Clear Sky Science · en
Wnt-3a exacerbates production of TNF-α in LPS stimulated microglia independent of the β-catenin canonical pathway
Why brain inflammation matters
Many brain disorders, including Parkinson’s disease, are now known to involve chronic inflammation. In this process, the brain’s resident immune cells, called microglia, can switch from protective helpers into overactive attackers that harm nearby neurons. This study asks a timely question: can a signaling molecule called Wnt‑3a, long thought to have calming, protective effects, actually make inflammation worse under certain conditions? The answer turns out to be more complicated than expected, and it highlights why the same molecule can be helpful in one context and harmful in another.
A messenger with two faces
Wnt proteins are a family of chemical messengers that help guide brain development, maintain brain cells in adulthood, and shape how cells respond to injury. One of the best studied members, Wnt‑3a, is usually associated with a “canonical” signaling route that stabilizes a protein called β‑catenin inside cells. In many systems, turning on this route seems to dampen inflammation and support neuron survival, so some researchers have wondered whether boosting Wnt‑3a might help protect vulnerable dopamine‑producing neurons in Parkinson’s disease. However, Wnt proteins can also signal through alternative, “non‑canonical” routes that may instead drive inflammation, and which pathway dominates can depend on the cell type and its state.
When calm cells stay calm
To explore these possibilities, the researchers studied primary microglia grown from mouse brains. They first looked at microglia in a resting, homeostatic state and exposed them to Wnt‑3a alone. Under these calm conditions, Wnt‑3a did not increase the release of key inflammatory molecules such as TNF‑α or IL‑1β. Even though some signaling inside the cells was activated, the microglia did not shift into a strongly inflammatory mode. This suggests that in a healthy, non‑inflamed brain, adding Wnt‑3a by itself may have little direct impact on microglial inflammatory output.
When inflamed cells are pushed further
The picture changed dramatically once the microglia were first triggered into an inflammatory state using bacterial lipopolysaccharide (LPS), a standard laboratory tool for mimicking infection. As expected, LPS alone drove a robust increase in TNF‑α production. But when Wnt‑3a was added together with LPS, microglia released significantly more TNF‑α than with LPS alone, while IL‑1β did not increase further. Detailed measurements showed that this boost in TNF‑α did not come from greater activation of the usual NFκB inflammatory route or from extra activation of the canonical β‑catenin pathway. Blocking the β‑catenin route with a protein called DKK1 left the Wnt‑3a‑driven TNF‑α surge intact, indicating that the expected “anti‑inflammatory” pathway was not responsible.
