Clear Sky Science · en
The vasoprotective role of Myeloid-derived suppressor cells in pathogenesis of aortic dissection
Why this matters for your heart
Aortic dissection is a sudden tear in the body’s main blood vessel that can be rapidly fatal, even in otherwise active adults. Doctors know that weakened vessel walls and high blood pressure play a part, but the role of the immune system has been harder to pin down. This study explores an unexpected set of “peacekeeper” cells in the blood and spleen that may actually protect the aorta from tearing, suggesting new ways to predict, prevent, or soften this deadly condition.
The danger of a torn artery
The aorta is the body’s largest artery, carrying blood from the heart to every organ. In an aortic dissection, the inner lining of this vessel suddenly splits, and blood forces its way between the layers of the wall, creating a false channel. This can cut off blood flow to vital organs or cause the vessel to burst. While surgeons can sometimes repair the damage, many patients die before they reach the operating room. Beyond known risks like long-term high blood pressure and tissue weakness, scientists are increasingly asking how inflammation—our own immune response—might tip the balance toward such a catastrophic tear.
Meet the body’s immune “brakes”
Myeloid-derived suppressor cells, or MDSCs, are a group of immature immune cells best known for slowing down immune attacks in cancer and chronic infection. They act like brakes on the immune system, toning down aggressive reactions that might otherwise damage healthy tissue. In heart disease, growing evidence suggests these cells can be protective, easing harmful inflammation and limiting scarring. The authors of this study wondered whether MDSCs might also be at work in aortic dissection, not as villains, but as emergency responders trying to shield the aorta from runaway inflammation.
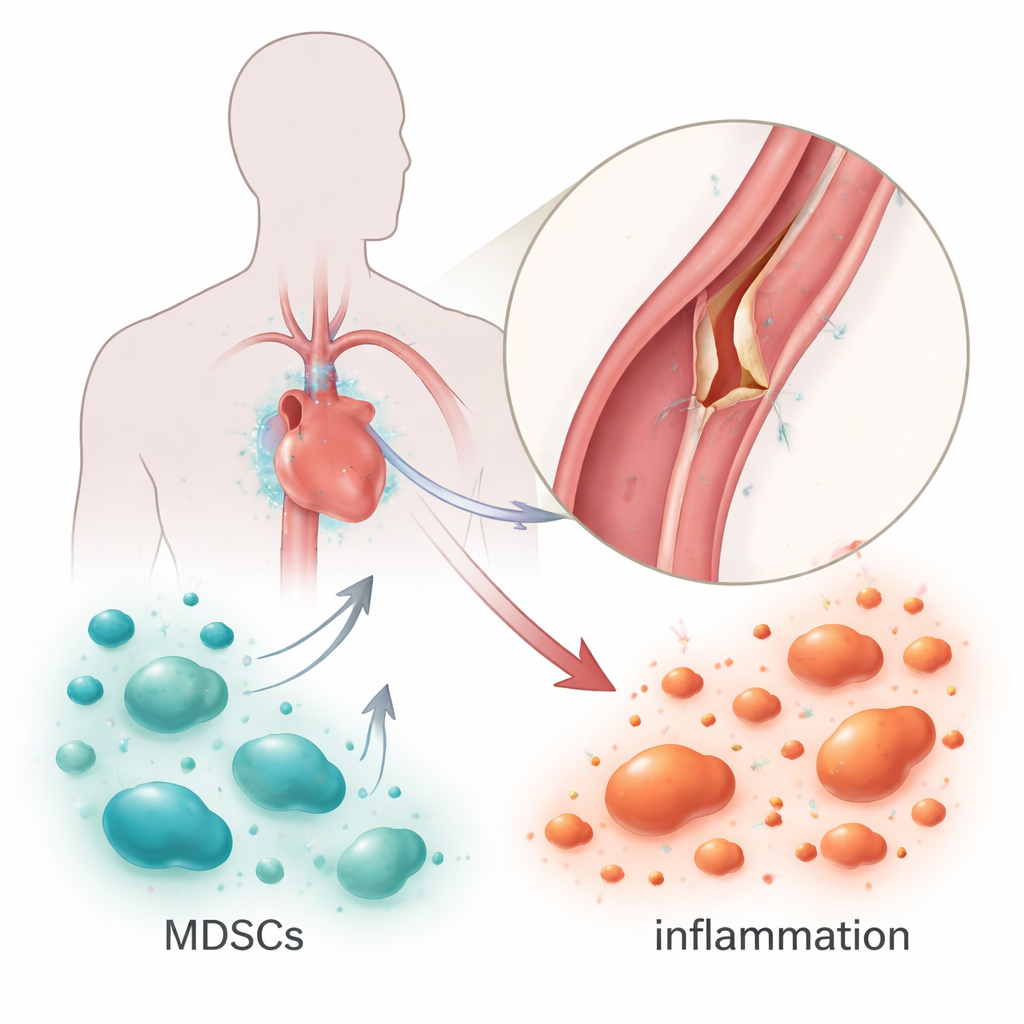
Building a tear in the lab
To investigate this, researchers created a mouse model of aortic dissection by weakening the vessel wall with a chemical in the drinking water and raising blood pressure using the hormone angiotensin II. Within 16 days, most of the mice developed classic signs of aortic dissection, including visible tears and separated layers in the aortic wall. The team then used flow cytometry, a technique for counting and classifying cells, to measure how many MDSCs were present in the blood and spleen. They found that a particular subtype, called monocytic MDSCs, expanded markedly in the spleen of mice with dissection, while levels in the circulating blood stayed about the same. This pattern suggests that these cells gather and become activated in immune organs rather than simply floating freely.
