Clear Sky Science · en
Effect of low-temperature intracanal sodium hypochlorite on root surface temperature reduction and organic tissue dissolution: an in vitro study
Cooling Down Root Canal Treatment
Anyone who has needed a root canal knows the main worry is pain during and after the procedure. Dentists already rely on a strong cleaning liquid called sodium hypochlorite to disinfect the inside of a tooth, but this solution can also irritate the tissues around the root. This study asked a simple, practical question: if we chill that cleaning liquid before using it, can we cool the tooth from the inside, potentially easing inflammation and pain, without losing its cleaning power?
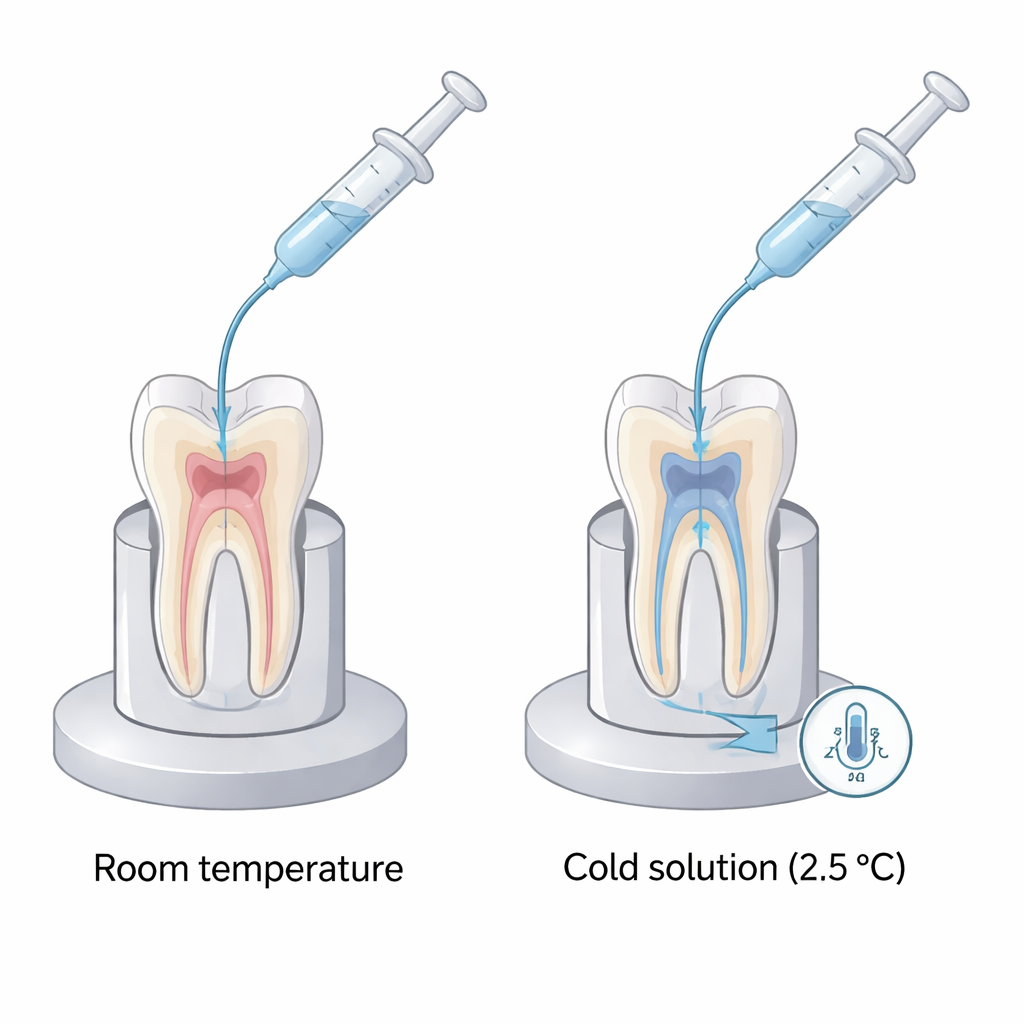
Why Dentists Care About Temperature
Modern root canal treatment depends on two partners working together: tiny instruments that scrape and shape the inner canal, and chemical solutions that wash away germs and soft, infected tissue. Sodium hypochlorite is the workhorse solution because it kills a wide range of microbes and dissolves organic material. But stronger or mishandled solutions can harm the delicate tissues at the tip of the root. At the same time, a trend called intracanal cryotherapy—using chilled fluids inside the canal—has emerged because cooling tissues can reduce blood flow, slow down inflammatory reactions, and lessen pain. What has not been clear is whether using this strong cleaner at low temperature changes how well it works or how much it cools the root surface.
How the Experiments Were Set Up
The researchers used two laboratory models to tease apart temperature effects from cleaning effects. First, they took extracted human premolar teeth, prepared their root canals as in real treatment, and attached tiny temperature sensors to the outside of the root near the tip. Each tooth was then irrigated twice, in separate sessions, using a negative-pressure system that draws liquid safely down to the end of the canal. One session used sodium hypochlorite at room temperature, and the other used the same solution chilled to about 2.5 °C. In the second model, they switched to 3D-printed plastic teeth fitted with glass capillaries packed with standardized strands of catgut, which mimic organic tissue. By weighing these capillaries before and after irrigation with warm or cold solution, they could precisely measure how much “tissue” was dissolved.
