Clear Sky Science · en
High fat diet remodels the gene regulatory networks in the preoptic area
Why your brain matters in diabetes
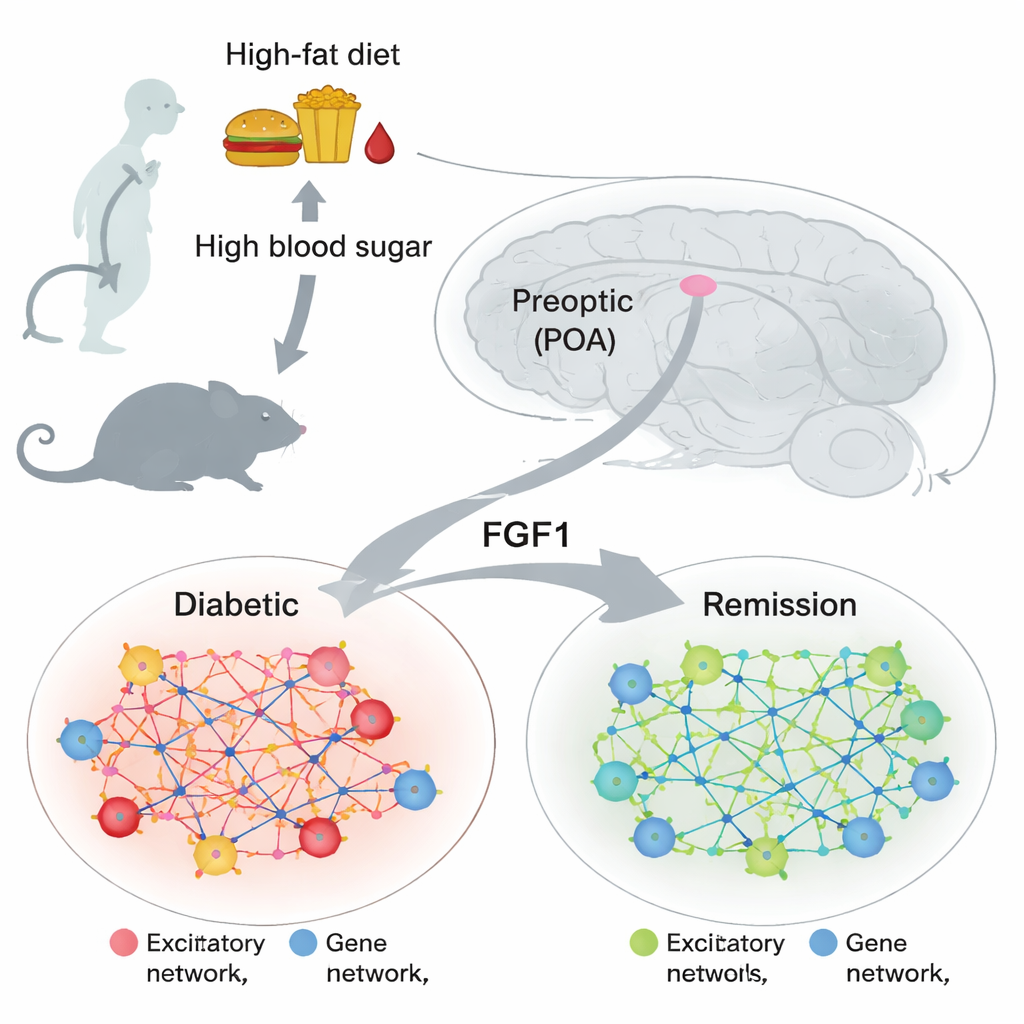
Most people think of diabetes as a problem of the pancreas and blood sugar, but your brain is deeply involved too. This study explores how a high fat diet and a powerful experimental treatment change gene activity in a small but important brain region called the preoptic area. By mapping which brain cells "reset" when diabetes goes into remission, the researchers hope to point the way to new therapies that tap into the brain’s own ability to help control blood sugar.
A small brain hub with big metabolic influence
The preoptic area sits at the front of the hypothalamus, a part of the brain that helps regulate body temperature, sleep, feeding, and hormone signals. Earlier work showed that cells in this region can influence how the body handles glucose and responds to warmth, but their role in diabetes was poorly understood. At the same time, other studies found that a single dose of a protein called fibroblast growth factor 1 (FGF1), given into the brain, can send obese diabetic mice into long-lasting remission. This raised a key question: which brain cells and gene programs change when blood sugar normalizes, and do similar changes appear in the preoptic area?
Reading gene activity cell by cell
To answer this, the team turned to single-cell RNA sequencing, a technique that measures which genes are switched on in thousands of individual cells at once. They re-analyzed an existing dataset from mice whose diabetes had gone into remission after FGF1 treatment, focusing on nerve cells in the hypothalamus. Using statistical tools, they identified groups of genes that rose or fell together as animals moved from a diabetic state to remission. These “remission modules” act like fingerprints of recovery: instead of tracking one gene at a time, they capture coordinated shifts across many genes within particular types of neurons.