What happens when the “brakes” are cut
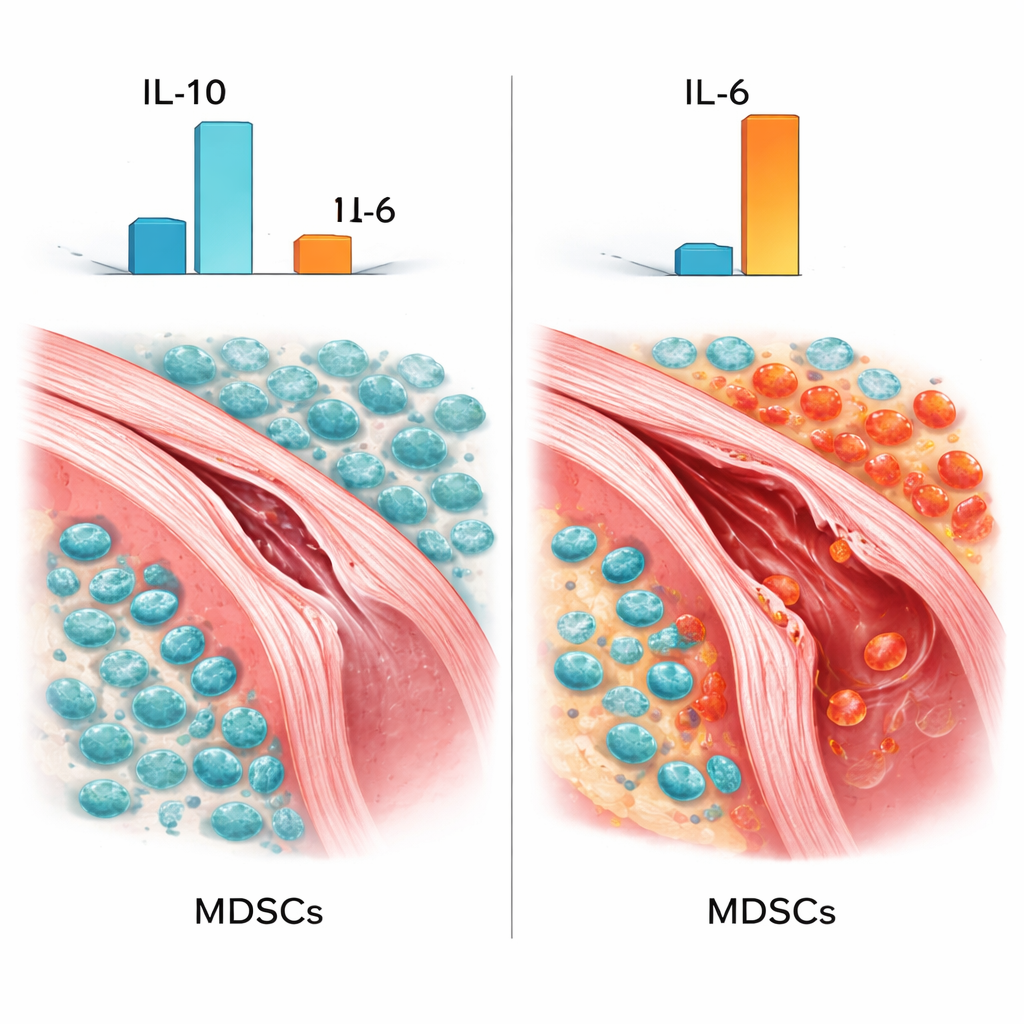
The crucial test was to see what happens if these protective cells are removed. Using the chemotherapy drug 5-fluorouracil, which selectively depletes fast-dividing cells including MDSCs, the researchers drastically reduced the number of MDSCs in the spleen. When they examined the aortas of these mice, the damage was far worse: tears were longer, the middle layer of the wall was more widely peeled apart, and more inflammatory cells had flooded into the tissue. Blood tests revealed that levels of IL-6, a pro-inflammatory signal linked to tissue injury, rose even higher, while IL-10, a calming, anti-inflammatory signal, dropped. In mice with intact MDSCs, both IL-6 and IL-10 were elevated, hinting that the body was mounting an inflammatory response while simultaneously trying to limit the damage. Removing MDSCs appeared to dismantle this protective counterweight.
A balancing act of signals
Further analysis showed that the number of MDSCs was strongly linked to the levels of these signaling molecules: more MDSCs went hand in hand with more IL-10 and less IL-6. This pattern fits with what has been seen in cancer and other diseases, where MDSCs help prevent immune overreaction by boosting soothing signals and damping inflammatory ones. The authors acknowledge that their model, based on chemically induced dissection in mice and a non-specific cell-killing drug, does not fully mirror human disease. Still, the consistent worsening of vessel injury when MDSCs were depleted, together with the shift toward a more damaging inflammatory profile, points to these cells as important guardians of the aortic wall.
What this means for future care
For non-specialists, the takeaway is that not all immune activity is helpful, and not all suppressive cells are harmful. In aortic dissection, this work suggests that a particular group of immune “brakes” helps keep inflammation in check, preserving the strength of the vessel wall. When those brakes fail or are removed, the aorta is more likely to tear badly. While the study was done in animals and human trials are still in the future, it raises the possibility that boosting or fine-tuning these protective cells—or the calming signals they release—could one day become part of strategies to prevent or treat aortic dissection.
Citation: Xu, Z., Wang, H., Lin, Z. et al. The vasoprotective role of Myeloid-derived suppressor cells in pathogenesis of aortic dissection. Sci Rep 16, 6231 (2026). https://doi.org/10.1038/s41598-026-37643-3
Keywords: aortic dissection, immune regulation, inflammation, myeloid-derived suppressor cells, vascular protection