Clear Sky Science · en
Nonlinear fractional stochastic delay modeling and computational analysis of herpes simplex virus type II dynamics
Why this virus still matters
Genital herpes, most often caused by herpes simplex virus type II (HSV‑II), affects hundreds of millions of people worldwide. Once acquired, the virus usually stays in the body for life, flaring up unpredictably and quietly spreading even when no sores are visible. This paper does not describe a new drug or vaccine; instead, it uses advanced mathematics and computer simulations to better understand how HSV‑II spreads through a population and how different control strategies might keep it in check.
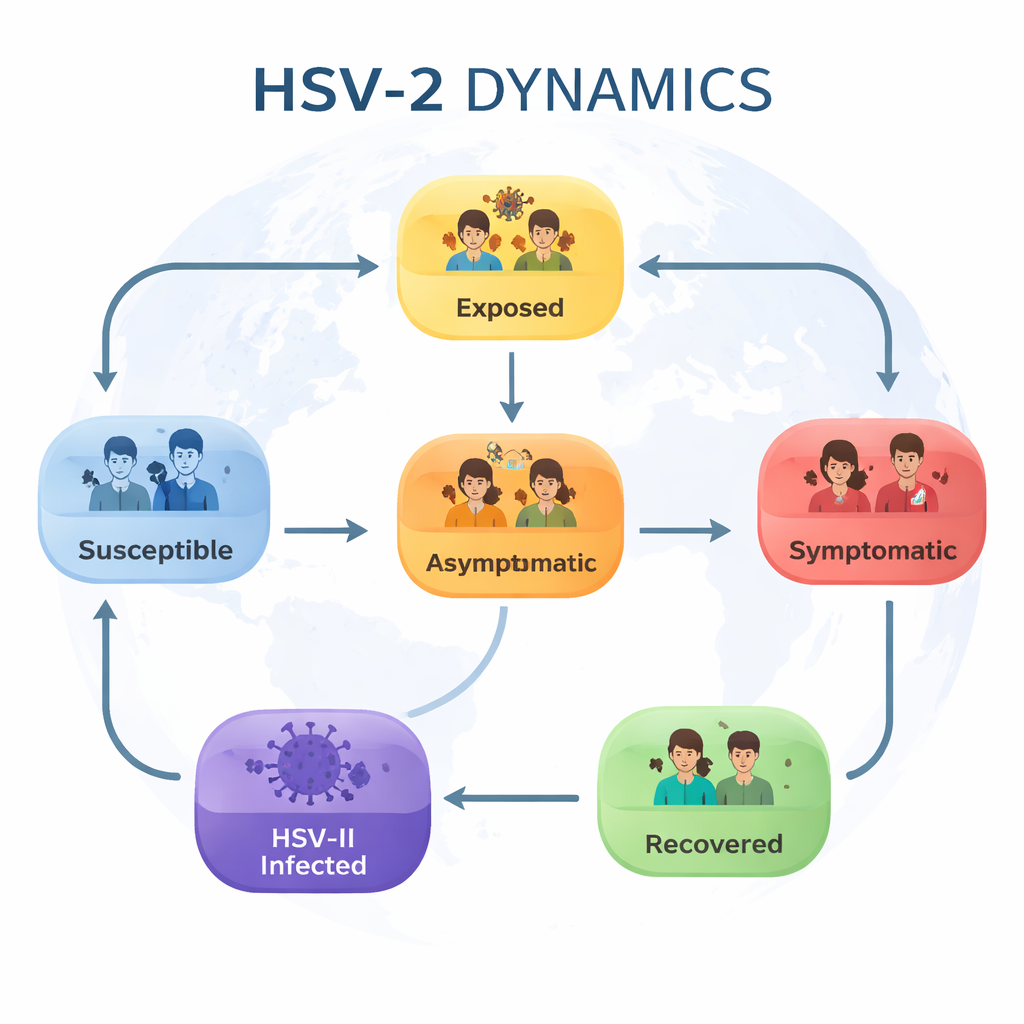
Breaking the outbreak into simple groups
The authors begin by dividing the population into six groups that capture the main stages of HSV‑II infection: people who are still vulnerable (susceptible), those recently infected but not yet infectious (exposed), people who carry the virus without symptoms (asymptomatic), those with obvious symptoms like genital sores (symptomatic), individuals with established HSV‑II infection, and people who have recovered for a time. They then describe how people move between these groups: becoming infected through contact, progressing from silent to symptomatic infection, receiving treatment, recovering, or losing temporary protection and becoming vulnerable again.
Adding memory, delay, and randomness
Real infections do not follow neat, clockwork rules, and the model reflects that. First, it includes a built‑in “memory” effect: past infection history influences current behavior and immunity, so the system does not forget what happened a moment ago. Second, there is an explicit time lag between infection and becoming contagious, mirroring incubation periods and delayed immune responses. Third, the model allows for random fluctuations, such as chance contacts or variable immune strength, by adding carefully designed noise terms. Together, these features create a richer and more realistic picture of how HSV‑II behaves over months and years, rather than assuming a perfectly smooth epidemic curve.
