Clear Sky Science · en
Molten salt-assisted one-pot synthesis of Ag nanoparticles supported on clay minerals for enhanced antibacterial performance
Why tiny silver and everyday clay matter
As antibiotic-resistant bacteria become harder to kill, scientists are racing to find new ways to stop infections. This study explores an unexpected duo—silver and common clay minerals—as a simple, low-cost weapon against dangerous germs. By cleverly heating silver salts with natural clays, the researchers created materials that can steadily release germ-killing silver in a controlled way, potentially useful for medical dressings, water purification, and other everyday protections.
Turning kitchen-shelf powders into germ fighters
The heart of this work is a straightforward "one-pot" recipe. The team mixed two types of clay minerals—montmorillonite, which looks like stacked sheets, and palygorskite, which forms tiny rods—with a silver salt and ordinary sodium nitrate. After grinding them together, they heated the mixture so the silver salt broke down on its own into metallic silver without any extra chemicals. Sodium nitrate melted and released charged particles that helped keep the newborn silver particles from clumping together. When the mixture cooled, the result was clay grains whose surfaces were evenly speckled with ultra-small silver particles. 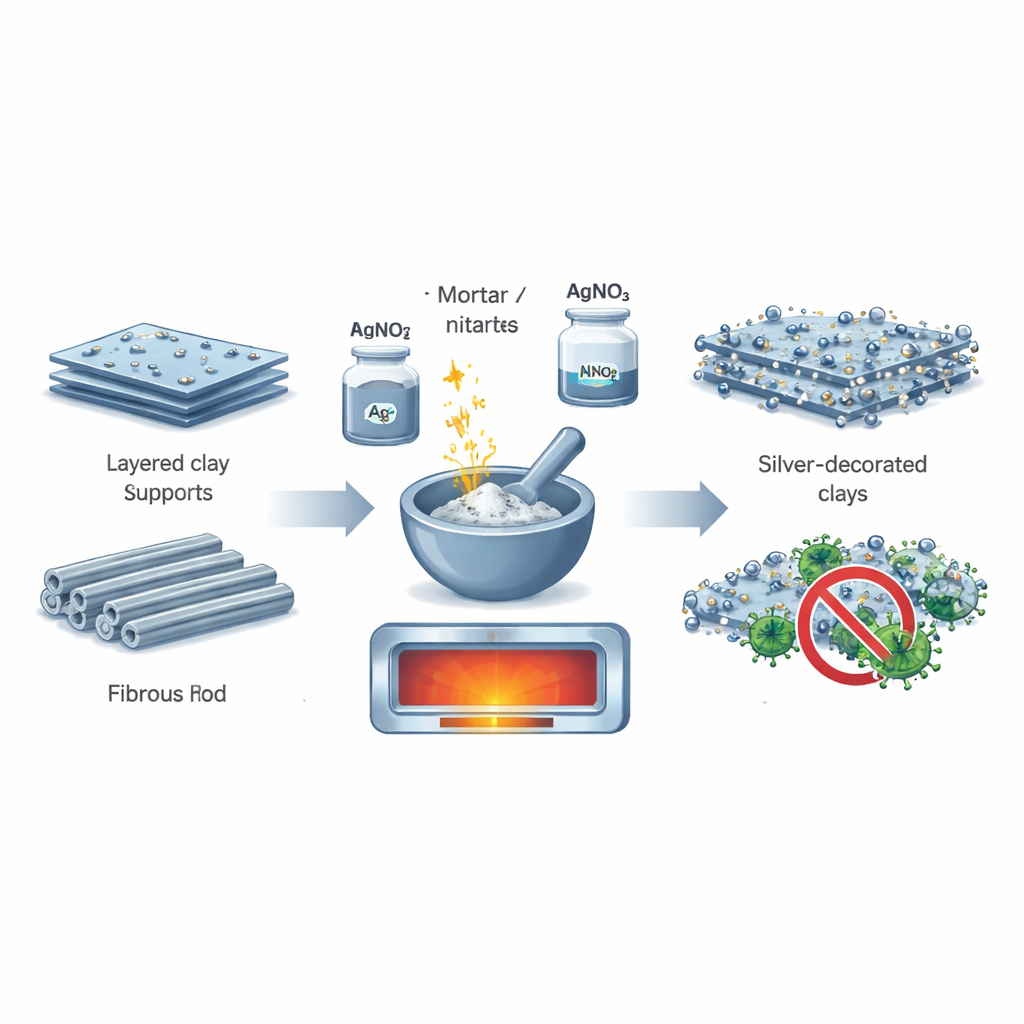
How clay shapes the silver specks
Although both clays started as simple natural minerals, their different shapes and surfaces turned out to matter a great deal. On the layered montmorillonite, silver nanoparticles averaged around 11 billionths of a meter in diameter. On the fibrous palygorskite, however, the particles were almost half that size, about 6 billionths of a meter. The rod-like palygorskite has more surface area and many negatively charged sites that attract silver ions and hold them in place as they turn into metal. This allowed more, smaller silver particles to spread evenly across the clay, preventing large clumps that would otherwise weaken the antibacterial effect.
