Clear Sky Science · en
Leveraging metabolic similarity in a 1H NMR database of medicinal plants to advance pharmacognostic insights
Why the chemistry of healing plants matters
When you drink an herbal tea or take a plant-based supplement, you are swallowing a swirling cocktail of hundreds of natural chemicals, not a single purified drug. Many traditional remedies work because of the combined action of these chemicals, yet modern science has often focused on isolating just one “active ingredient.” This study shows how a powerful analytical technique called proton nuclear magnetic resonance (1H NMR) can capture the whole chemical “fingerprint” of medicinal plants and use it to compare hundreds of herbs at once—helping to ensure quality, trace origins, and even identify home-grown substitutes for imported species.
Seeing plants as chemical neighborhoods
Instead of chasing one miracle molecule at a time, the researchers built a large-scale chemical map of 656 traditional medicinal plant samples from Asia and Europe. Using 1H NMR, they recorded broad, highly reproducible spectra that act like barcodes for each sample’s overall mix of metabolites. By treating each spectrum as a fingerprint and analyzing them with multivariate statistics, they could place every herb within a “chemical neighborhood” of related species. This macroscopic view reveals which plants share similar chemistry, which stand apart, and how environmental factors such as geography shift a plant’s profile without needing to identify every individual compound.
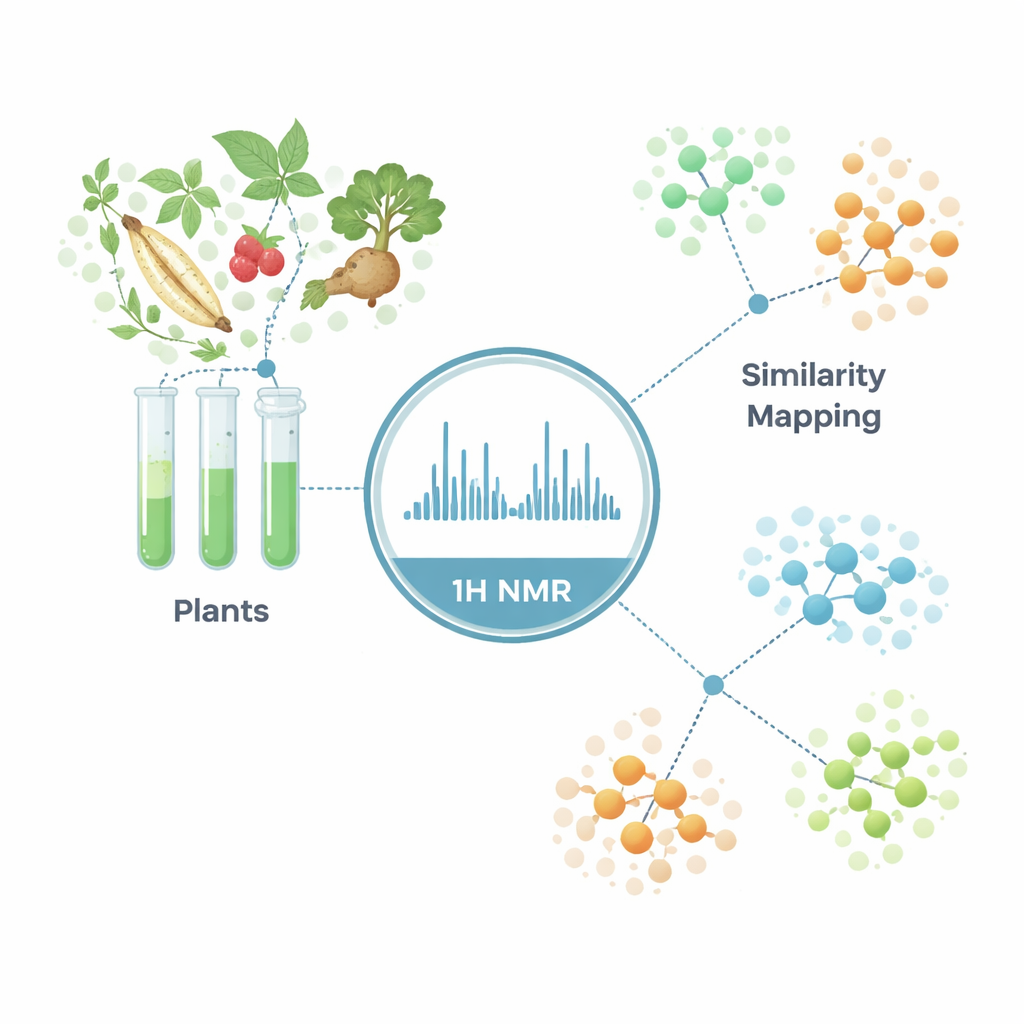
Sorting look‑alike herbs and checking their passports