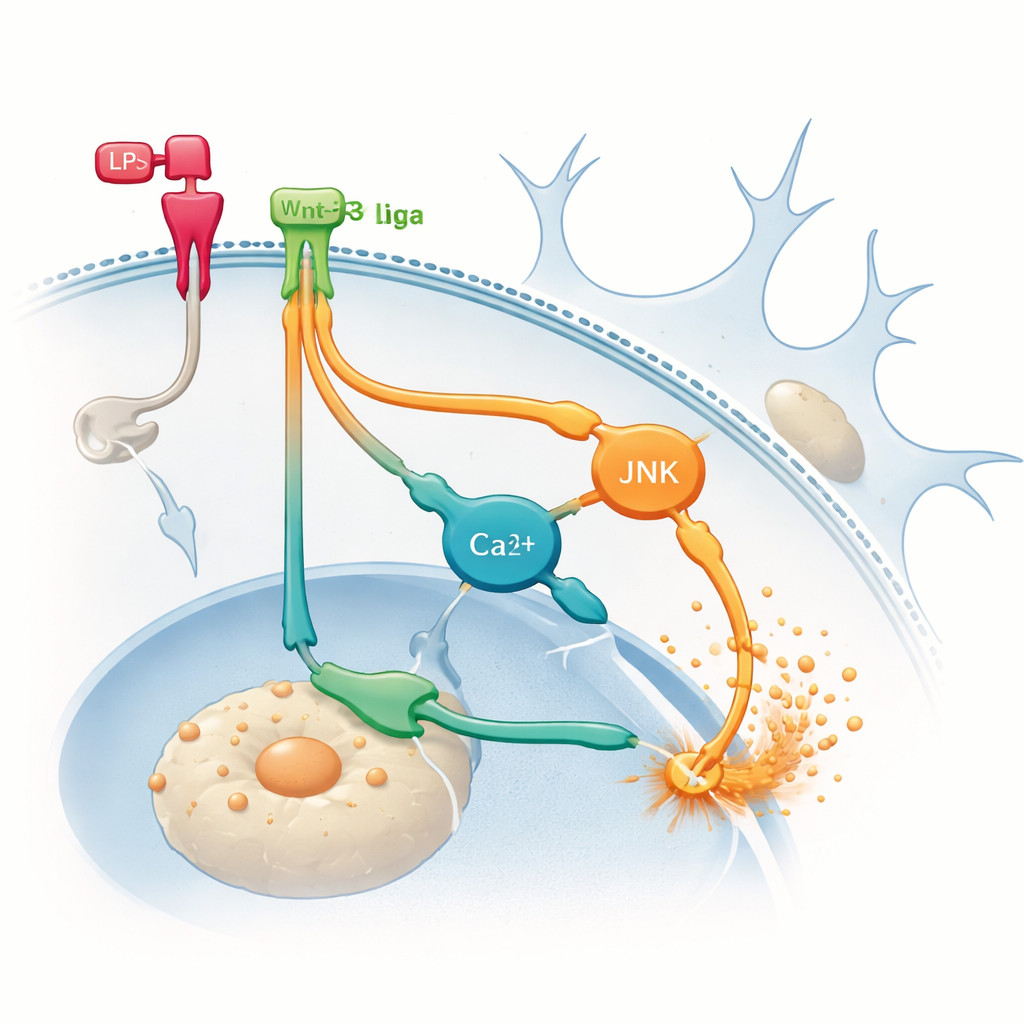
Hidden routes that fan the flames
To find where the extra inflammation was coming from, the team turned to the alternative non‑canonical routes that Wnt proteins can use. They pharmacologically blocked two signaling branches inside microglia: one involving a protein called JNK, and another tied to changes in calcium levels within the cell. Inhibiting either branch reduced TNF‑α production induced by LPS and, importantly, prevented Wnt‑3a from further boosting TNF‑α. These results suggest that, once microglia are already inflamed, Wnt‑3a can switch to predominantly using non‑canonical routes that amplify inflammatory signals rather than calming them. In other words, the same messenger can take a different internal path depending on the cell’s starting state, with very different outcomes.
Testing in a Parkinson’s model
The researchers then asked whether this behavior of Wnt‑3a in microglia would translate into bigger effects on neurons in a living brain. They infused Wnt‑3a directly into the brain ventricles of mice receiving the toxin MPTP, a widely used model of Parkinson’s‑like damage to dopamine neurons and microglial activation. As expected, MPTP reduced the number of dopamine neurons and increased microglial numbers in a midbrain region critical for movement. However, extra Wnt‑3a did not further worsen neuron loss or significantly change microglial counts compared with vehicle‑treated animals. In this complex in vivo setting, Wnt‑3a neither rescued nor clearly aggravated the toxin’s damaging effects over the timeframe studied.
What this means for future therapies
Overall, the study shows that Wnt‑3a is not a simple on–off switch for brain inflammation. In quiet microglia it has little effect, but in already inflamed cells it can selectively crank up the release of TNF‑α through non‑canonical signaling routes, independent of the classical β‑catenin pathway. At the same time, adding Wnt‑3a in a Parkinson’s‑like mouse model did not obviously protect or further harm dopamine neurons in the short term. For people interested in future treatments, the key message is that targeting Wnt signaling will require a careful appreciation of cellular context. Therapies based on Wnt‑3a or related molecules may help in some situations but could backfire in others if they inadvertently intensify harmful inflammation.
Citation: Federici, G., Stayte, S., Rentsch, P. et al. Wnt-3a exacerbates production of TNF-α in LPS stimulated microglia independent of the β-catenin canonical pathway. Sci Rep 16, 8222 (2026). https://doi.org/10.1038/s41598-026-37653-1
Keywords: brain inflammation, microglia, Wnt signaling, Parkinson’s disease, TNF-alpha