Slow, steady silver for stronger protection
These silver-decorated clays do not simply act as coatings; they work as tiny silver reservoirs. When immersed in water, they gradually release silver ions, which are the main killers of bacteria. The palygorskite-based material released silver about eight times more efficiently than the montmorillonite version over twelve hours. At first, there was a quick burst as silver near the outer surface dissolved, followed by a slower, sustained release from deeper within the clay. This controlled trickle of silver is important: too little has no effect, while a sudden high dose can be toxic to healthy cells. The structure of palygorskite hit a better balance, offering a steady supply of active silver in contact with microbes. 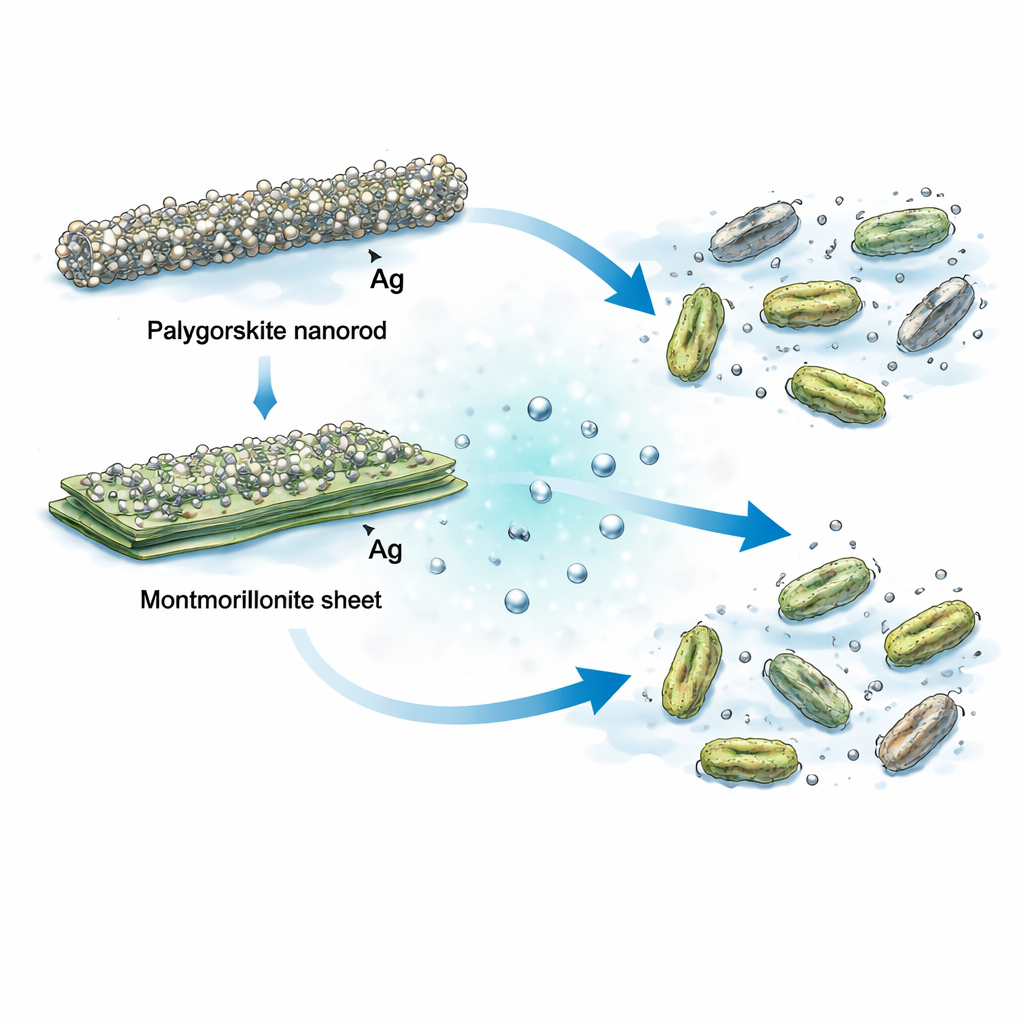
Putting the new materials to the test
To see whether this design really matters in practice, the researchers tested the materials against two common and medically important bacteria: Escherichia coli, which has a thin, flexible outer wall, and Staphylococcus aureus, which has a thicker, tougher one. In liquid cultures, both silver–clay materials slowed down or stopped bacterial growth, but the palygorskite version was clearly stronger. It reached almost complete killing of E. coli at a lower dose and showed superior performance against S. aureus as well. The smaller silver particles on palygorskite and its faster silver-ion release allowed more silver to reach and penetrate the cells. In addition, the rigid clay rods themselves can scrape and weaken bacterial surfaces, opening extra doorways for silver to get inside.
What this means for future antibacterial tools
For non-specialists, the takeaway is that pairing a familiar metal with simple natural clays can turn cheap, abundant materials into sophisticated germ-fighting tools. The study shows that not all clays are equal: palygorskite, with its rod-like structure, produces smaller silver particles, releases silver more effectively, and kills bacteria more efficiently than its layered cousin. Because the process uses no harsh reducing chemicals and relies on easily scaled heating and mixing, it could be adapted for coatings, filters, wound dressings, and other applications where long-lasting, low-cost antibacterial protection is needed.
Citation: Wang, Q., He, Q., Huang, G. et al. Molten salt-assisted one-pot synthesis of Ag nanoparticles supported on clay minerals for enhanced antibacterial performance. Sci Rep 16, 6717 (2026). https://doi.org/10.1038/s41598-026-37682-w
Keywords: silver nanoparticles, antibacterial materials, clay minerals, drug-resistant bacteria, nanocomposites