Clear Sky Science · en
Type I interferon signaling defines a novel disease signature in xeroderma pigmentosum C human keratinocytes
Why sunlight can be so dangerous for some people
For most of us, sunlight means warmth and vitamin D. But for people with a rare genetic condition called xeroderma pigmentosum C (XP-C), even modest daylight can cause severe skin damage and an extreme risk of skin cancer. This study looks beyond the well-known DNA repair problem in XP-C and uncovers a hidden layer of overactive immune‑like signaling inside their skin cells, offering new clues to why their skin is so vulnerable and inflamed after sun exposure.
When DNA repair fails in the skin
Our skin cells constantly repair DNA damage caused by ultraviolet B (UVB) rays from the sun. A protein called XPC is one of the first "sensors" that spots UV‑induced lesions and launches a large repair team. In XP-C, mutations in the XPC gene cripple this early warning step, so UV damage piles up instead of being fixed. Patients with XP-C develop skin cancers thousands of times more often than the general population and must avoid sunlight rigorously. While this DNA repair defect is well known, how it rewires the cells’ internal communication systems—especially those controlling inflammation—has been much less clear.
Probing cell signals over time
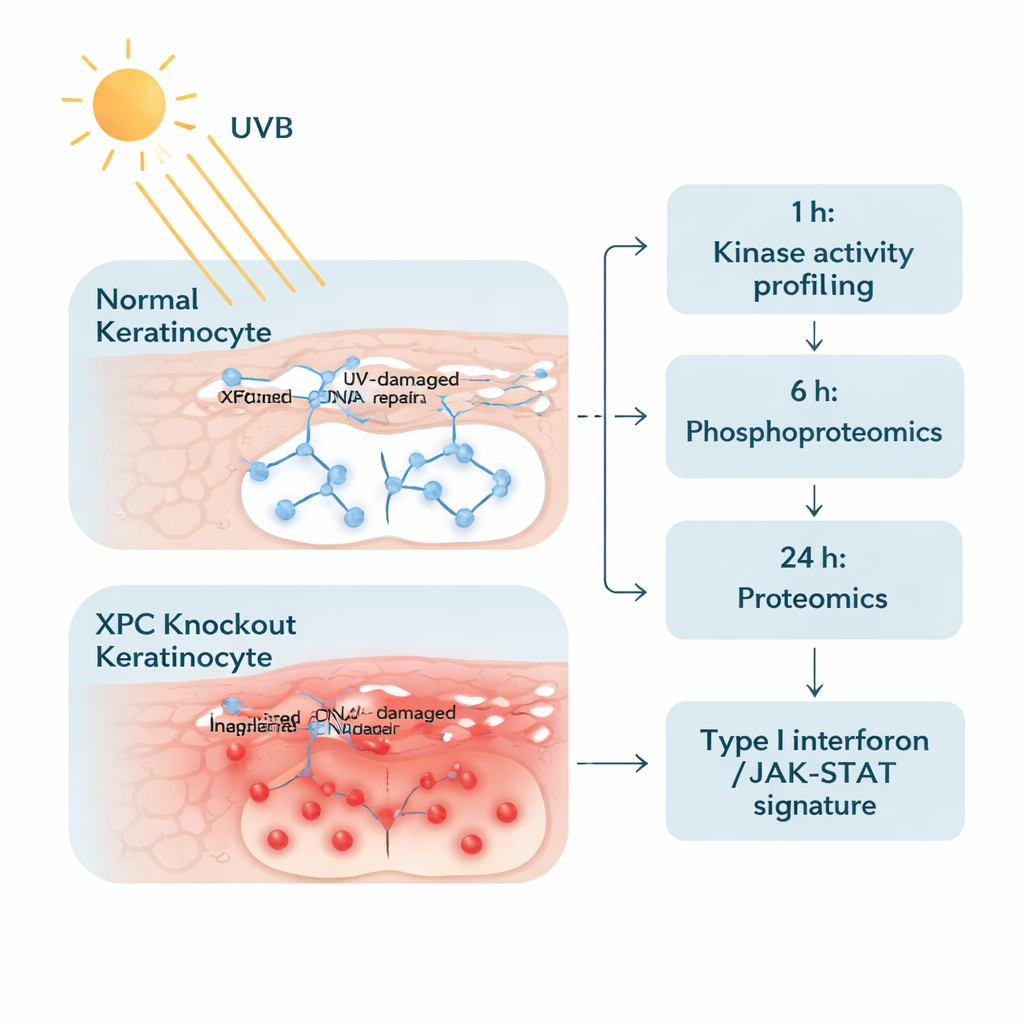
To tackle this problem, the researchers engineered human keratinocytes—the main cells in the outer skin layer—to completely lack XPC, and compared them to otherwise identical normal cells. They exposed both cell types to a carefully chosen, real‑world level of UVB similar to a moderate sunburn dose. Then they examined the cells in two stages. One hour after irradiation, they measured the activity of many enzymes called protein tyrosine kinases, which switch signals on and off by adding small phosphate tags. Twenty‑four hours later, they used advanced mass spectrometry to take a broad snapshot of thousands of proteins and see which ones rose or fell in abundance. This two‑step approach let them track both the early “alarm bells” and later “downstream responses” within the cells.
