Clear Sky Science · en
Enhanced meropenem activity by a microbiome derived peptide targeting oxacillinase 48 carbapenemase in carbapenem resistant Klebsiella pneumoniae isolates
Why this matters for everyday health
Antibiotic resistance is making once-treatable infections harder and sometimes impossible to cure, especially in hospitals. This study explores a new way to "re-arm" an existing last-resort antibiotic, meropenem, against a dangerous bacterium called Klebsiella pneumoniae that often infects intensive care patients. Instead of inventing a brand-new drug, the researchers test a small helper molecule, a peptide from the human microbiome, that disables one of the bacterium’s key resistance tools so meropenem can work again.
A stubborn hospital germ and its chemical shield
Klebsiella pneumoniae is a common cause of pneumonia, blood infections, and urinary tract infections in hospitalized patients. Some strains have become resistant to carbapenems, a powerful class of antibiotics that doctors normally save for severe, hard-to-treat cases. Many of these bacteria make a special enzyme called OXA-48, which acts like a chemical shield that breaks down carbapenem drugs before they can kill the microbe. In the Middle East, including Iran, OXA-48-producing strains are especially widespread, and treatment options are dwindling as resistance even to newer drug combinations appears. This has made OXA-48 a priority target for new therapies.
Designing a helper peptide from the human microbiome
The researchers previously used computer models to scout databases of natural human antimicrobial peptides—short protein fragments made by our own bodies and resident microbes—for candidates that might stick tightly to the OXA-48 enzyme. Through virtual docking and long molecular dynamics simulations, they refined these candidates and selected one, named M104, that was predicted to bind stably to the enzyme’s active site and block its function while remaining water-soluble and chemically stable. In this study, they put M104 to the test in the lab, combining it with meropenem against 20 patient-derived Klebsiella isolates: 10 that produced OXA-48 and were resistant to meropenem, and 10 that lacked carbapenemase enzymes and were drug-susceptible.
Making an old antibiotic work again
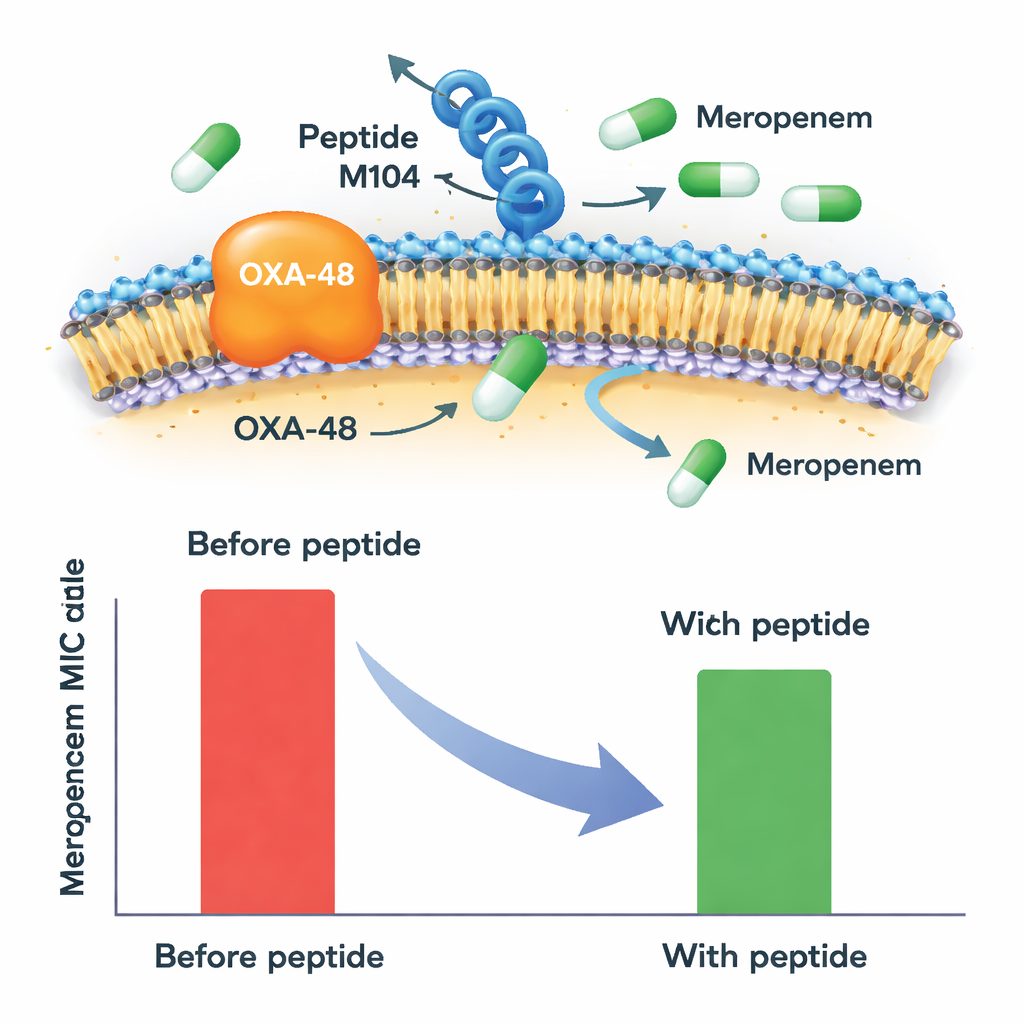