The team first asked whether their database could reflect botanical family trees and support quality control. They focused on genera such as Angelica and Glycyrrhiza (the licorice group), widely used in East Asian medicine but represented by multiple species and origins. By clustering the NMR fingerprints, they showed that most samples labeled as the same genus grouped together in chemical space. Intriguingly, a plant long classified separately, Ostericum koreanum, nestled firmly inside the Angelica cluster—matching a recent taxonomic revision based on genetics. The method also picked up subtler differences: fruits of Schisandra chinensis from Korea and China were chemically similar and formed one cluster, while a commercial sample from the Netherlands sat far apart, hinting at different cultivation or processing and raising questions about consistent therapeutic effects.
Finding safe stand‑ins for scarce or imported herbs
Beyond labeling and origin checks, the database can spotlight metabolically similar plants that might stand in for one another. This is important when a traditional herb is expensive, endangered, or restricted by international rules on sharing biological resources. The researchers compared the chemical fingerprints of taxonomically distant species such as Taxus chinensis (source of the cancer drug paclitaxel) and European mistletoe (Viscum album), as well as pairs like South American cat’s claw (Uncaria tomentosa) and East Asian relatives. Despite their different histories and uses, these plants shared notable chunks of their metabolite profiles. Follow-up analysis with high-resolution mass spectrometry and molecular networking confirmed overlapping families of bioactive molecules, including compounds linked to anti-cancer, immune-modulating, and neuroprotective actions. This does not prove they are interchangeable medicines, but it provides a rational shortlist of candidates for further pharmacological testing.
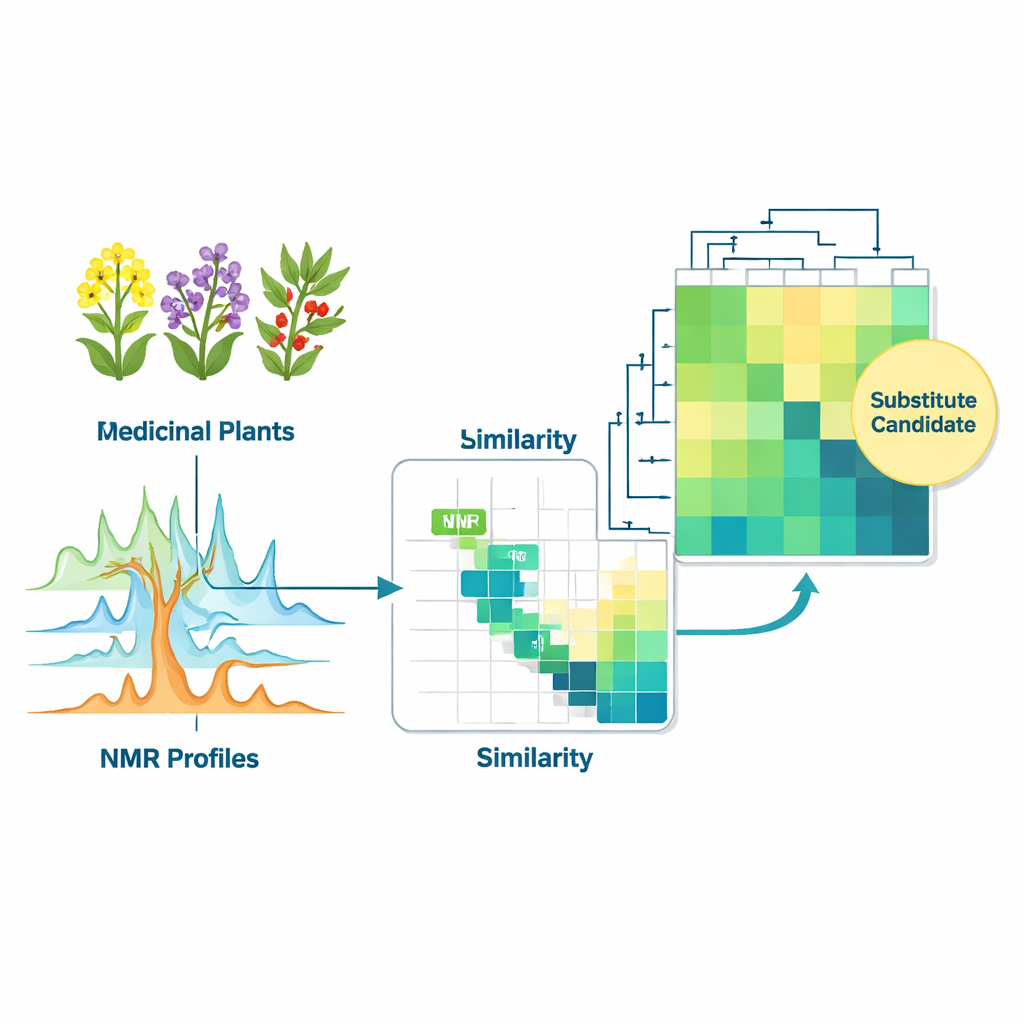
Making sense of complex herbal recipes