A single number that signals danger
A central outcome of the analysis is the basic reproduction number, often written as R�0. This single number summarizes, on average, how many new infections one contagious person causes in a mostly uninfected population. If R�0 is below 1, outbreaks tend to fade; if it is above 1, the infection can settle in and persist. The authors show that their HSV‑II system has two key steady states: one where the virus disappears and another where it remains a long‑term presence. By examining how R�0 depends on contact rates, the fraction of cases that are silent, recovery speeds, and death or removal rates, they identify which levers matter most for tipping the system from persistence toward elimination.
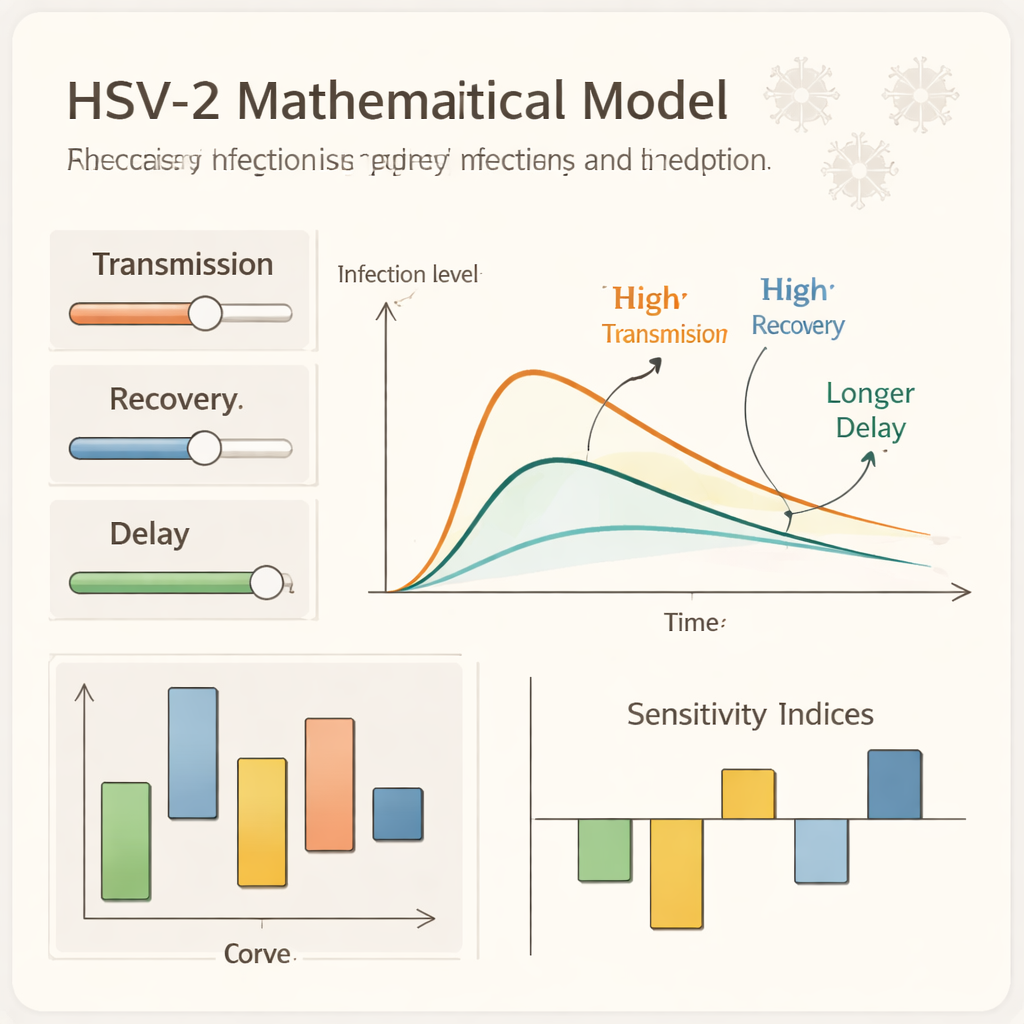
Testing the model on the computer
To explore these ideas numerically, the team builds a special kind of computer scheme that mimics the underlying biology even when using relatively large time steps. This nonstandard approach guarantees that simulated population sizes never become negative and remain within realistic bounds. Running the model under many settings, they track how the numbers of susceptible, exposed, asymptomatic, symptomatic, infected, and recovered people change over time. They find, for example, that stronger memory effects can prolong low‑level, smoldering infection, while longer delays shift and stretch epidemic peaks. Their sensitivity calculations show that increasing transmission‑related parameters drives R�0 up, whereas speeding up recovery or removal pushes R�0 down, highlighting where prevention and treatment efforts are most effective.
What this means for everyday health
For non‑specialists, the message of this work is that the future of HSV‑II in a community is not random fate. By carefully modeling how people move through different stages of infection, and by accounting for delays, lingering immune effects, and chance events, the authors build a tool that can test “what‑if” scenarios before trying them in the real world. Their results suggest that reducing opportunities for transmission and improving treatment and recovery can jointly drive the virus toward extinction in a population. While this study does not offer an immediate cure, it provides a robust framework for designing smarter public health strategies, evaluating potential vaccines, and ultimately reducing the long‑term burden of genital herpes.
Citation: Raza, A., Alsulami, M., Lampart, M. et al. Nonlinear fractional stochastic delay modeling and computational analysis of herpes simplex virus type II dynamics. Sci Rep 16, 7009 (2026). https://doi.org/10.1038/s41598-026-37658-w
Keywords: genital herpes, HSV-2 transmission, mathematical modeling, stochastic dynamics, fractional-order delay