When meropenem was used alone, all OXA-48-positive Klebsiella isolates were clearly resistant. Adding peptide M104 changed that picture. At its highest tested level, M104 cut the amount of meropenem needed to stop bacterial growth by at least half—and in some cases by as much as six-fold—for every resistant OXA-48 producer tested. This pushed many strains from clearly resistant into an intermediate or even susceptible range, based on accepted clinical thresholds. Importantly, M104 had no killing effect on its own and did not boost meropenem when the bacteria lacked the OXA-48 enzyme, pointing to a very specific action on that resistance mechanism rather than a broad, nonselective toxicity.
Challenges with slimy biofilms and safety checks
Many hospital infections involve biofilms—slimy, layered communities of bacteria that cling to devices and tissues and are much harder to eradicate than free-floating cells. The team therefore tested whether M104 could help meropenem prevent biofilms from forming or clear those already in place for strong biofilm-forming Klebsiella strains. While the peptide-meropenem combination generally halved the drug amount needed to block biofilm growth or begin to clear mature biofilms, these effects were modest and did not reach clear statistical significance in this small sample. On the safety side, M104 showed no detectable damage to human red blood cells and did not reduce the viability of human skin fibroblast cells after 24 hours at a concentration higher than what was used in bacterial tests; only after 48 hours did cell survival drop by about 10 percent. Sequence comparisons also suggested little risk of the peptide accidentally targeting other bacterial or human proteins.
What this could mean for future treatments
Overall, the findings suggest that peptide M104 can specifically disarm the OXA-48 resistance enzyme and restore much of meropenem’s punch against otherwise untreatable Klebsiella pneumoniae, at least for bacteria growing in a free-floating state. Although its impact on tough, established biofilms was limited under the tested conditions, the study introduces the first reported peptide-based blocker of OXA-48 and demonstrates that such targeted helpers can be both effective and relatively gentle on human cells in the short term. With further testing in more bacterial strains, other carbapenem drugs, optimized formulations, and animal models, this kind of precision add-on therapy could help extend the useful life of our most powerful antibiotics rather than leaving doctors to face resistant infections with an empty toolbox.
Citation: Sadeghi, S., Faramarzi, M.A. & Siroosi, M. Enhanced meropenem activity by a microbiome derived peptide targeting oxacillinase 48 carbapenemase in carbapenem resistant Klebsiella pneumoniae isolates. Sci Rep 16, 7589 (2026). https://doi.org/10.1038/s41598-026-37644-2
Keywords: antibiotic resistance, carbapenem-resistant Klebsiella, OXA-48 enzyme, meropenem, peptide inhibitors