkaline water, the removal dropped sharply because protective layers formed on the iron, blocking its cleaning action.
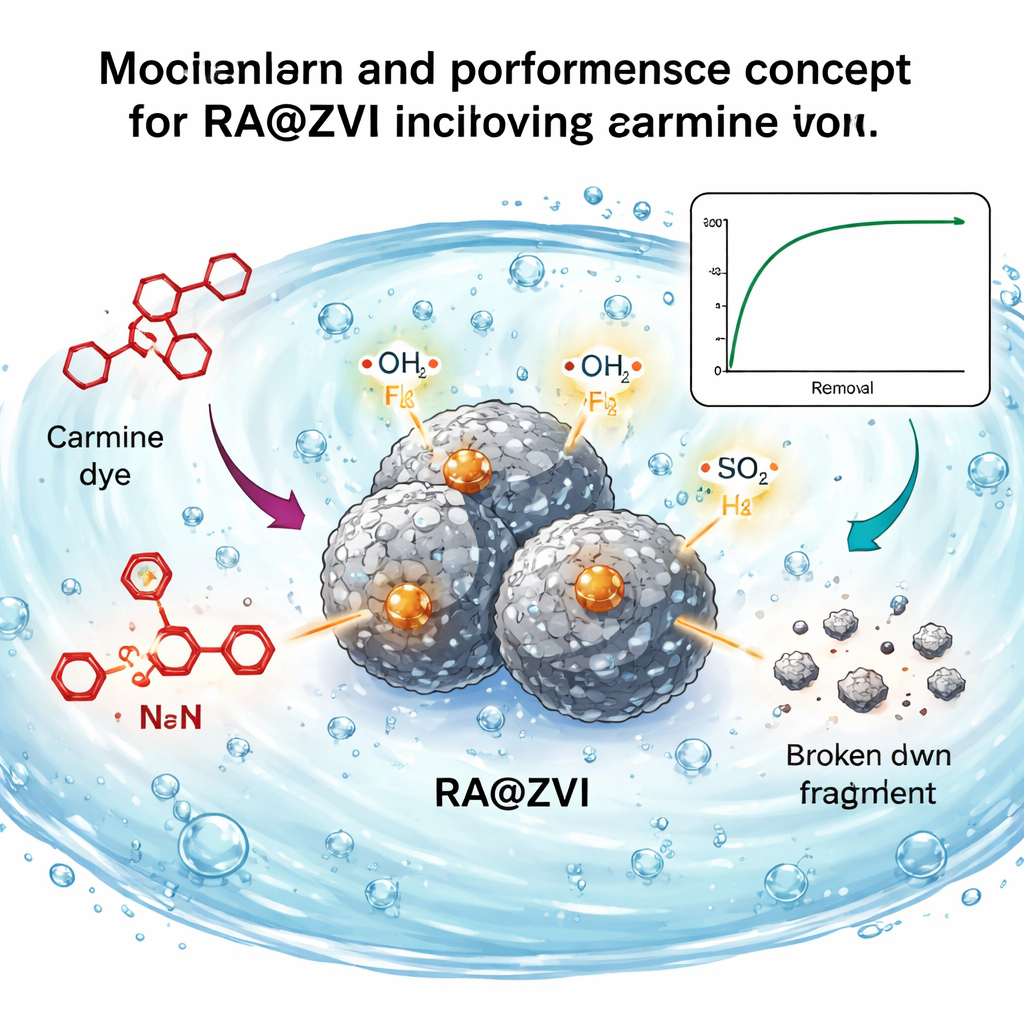
What Happens at the Tiny Scale
Using advanced microscopes and X‑ray methods, the scientists examined the structure of RA@ZVI. They observed micrometer‑sized iron particles well spread out in a porous carbon framework made from the anthracite. This structure offers many active spots for the dye to reach. After the cleaning tests, the iron particles appeared corroded and partially covered with carbon‑rich residues, showing they had taken part in chemical reactions. Spectral analysis of the dye solution revealed that RA@ZVI not only grabbed the dye molecules but actually broke apart key parts of their structure, especially the azo bond that links two rings together and the anthraquinone ring system that gives carmine its intense color.
Invisible Helpers: Short‑Lived Reactive Species
The researchers also looked at which short‑lived “helper” molecules were doing the damage to the dye. By adding special chemicals that selectively block certain reactive species, they found that two radicals—hydroxyl radicals and superoxide radicals—play central roles. The metallic iron in RA@ZVI reacts with oxygen and small amounts of peroxide formed in the water, generating these extremely reactive radicals. Working together with the iron itself, they attack the dye molecules, breaking them down into smaller, less harmful substances that eventually can become carbon dioxide and water.
Why This Matters for Everyday Life
In simple terms, this work shows how a troublesome industrial waste can be turned into a low‑cost, reusable cleaning agent for polluted water. By carefully choosing heating conditions and ingredient ratios, the team created a material that almost completely removes a difficult red dye under realistic treatment conditions, without relying on expensive or highly toxic chemicals. If scaled up, this approach could help reduce both red mud piles and the bright, persistent dyes found in printing and dyeing wastewater, moving us a step closer to cleaner factories and cleaner rivers.
Citation: Wang, Z., Tuo, B., Li, S. et al. Removal of carmineusing red mud-supported ZVI materials. Sci Rep 16, 6524 (2026). https://doi.org/10.1038/s41598-026-37767-6
Keywords: red mud, wastewater treatment, azo dye removal, zero valent iron, carmine