An inflammatory pathway lights up
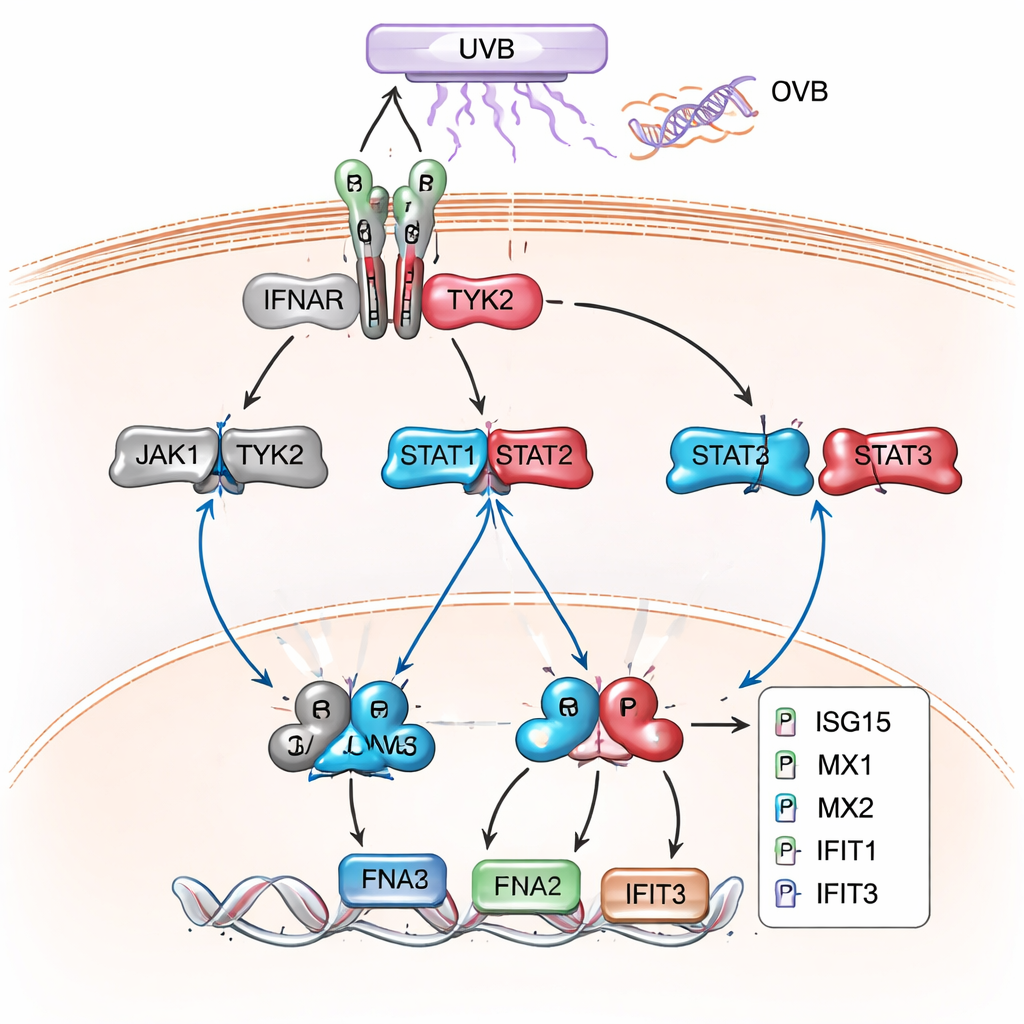
The early kinase screen revealed that XPC‑deficient keratinocytes respond to UVB with a surge of phosphorylation on more than a hundred sites compared with normal cells. Notably, many of these changes clustered in the JAK/STAT signaling pathway—a central communication route usually used by immune messengers such as interferons. Markers linked to JAK1, JAK2, JAK3, TYK2, and STAT proteins were more active in XP-C cells, even before UVB, and were further boosted after irradiation. This suggested that the cells were primed in a “high‑alert” state, ready to amplify inflammatory messages whenever they encountered UV‑induced stress.
Interferon‑style alarm genes in skin cells
The later, broader protein survey confirmed and extended this picture. In XPC knockout cells, especially after UVB, hundreds of proteins were altered, and a large cluster corresponded to genes normally switched on by type I interferons—the same antiviral signals our bodies use to fight infections. Proteins such as MX1, MX2, IFIT1, IFIT2, IFIT3, ISG15, OAS1, and IRF9, classic “interferon‑stimulated genes,” were strongly elevated. Network and pathway analyses linked these proteins back to JAK/STAT and type I interferon signaling as the dominant theme. Follow‑up western blot experiments confirmed that key STAT proteins were more heavily phosphorylated, and that these interferon‑responsive proteins were produced at far higher levels in XP-C cells than in normal keratinocytes, both at rest and especially after UVB.
What this means for patients and future treatments
Together, these results show that XP-C is not only a disease of faulty DNA repair; it is also marked by a persistent, interferon‑like inflammatory state inside skin cells, driven through the JAK/STAT pathway and intensified by UV light. For a layperson, this means that XP-C keratinocytes behave as if they are constantly fighting an invisible infection whenever they see sunlight, layering chronic inflammation on top of unrepaired DNA damage. While the study does not test treatments directly, it raises the possibility that carefully tuned drugs targeting JAK/STAT or related inflammatory circuits might one day help reduce UV‑triggered damage in XP-C and perhaps in other inflammatory skin conditions that share this molecular signature.
Citation: Nasrallah, A., Rezvani, HR., Belmudes, L. et al. Type I interferon signaling defines a novel disease signature in xeroderma pigmentosum C human keratinocytes. Sci Rep 16, 6559 (2026). https://doi.org/10.1038/s41598-026-37662-0
Keywords: xeroderma pigmentosum, skin cancer, DNA repair, interferon signaling, JAK STAT pathway