Traditional medicine rarely uses herbs in isolation; instead, multi-herb prescriptions are designed so that different ingredients reinforce or temper each other. The team applied NMR profiling to Huanglian Jiedu Decoction, a classic four-herb formula used for inflammatory and infectious conditions. By comparing the spectra and statistical positions of each single herb—Coptis, Phellodendron, Scutellaria, and Gardenia—with those of their mixtures, they showed that the overall mixture’s chemical profile could be approximated as a weighted combination of its parts. At the same time, NMR was sensitive enough to distinguish chemically similar ingredients such as Coptis and Phellodendron, both rich in the alkaloid berberine but carrying distinct side components. This kind of mixture-level mapping helps researchers see how each herb contributes to the ensemble effect and whether swapping one component might subtly shift the formula’s behavior.
What this means for future plant-based medicines
To a non-specialist, the key message is that herbal medicines can now be studied and managed at scale without losing their complexity. By using 1H NMR as a stable, whole-profile fingerprinting tool, scientists can cluster herbs by chemical similarity, check authenticity across time and place, and rationally propose local substitutes when imports are scarce or regulated. While chemical resemblance alone cannot guarantee the same clinical effects—follow-up biological and clinical tests are essential—this macroscopic, database-driven approach offers a powerful starting map. It brings centuries of empirical herbal practice into a framework that modern pharmacology and regulators can use to design safer, more consistent, and more sustainable plant-based therapies.
Citation: Seo, S., Erol, Ö., Kim, H. et al. Leveraging metabolic similarity in a 1H NMR database of medicinal plants to advance pharmacognostic insights. Sci Rep 16, 6691 (2026). https://doi.org/10.1038/s41598-026-37725-2
Keywords: medicinal plants, NMR metabolomics, herbal medicine, natural product drug discovery, metabolic profiling