High fat diet, brain cells, and dialed-down energy use
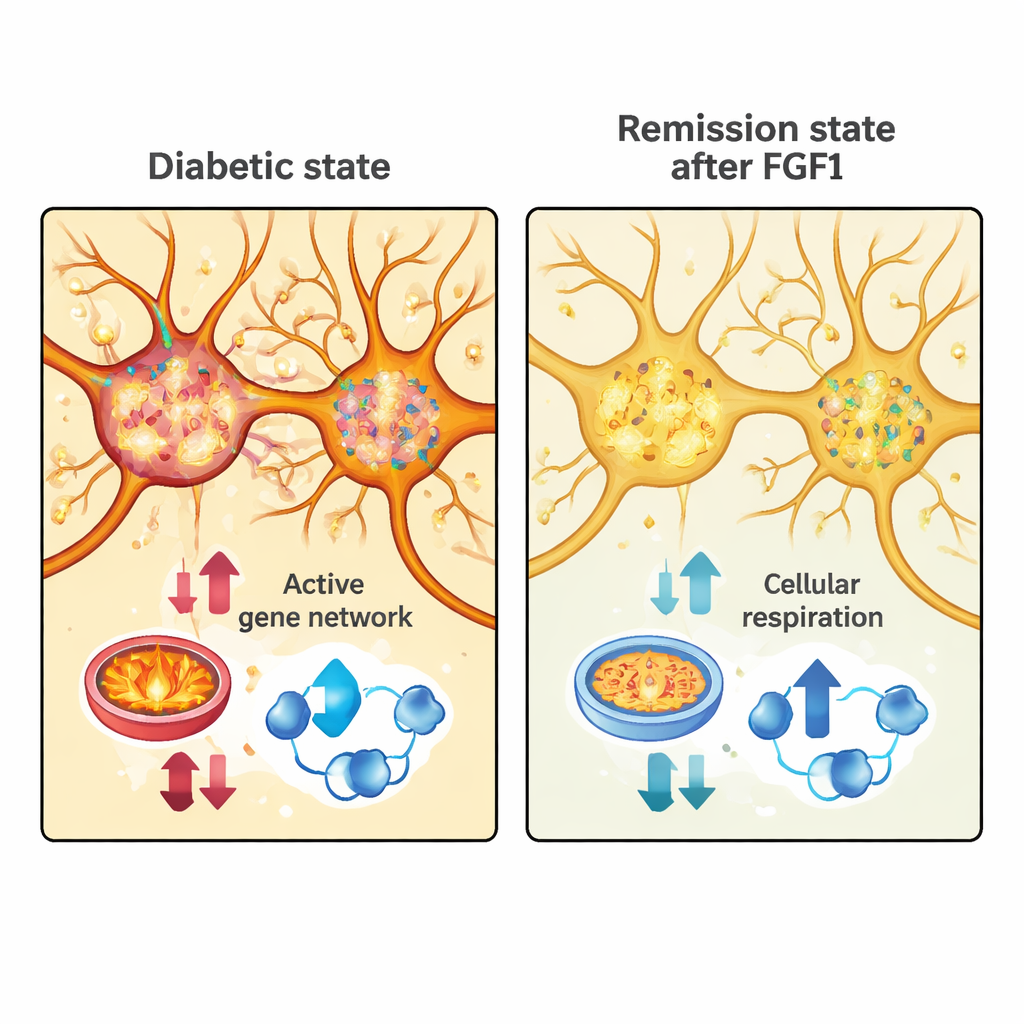
The researchers then compared these remission modules to gene activity in another dataset that sampled cells from the preoptic area of normal mice. They first separated neurons into two broad classes: excitatory cells, which tend to increase activity in their circuits, and inhibitory cells, which dampen it. In diabetic mice treated with FGF1, both types of neurons showed broad reductions in genes linked to energy production—especially those involved in oxidative phosphorylation, cellular respiration, and the mitochondrial machinery that fuels active cells. In excitatory neurons, a large “turquoise” gene module tied to energy use and synaptic activity was strongly turned down in remission, hinting that these cells become less metabolically demanding when blood sugar is brought under control.
Tracing remission signatures to specific neuron groups
Next, the team asked whether these remission-related gene patterns lined up with particular cell populations in the preoptic area. By computationally merging the two datasets, they found that some preoptic neuron clusters expressed many of the same genes that changed with FGF1-induced remission. They highlighted several genes—such as Trpc4, Dgkg, and Ryr3—that sat at the crossroads of these analyses. Using a highly sensitive microscopy method called RNAscope, they showed that these genes are indeed expressed in distinct zones of the mouse preoptic area, mostly in dorsal subregions. This validates them as real markers of specific neuron groups that may participate in the brain’s response to metabolic disease.
What this means for future diabetes treatments
For non-specialists, the upshot is that diabetes is not just a blood sugar problem—it is also a brain network problem. The study shows that when diabetes goes into remission in mice, certain preoptic neurons shift into a lower-energy state and change their gene activity in coordinated ways. By defining these remission modules and linking them to concrete cell types and marker genes, the work provides a roadmap for future experiments: researchers can now target these specific neurons to test how they influence glucose control, obesity, and related complications. In the long run, better understanding of these brain circuits could help inspire treatments that complement or even bypass failing organs in the body by harnessing the brain’s built-in control over metabolism.
Citation: Lazaro, O., Beimfohr, C., App, B. et al. High fat diet remodels the gene regulatory networks in the preoptic area. Sci Rep 16, 7042 (2026). https://doi.org/10.1038/s41598-026-37692-8
Keywords: brain and diabetes, preoptic area, high fat diet, gene networks, FGF1 remission