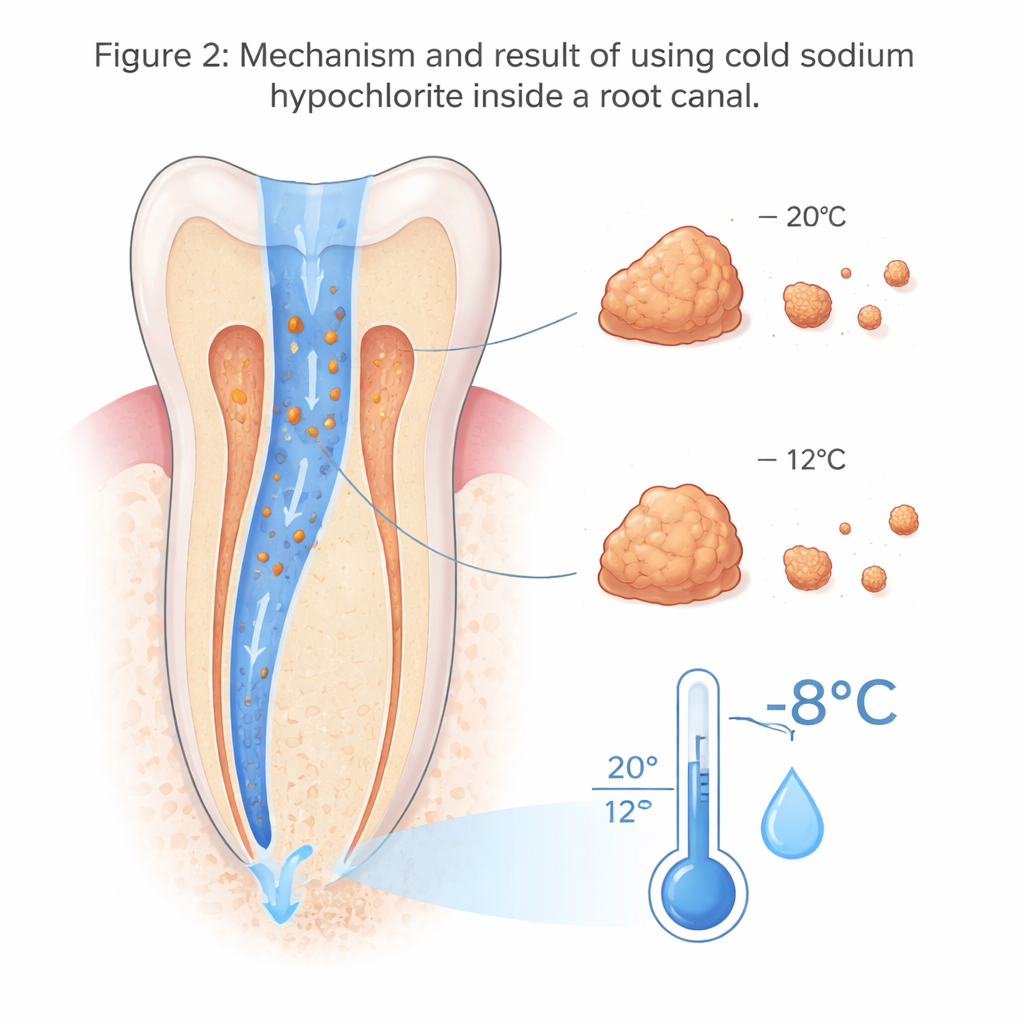
What Cooling the Solution Actually Did
On the real teeth, room-temperature irrigation produced only a small drop of about 0.9 °C at the root surface. In contrast, the cold solution produced a much more pronounced cooling of roughly 8.3 °C, a statistically significant change that suggests chilled fluid can noticeably lower the temperature at the root tip area for several minutes. Importantly, the starting temperature of the teeth was similar in both cases, so the difference came from the solution temperature itself. On the 3D-printed teeth, however, the amount of organic material dissolved was nearly the same whether the liquid was cold or at room temperature. Within the sensitivity of their method, chilling sodium hypochlorite did not blunt its ability to break down soft tissue.
Strengths, Limits, and Real-World Meaning
By combining natural teeth for realistic heat behavior with standardized printed teeth for precise weighing, the study offers a clearer picture of what cooling actually achieves. Still, it has limits: all work was done outside the body, where there is no blood circulation or natural temperature control, and only one irrigation system was tested. The amount of organic material was also relatively small, which might hide subtle differences in cleaning between temperatures. Even so, the findings line up with clinical reports that cold irrigation can lessen post-treatment discomfort, and they show that meaningful cooling is possible without clearly sacrificing cleaning performance under these conditions.
A Colder, Gentler Root Canal?
For patients, the takeaway is straightforward: using chilled sodium hypochlorite inside the tooth appears to cool the root tip significantly while still dissolving soft tissue about as well as the usual room-temperature solution. That combination—better cooling with unchanged cleaning—means cold irrigants could become a simple, low-cost way to make root canal therapy kinder to the tissues around the tooth, and potentially less painful afterward, without compromising the success of the treatment.
Citation: Iparraguirre Nuñovero, M.F., Hungaro Duarte, M.A., Xavier da Silva Neto, U. et al. Effect of low-temperature intracanal sodium hypochlorite on root surface temperature reduction and organic tissue dissolution: an in vitro study. Sci Rep 16, 6849 (2026). https://doi.org/10.1038/s41598-026-37704-7
Keywords: root canal, intracanal cryotherapy, sodium hypochlorite, postoperative pain, endodontic irrigation